10 Failure Mode and Effects Analysis Examples for Medical Devices
Introduction In the high-stakes realm of medical device development, ensuring patient...

In the complex landscape of medical device design, the challenge of ensuring patient safety is increasingly daunting. By employing Failure Mode and Effects Analysis (FMEA), manufacturers can proactively identify potential failure modes, thereby enhancing product reliability and compliance with safety standards. This evolution necessitates a reevaluation of existing practices to address new risks.
How can the latest advancements in FMEA methodologies be leveraged to safeguard against emerging risks in medical devices? This article delves into ten compelling examples of failure modes that underscore the critical importance of FMEA in enhancing medical device safety.
In the design of medical equipment, the absence of a systematic approach to identifying potential failures can jeopardize patient safety. Voler Systems employs Failure Mode and Effects Assessment as a fundamental element in this process. This systematic approach identifies failure mode examples, ensuring adherence to rigorous safety and efficacy standards for each unit.
Incorporating failure mode and effects analysis into the design process enhances product reliability and ensures compliance with regulatory requirements, prioritizing patient health. This proactive approach allows for early detection of issues, leading to timely interventions that significantly reduce risks.
Recent advancements in failure mode and effects analysis methodologies have further enhanced its application in medical equipment safety, enabling more thorough risk assessments and better outcomes. These advancements not only enhance safety but also improve overall patient outcomes in medical device usage.
Real-world failure mode examples illustrate how failure mode and effects analysis has effectively mitigated risks in various medical devices, reinforcing its importance in ensuring safety and compliance as the healthcare landscape evolves.
Identifying potential failures within vehicle systems is essential for ensuring safety and reliability in the automotive sector. Failure Mode and Effects Assessment plays a crucial role in this process. Producers utilize failure mode examples and effects analysis to meticulously examine essential elements like:
Evaluating each failure mode's impact on vehicle performance, including various failure mode examples, enables proactive risk management, significantly reducing accident risks. This approach enhances consumer trust and improves safety statistics in the automotive industry.
Recent applications of failure mode and effects analysis have demonstrated its effectiveness in addressing the complexities of modern vehicle architectures, particularly in the context of electrification and autonomous driving. This underscores its importance as a foundational tool for quality assurance in automotive design and manufacturing.
Moreover, the Automotive Failure Mode and Effects Analysis Software market is anticipated to attain $1.02 billion by 2033, expanding at a CAGR of 10.5%, indicating the rising dependence on failure analysis methodologies. Key applications include:
These are important failure mode examples for early-stage risk identification and manufacturing risk control, vital for ensuring compliance with regulatory standards. However, addressing challenges such as data privacy concerns and integration complexities with legacy systems is vital for manufacturers to fully leverage the benefits of failure analysis methodologies.
Failure Mode and Effects Analysis plays a critical role in enhancing aircraft safety within the aerospace sector. By identifying potential failure mode examples in critical systems such as navigation, propulsion, and communication, manufacturers can implement corrective actions before issues arise. Without failure mode and effects analysis, critical safety issues may go unaddressed, leading to severe consequences. This proactive strategy ensures compliance with aviation regulations and protects passengers and crew, highlighting the critical role of failure mode and effects analysis in maintaining safety standards. The absence of such analysis could lead to catastrophic failure mode examples, highlighting its importance in aviation safety.
Wearable health monitors face significant challenges in ensuring user safety due to potential failure mode examples that can compromise their effectiveness. These devices, including fitness trackers and medical alert systems, utilize Failure Mode and Effects Assessment to proactively identify possible failure mode examples that could endanger user safety. Manufacturers can enhance the design and functionality of wearable devices by evaluating key factors like sensor accuracy, battery longevity, and data transmission dependability.
Voler Systems emphasizes essential strategies for preventing overheating in wearable technology, which is crucial given the rising concerns highlighted by incidents like the Fitbit recall. This risk assessment guarantees that wearables provide accurate health data and comply with regulations throughout the development process, thereby improving patient outcomes and user satisfaction.
Recent advancements in failure mode examples and effects analysis, particularly the integration of real-world evidence (RWE) in regulatory submissions, have refined evaluation processes. This allows manufacturers to understand failure rates better and enhance safety protocols. Consequently, wearables that include failure mode and effects analysis are less likely to experience critical failures, thereby increasing their reliability and accuracy in health monitoring.
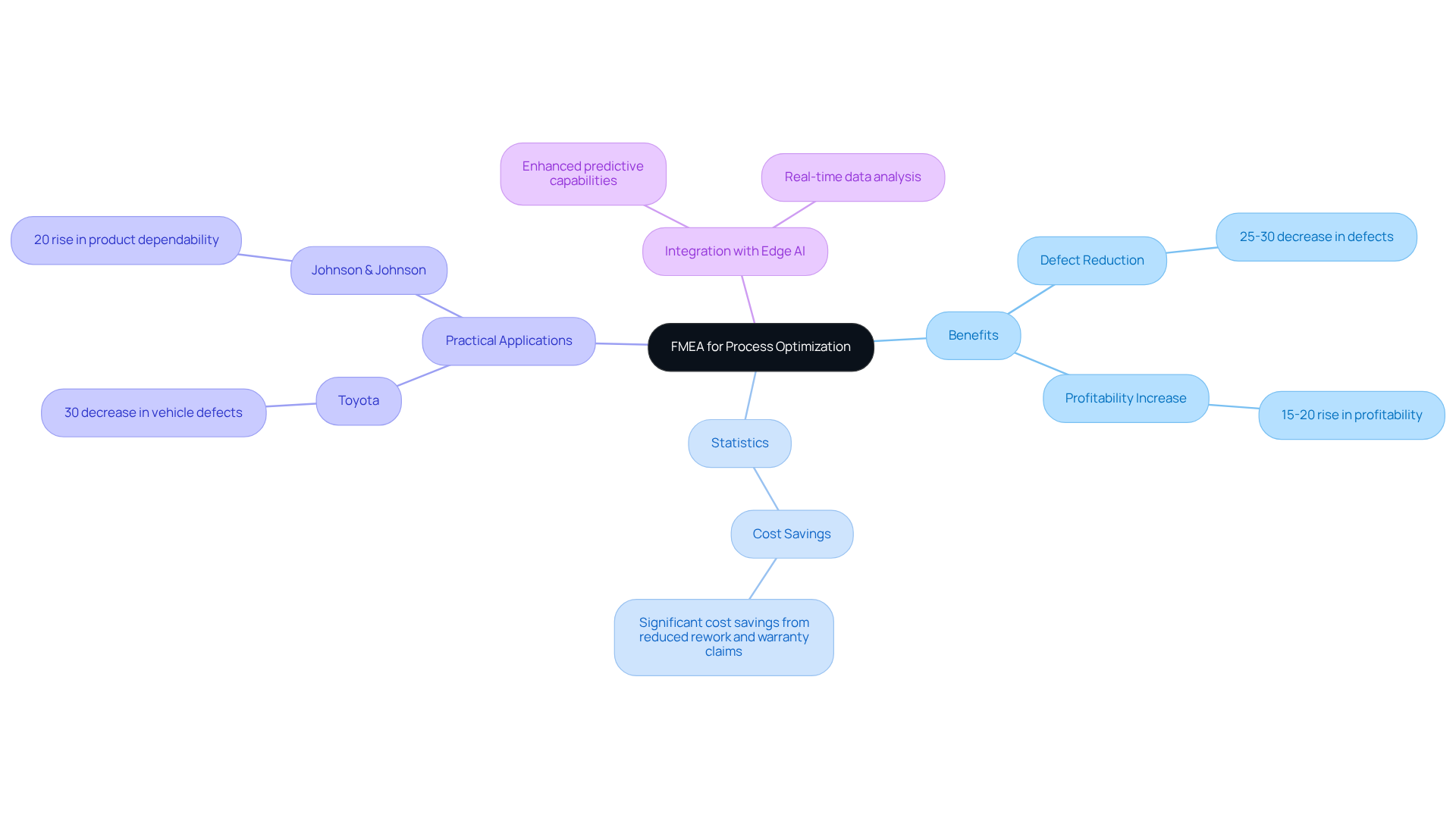
In the manufacturing sector, the implementation of Failure Mode and Effects Analysis (FMEA) is crucial for enhancing process efficiency and reducing defects. Manufacturers can identify potential failure mode examples by systematically assessing each step in the production line, which may lead to item defects or inefficiencies. This proactive approach enhances quality and substantially reduces costs associated with rework and waste.
Statistics indicate that applying FMEA can lead to a 25-30% decrease in defects, while organizations utilizing this method experience a 15-20% rise in profitability due to diminished risks associated with failure mode examples and reduced expenses related to warranty claims and recalls, underscoring its critical role in achieving operational excellence.
Practical applications, such as those observed in firms like Toyota, which achieved a 30% decrease in vehicle defects, and Johnson & Johnson, which noted a 20% rise in product dependability, illustrate how FMEA has effectively lowered defect rates and enhanced product reliability.
By incorporating Edge AI into their FMEA processes, manufacturers, including those collaborating with Voler Systems, can further improve their quality management systems, promoting a culture of ongoing enhancement and ensuring that potential issues are tackled before they develop into expensive problems. This strategic integration not only enhances quality management but also solidifies Voler Systems' position at the forefront of medical device innovation.
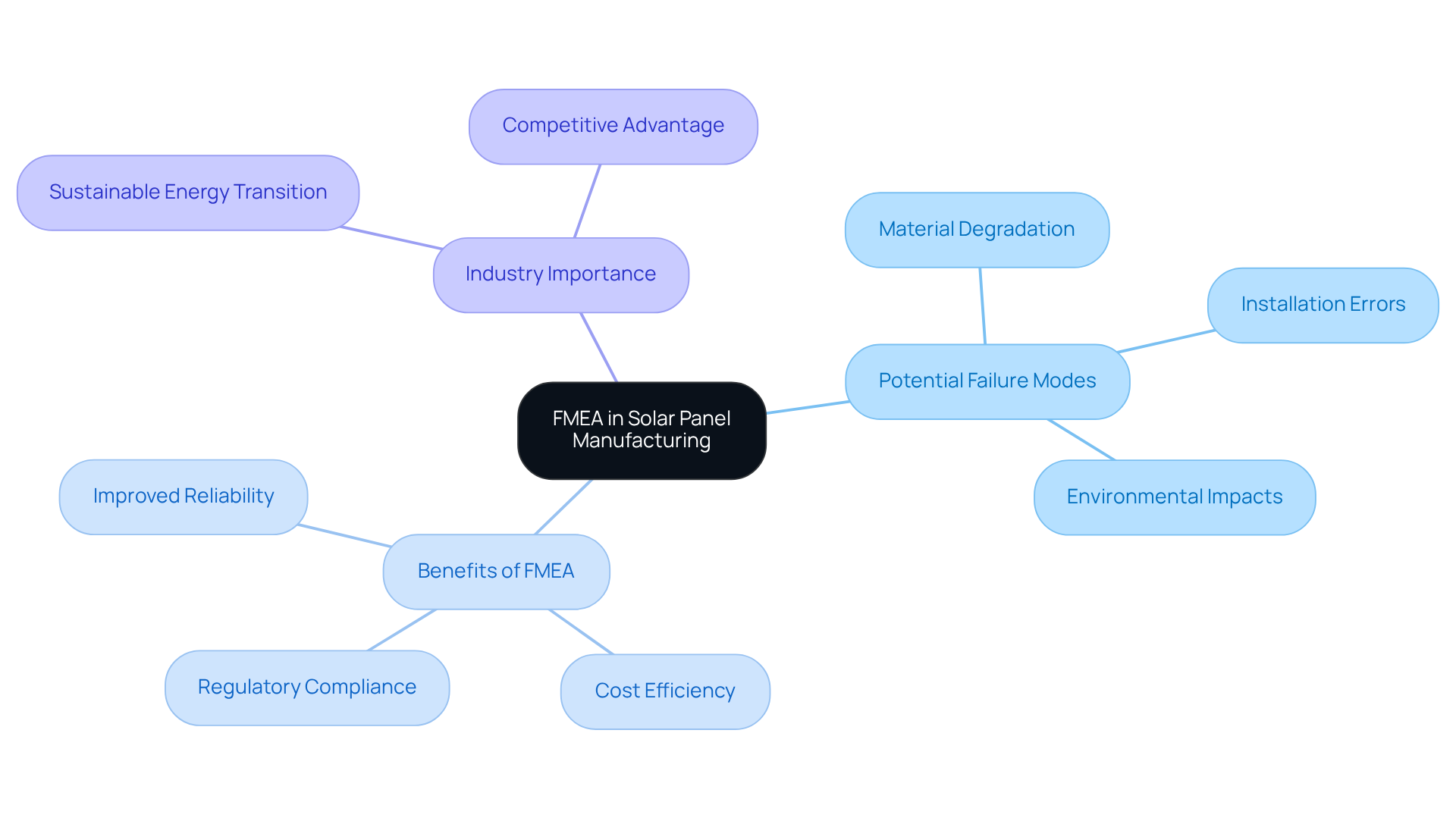
In the renewable energy sector, particularly in solar panel manufacturing, the implementation of Failure Mode and Effects Analysis (FMEA) is essential for enhancing product reliability and efficiency. By systematically identifying potential failure mode examples, such as:
manufacturers can significantly improve the reliability and efficiency of solar panels. This proactive approach enhances performance and supports the transition to sustainable energy solutions. For instance, recent applications of FMEA in solar technology have led to improved designs that mitigate risks associated with severe weather conditions, thereby extending the lifespan and efficiency of solar installations.
As the industry faces increasing demands for renewable energy, the importance of FMEA in ensuring high-quality, durable solar products cannot be overstated. Without FMEA, manufacturers risk operational inefficiencies and increased costs due to the presence of failure mode examples. Implementing FMEA not only improves product reliability but also ensures compliance with evolving regulatory standards, positioning manufacturers as leaders in the competitive renewable energy market.
In the consumer electronics industry, the absence of a systematic approach to failure analysis can lead to significant reliability issues. Manufacturers can identify weaknesses that may lead to malfunctions by systematically examining potential failure mode examples in smartphones, laptops, and home appliances. This proactive risk management approach enhances product quality and significantly boosts customer satisfaction and brand loyalty.
For instance, organizations that have adopted failure mode and effects analysis have reported a reduction in defect rates, achieving decreases in lot reject rates from 5,500 parts per million (PPM) to as low as 900 PPM. These enhancements demonstrate how failure mode examples in failure mode and effects analysis uphold high standards and reliability in consumer electronics, fostering trust and loyalty among consumers.
For medical equipment manufacturers, especially those creating wearable sensors, incorporating FMEA into the design process is essential. Voler Systems highlights the significance of comprehending user behavior and environmental elements, ensuring that equipment operates dependably across various contexts.
By utilizing AI-powered engineering development, Voler Systems aids precise sensing and smooth data communication, further improving the dependability and efficiency of medical instruments. Ultimately, neglecting FMEA could jeopardize not only product reliability but also the trust that consumers place in medical devices.
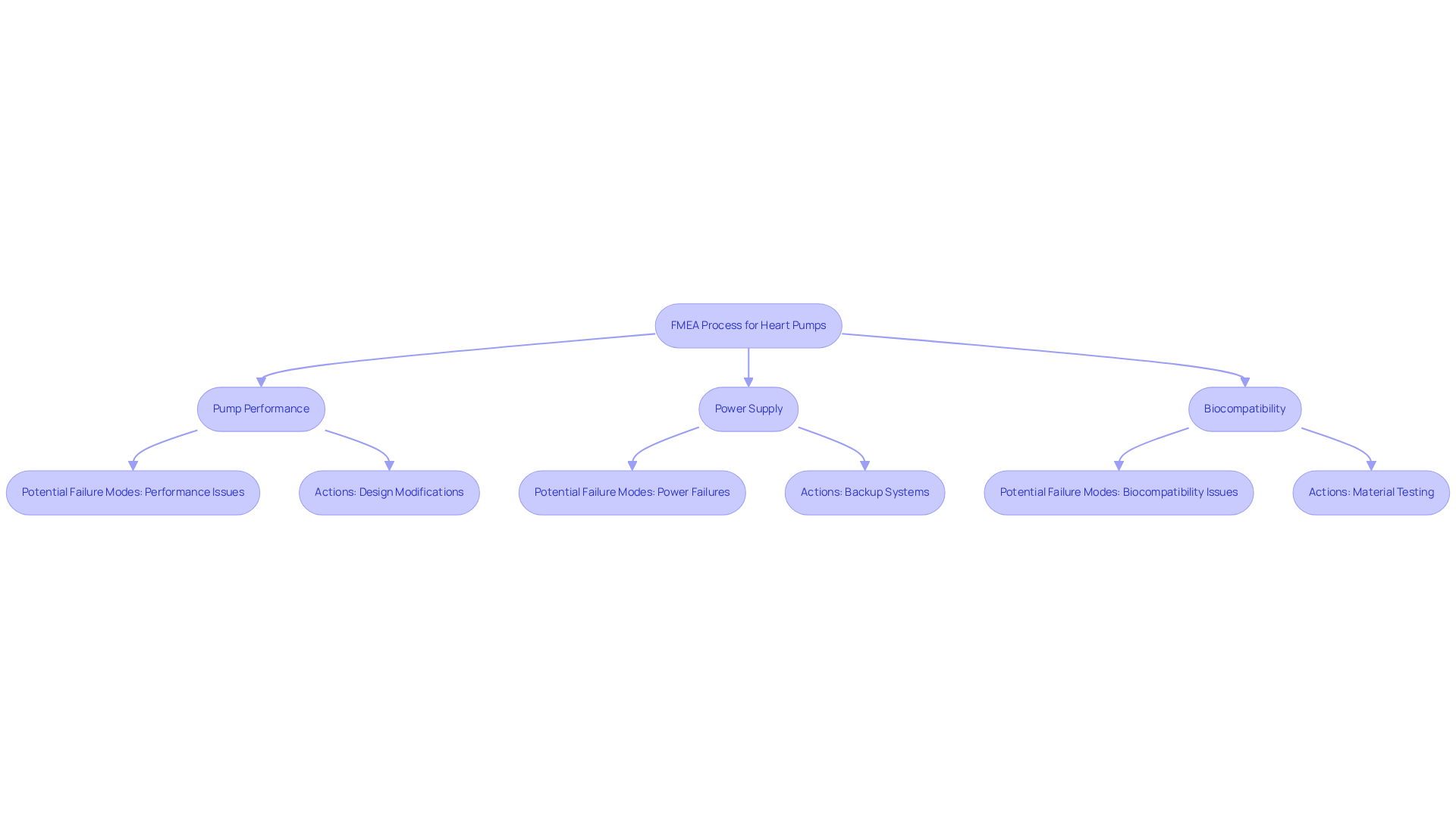
In the design and development of heart pumps, ensuring patient safety is paramount, making FMEA a critical process. By systematically identifying potential failure mode examples related to:
manufacturers can implement design modifications that significantly enhance reliability. This thorough analysis ensures compliance with regulatory standards and protects patients from harm.
Voler Systems provides essential documentation compliance assistance for medical apparatus startups, including guidance on user manuals and documentation practices, helping them navigate the complex regulatory landscape effectively.
Recent FDA alerts regarding Impella heart pumps underscore the necessity of analyzing failure mode examples to mitigate risks associated with equipment malfunctions. The FDA reported serious injuries linked to differential pressure sensor drift in 2.5% of Impella RP cases, highlighting the consequences of inadequate failure mode examples in the analysis.
Ultimately, prioritizing failure mode examples and effects analysis is essential for the advancement of safe and effective heart pump technologies.
In the rapidly evolving landscape of smart home technology, the potential for failure poses significant challenges to user safety and experience. By systematically identifying potential failure mode examples related to:
manufacturers can create products that are not only functional but also secure. This proactive approach mitigates risks, ensuring products align with consumer expectations and regulatory standards. Neglecting these practices can lead to project delays and increased costs.
Robust testing systems are essential for ensuring quality and reliability in electronic device design. Recognizing common manufacturing test errors enhances quality and efficiency, leading to better development outcomes.
In an era where technological advancements are rapid, the need for regular updates to Failure Mode and Effects Analysis procedures is paramount for effective risk management. As new technologies arise and existing items advance, revisiting and modifying FMEA analyses becomes crucial to identify potential new failure mode examples. This continuous evaluation enhances safety and dependability. It also guarantees adherence to changing regulatory standards, especially in the medical equipment sector, which must comply with strict regulations such as ISO 14971.
Voler Systems provides expert documentation compliance assistance tailored for medical equipment startups, including the creation of user manuals that fulfill regulatory requirements. Notably, 67% of manufacturers utilizing FMEA software for risk prevention have reported improved item consistency and a reduction in defects, underscoring the methodology's effectiveness as a dynamic tool that improves safety and operational performance.
Furthermore, Voler Systems has significant experience supporting legacy test equipment for medical product companies, ensuring that even older technologies meet current compliance standards. Organizations that utilize failure mode examples in their product development processes can significantly reduce defects and enhance reliability, ultimately resulting in safer medical devices and improved patient outcomes. By neglecting to adapt FMEA processes, organizations risk not only their compliance but also the safety of the patients they serve.
Failure Mode and Effects Analysis (FMEA) serves as a cornerstone in ensuring safety across industries, particularly in the medical device sector. By systematically identifying potential failure modes, organizations can proactively address risks, ensuring that products meet both safety and regulatory standards. This approach not only protects patients but also fosters trust in medical technologies, which is vital for maintaining compliance and ensuring patient safety in a rapidly changing environment.
Throughout the article, various sectors including automotive, aerospace, renewable energy, and consumer electronics have been discussed, highlighting how FMEA contributes to improved safety, reliability, and operational efficiency. Each industry showcases real-world examples of how effective risk management practices lead to enhanced product performance and reduced defect rates. The integration of advanced methodologies and continuous updates to FMEA processes further solidify its importance in adapting to evolving technologies and regulatory requirements.
Manufacturers must implement FMEA in their product development processes to prioritize safety and compliance. As industries face increasing demands for innovation and reliability, adopting comprehensive failure analysis strategies will be crucial. Organizations must commit to ongoing risk assessment and improvement, ensuring that they not only meet current standards but also position themselves as leaders in quality and innovation.
What is Failure Mode and Effects Analysis (FMEA) and why is it important in medical device design?
FMEA is a systematic approach used to identify potential failures in medical equipment design. It is important because it helps ensure adherence to safety and efficacy standards, enhances product reliability, and prioritizes patient health by allowing for early detection of issues.
How does FMEA contribute to patient safety in medical devices?
FMEA contributes to patient safety by enabling proactive risk management, which allows for timely interventions that significantly reduce risks associated with medical devices.
What recent advancements have been made in FMEA methodologies for medical equipment?
Recent advancements in FMEA methodologies have improved risk assessments and outcomes in medical equipment safety, leading to enhanced overall patient outcomes.
How is FMEA applied in the automotive industry?
In the automotive industry, FMEA is used to identify potential failures in vehicle systems such as brakes, steering mechanisms, and electronic systems, enabling proactive risk management and enhancing safety and reliability.
What impact does FMEA have on consumer trust in the automotive sector?
By significantly reducing accident risks through meticulous evaluation of failure modes, FMEA enhances consumer trust and improves safety statistics in the automotive industry.
What is the projected growth of the Automotive Failure Mode and Effects Analysis Software market?
The Automotive Failure Mode and Effects Analysis Software market is anticipated to reach $1.02 billion by 2033, expanding at a CAGR of 10.5%.
What are some key applications of FMEA in the automotive industry?
Key applications of FMEA in the automotive industry include Design Failure Mode Effects Analysis and Process Failure Mode Effects Analysis, which are vital for early-stage risk identification and manufacturing risk control.
What role does FMEA play in the aerospace industry?
In the aerospace industry, FMEA is critical for enhancing aircraft safety by identifying potential failures in systems like navigation, propulsion, and communication, allowing manufacturers to implement corrective actions before issues arise.
What could happen if FMEA is not utilized in aviation safety?
Without FMEA, critical safety issues may go unaddressed, potentially leading to catastrophic failures and severe consequences for passengers and crew.
How does FMEA ensure compliance in the aerospace sector?
FMEA ensures compliance with aviation regulations by identifying and addressing potential failure modes, which helps maintain safety standards in aircraft design and operation.
