Understanding the Software Product Development Process for Medical Devices
Introduction Understanding the software product development process for medical devices...

Navigating the complex landscape of medical device design necessitates a thorough understanding of regulatory standards, particularly the IEC 62304 checklist. This essential framework guides manufacturers in developing safe and effective software for healthcare products while underscoring the critical processes that ensure compliance. With evolving regulations and increasing market demands, organizations must consider how to effectively implement these requirements to avoid costly pitfalls. This article explores ten vital components of the IEC 62304 checklist, providing insights and strategies that empower manufacturers to streamline their compliance efforts and enhance product quality.
Since its founding in 1979, has established itself as a leader in electronic product design, particularly within the , where adherence to the 62304 checklist is critical. The organization boasts a team of seasoned engineers, each possessing over 20 years of experience, who provide essential support in navigating the complexities of .
The is recognized as a consensus standard by , underscoring its importance; it ensures that software used in healthcare products is developed through a comprehensive, risk-oriented process. Recent data indicates that a significant proportion of healthcare equipment companies are achieving compliance with the 62304 checklist, further emphasizing its role in enhancing .
capitalizes on its extensive knowledge of these regulatory standards to assist clients in , ensuring that their products are not only safe but also ready for market. This commitment to quality and precision positions as a trusted partner in the healthcare industry, facilitating successful navigation through the .
' role in compliance, then explore the branches to understand the company's background, the significance of the IEC 62304 checklist, and the advantages of achieving compliance. Start at the center with Voler Systems' role in compliance, then explore the branches to understand the company's background, the significance of the IEC 62304 checklist, and the advantages of achieving compliance.](https://images.tely.ai/telyai/ddhqtbwo-start-at-the-center-with-voler-systems-role-in-compliance-then-explore-the-branches-to-understand-the-companys-background-the-significance-of-the-iec-62304-checklist-and-the-advantages-of-achieving-compliance.webp)
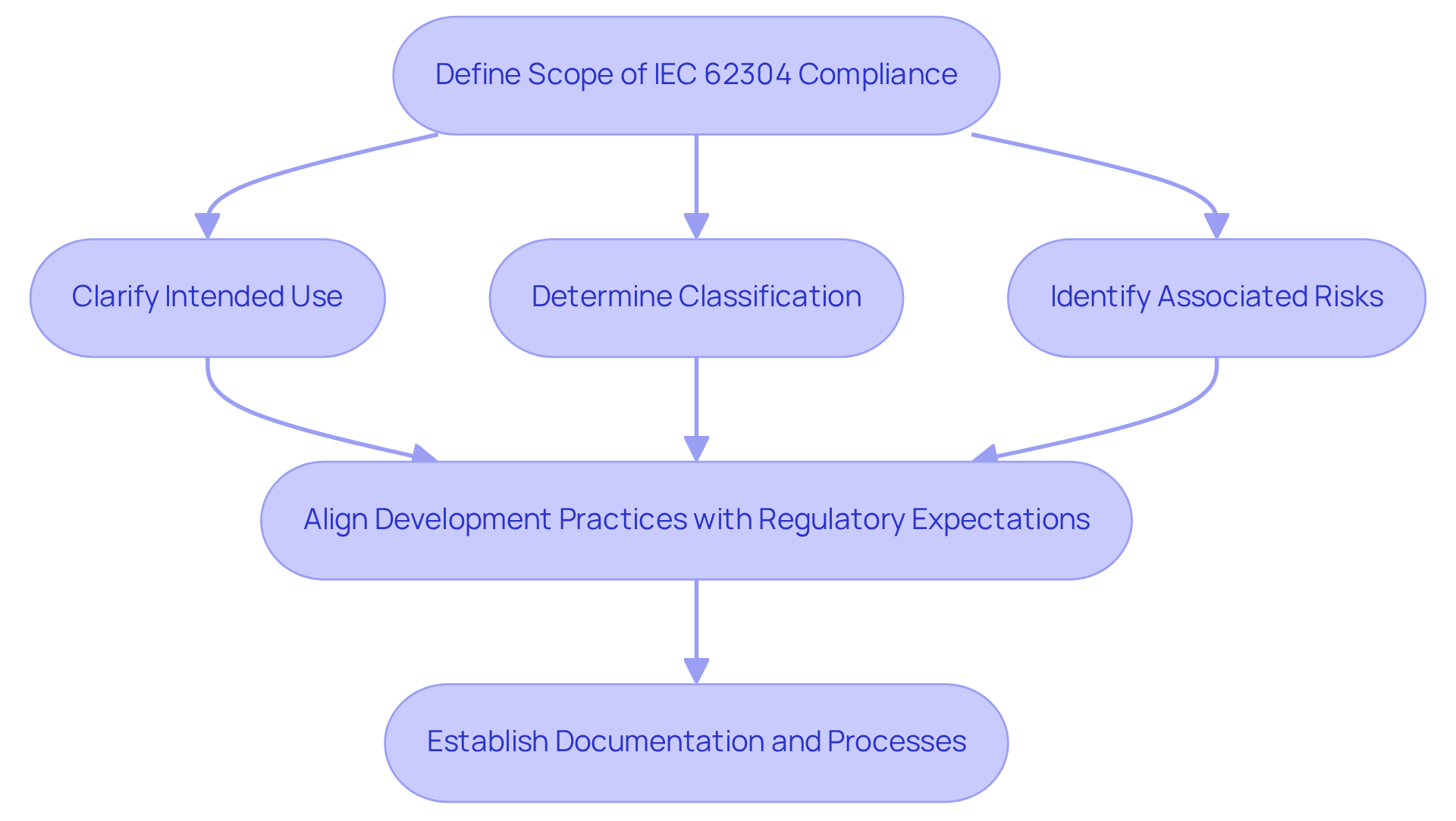
The outlines the life cycle processes for medical device applications, which include development, maintenance, and . Establishing an early definition of the is crucial, as it clarifies the software's intended use, classification, and associated risks. This proactive clarity allows teams to align their development practices with , ensuring that all necessary documentation and processes are established from the outset. By addressing these elements early, manufacturers can streamline regulatory efforts, mitigate the risk of costly revisions later, and enhance overall . Moreover, with the updated IEC 62304 introducing more sophisticated risk-based approaches, the importance of the for early scope definition becomes even more pronounced.
provides essential , assisting healthcare product startups in navigating these regulatory challenges. This support includes guidance on user manuals and actionable steps in the , ensuring that their innovative technologies comply with the necessary standards for safety and efficacy.
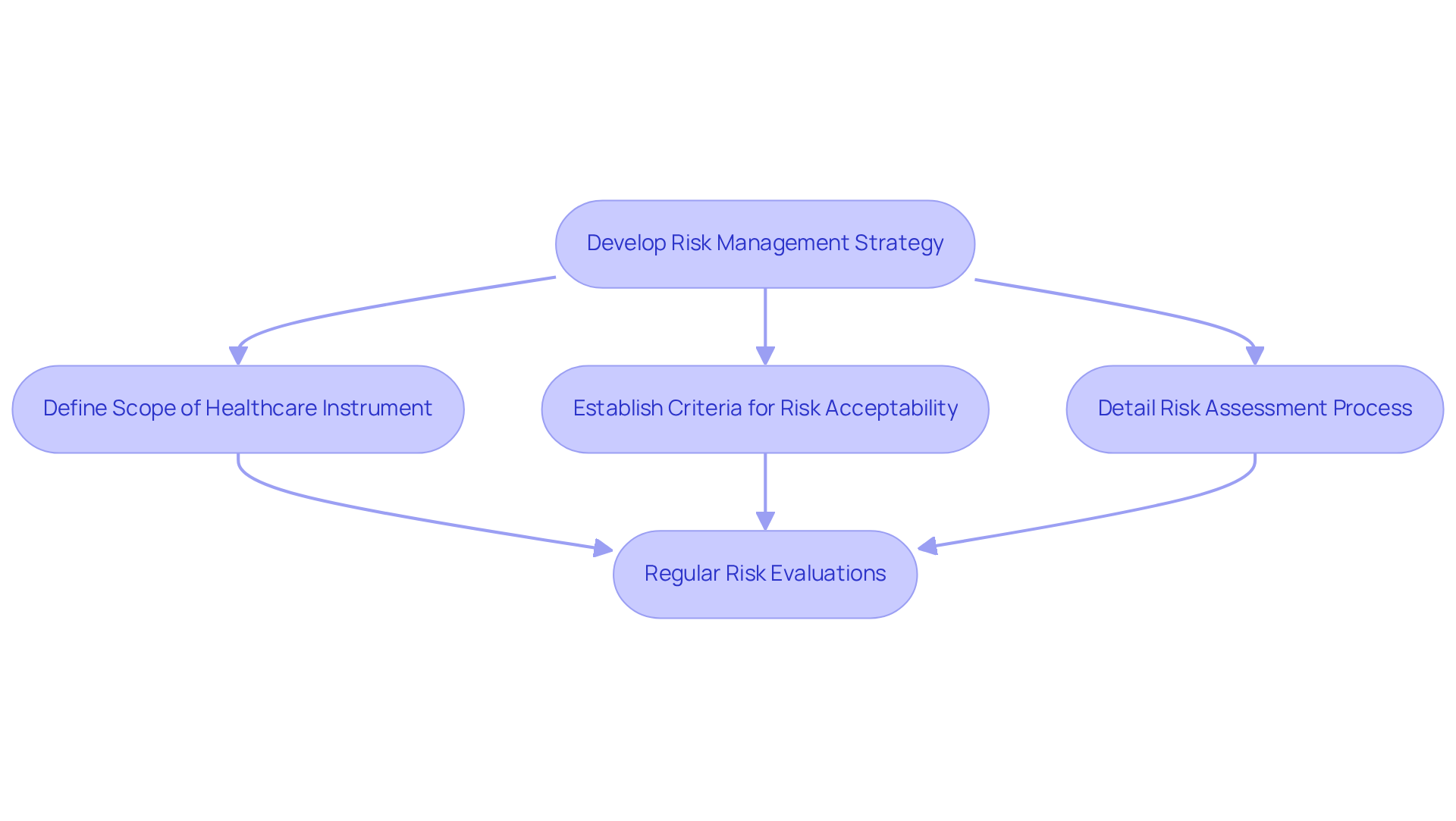
serves as a cornerstone of IEC 62304 compliance, requiring manufacturers to systematically identify, assess, and mitigate risks associated with their software. This process begins with the development of a comprehensive that aligns with , which outlines the framework for in s. Key components of this strategy include:
Regular risk evaluations are essential throughout the , ensuring that potential hazards are promptly identified and effectively managed. This proactive approach not only enhances but also supports compliance with , ultimately facilitating the successful market entry of health equipment. Furthermore, the integration of into risk assessments is vital for validating initial assumptions and uncovering new hazards, thereby reinforcing the ongoing nature of effective .
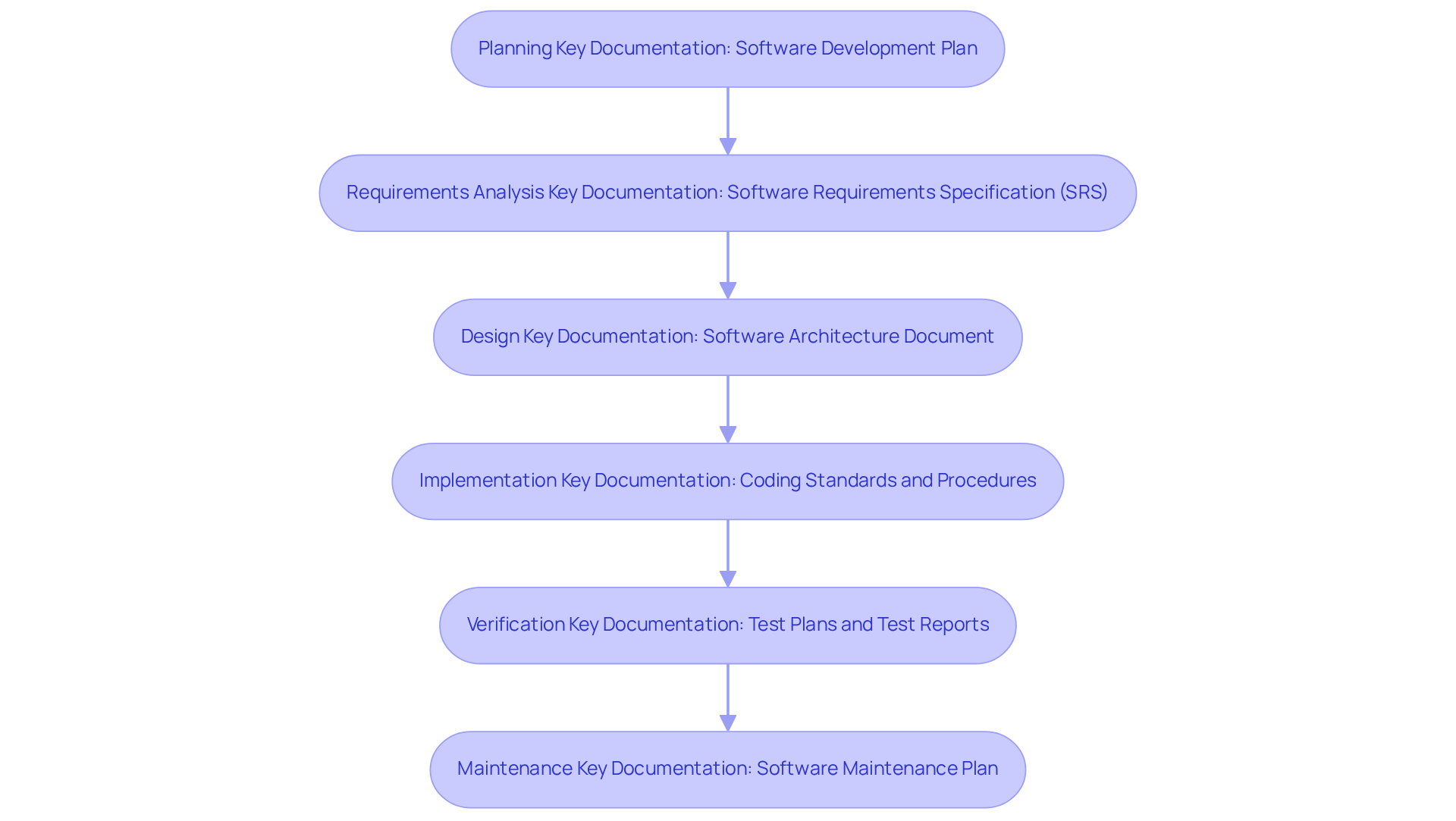
The 62304 checklist mandates (SDLC) for . This includes critical phases such as planning, requirements analysis, design, implementation, verification, and maintenance. Each phase must be meticulously documented to establish a transparent audit trail that demonstrates compliance with regulatory standards.
Key documentation includes a , which outlines the processes, deliverables, and timelines for each development stage. This rigorous approach not only fosters compliance but also enhances the overall quality and reliability of . As we approach 2026, maintaining such documentation becomes increasingly vital, as it directly influences and market readiness.
Recent statistics reveal that approximately 70% of healthcare technology firms prioritize , acknowledging its significance in ensuring product safety and effectiveness. To assist startups in this endeavor, offers tailored , ensuring that all regulatory requirements are met efficiently. Strong documentation practices are not merely a regulatory obligation; they are a fundamental component of .
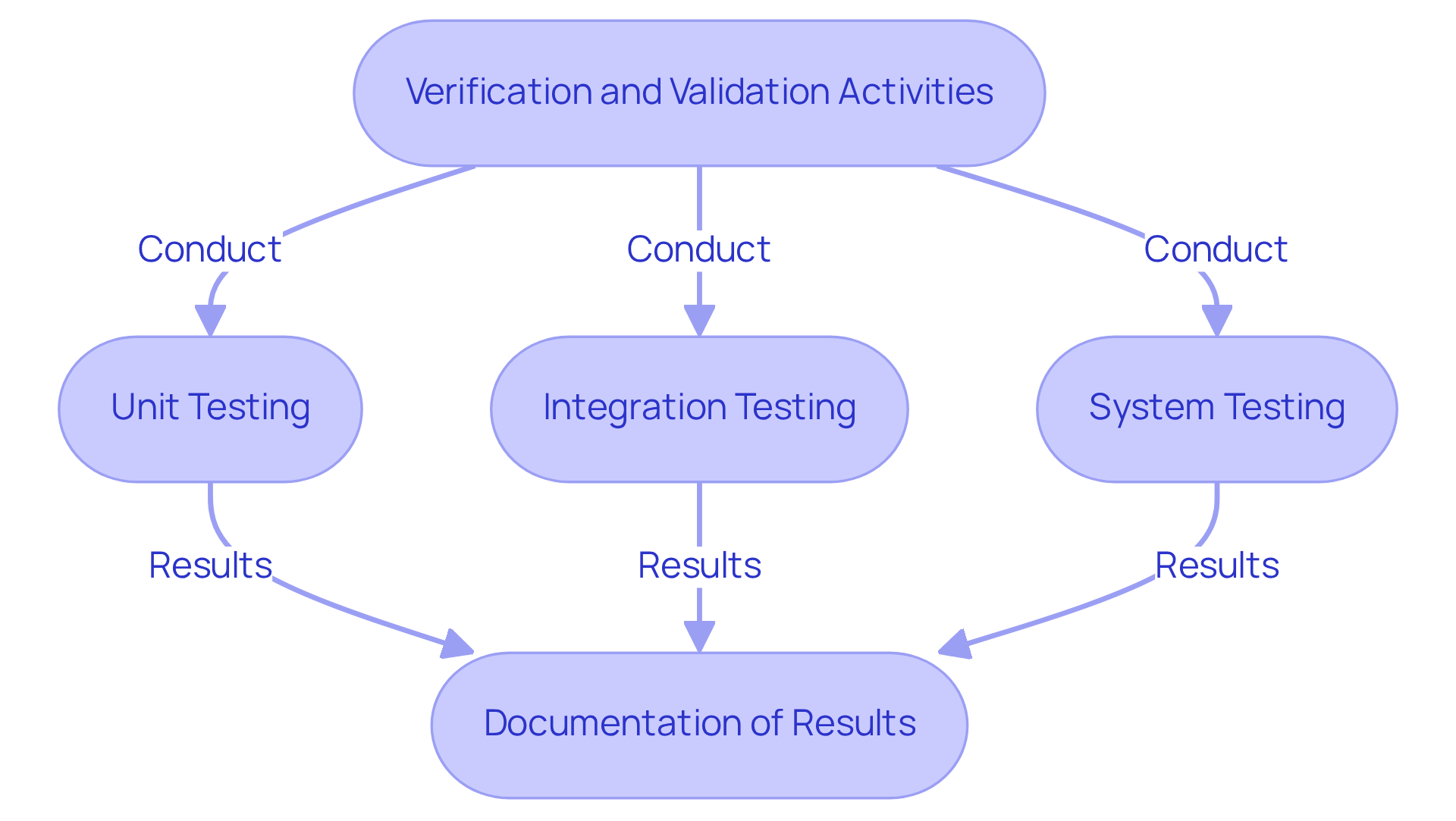
(V&V) are critical components of the . Verification ensures that software meets , while validation confirms that it fulfills its intended use in real-world scenarios. Producers should establish a that includes:
Along with of all results to ensure during audits. The significance of robust testing systems cannot be overstated, as they are vital for ensuring the . Furthermore, offers comprehensive documentation compliance support, which is particularly advantageous for startups in the medical technology sector, assisting them in navigating the complexities of compliance from prototype to production.
Traceability is a fundamental requirement of the , ensuring that every requirement is directly linked to its corresponding design and testing activities. This linkage is effectively documented through a , which outlines the relationships between user needs, system requirements, design specifications, and test cases.
By implementing an RTM, companies can uphold adherence to while significantly improving the overall . Studies indicate that over 70% of utilize an RTM to maintain this critical traceability.
To create an effective RTM for , it is essential to clearly define is traceable throughout the development lifecycle. Expert insights suggest that regular reviews and updates to the RTM are crucial for maintaining its accuracy and relevance, particularly as project specifications evolve.
This structured approach not only aids in meeting regulatory expectations but also within development teams.
Effective is essential for overseeing software changes and ensuring that all versions are thoroughly documented and traceable. The requires manufacturers to implement robust that include version control, change management, and build management. By maintaining comprehensive records of all changes and their justifications, manufacturers demonstrate compliance and facilitate the audit process. Currently, approximately 70% of device manufacturers have adopted , reflecting a growing recognition of their importance in enhancing product quality and safety.
Best practices in version control involve the implementation of , conducting regular audits, and ensuring that all team members are well-versed in . As highlighted in ' documentation, support for startups in the encompasses critical requirements such as tracking all software-related artifacts and adhering to change control procedures, which ensures that every modification is traceable and accountable.
Furthermore, with the expected growth of the software market, companies must prepare for ongoing compliance with the updates, underscoring the urgency of adopting effective version control practices. Real-world examples illustrate how efficient version control has led to improved compliance and reduced time to market, ultimately contributing to the development of safer healthcare products. For additional insights, refer to user manuals and case studies provided by , which detail successful implementations of these practices.
Frequent audits and evaluations are essential for maintaining compliance with the . These audits assess the effectiveness of current processes, identify gaps or non-conformities, and provide actionable recommendations for improvement. By establishing a routine audit schedule, manufacturers not only ensure but also promote the . Notably, 56% of organizations have reported identifying adherence gaps during regular audits, underscoring the critical role these evaluations play in ensuring conformity to .
With ' comprehensive , which includes , can effectively navigate these regulatory challenges. Furthermore, - such as integrating risk management and maintaining thorough documentation - bolster regulatory efforts and enhance overall product safety and reliability.
Training is crucial for ensuring that all team members comprehend the requirements of the 62304 checklist and their specific roles in achieving compliance. Manufacturers should implement a comprehensive that covers the principles of the standard, risk management practices, documentation requirements, and includes the 62304 checklist. Regular training sessions and workshops are vital for maintaining competency and keeping the team updated on changes to . Notably, a significant percentage of healthcare equipment manufacturers recognize the importance of this training, with many providing ongoing education on to enhance team proficiency in . This commitment to training fosters a culture of compliance and equips teams with the necessary skills to effectively navigate the complexities of .
With the set to be released in August 2026, it is imperative for manufacturers to align their s with these new regulations, particularly regarding and validation procedures. As Laura Friedl-Hirst states, "Manufacturers must evaluate any modifications, examine development plans, and ensure continuous adherence to the updated standard." This underscores the urgency of in the evolving landscape of healthcare device software.
plays a pivotal role in assisting manufacturers by providing extensive documentation adherence support, ensuring that teams are well-prepared to meet the challenges of innovative healthcare technology development.
Continuous enhancement serves as a cornerstone of quality management, particularly regarding adherence to the . To foster a culture of , manufacturers must:
This proactive approach not only bolsters adherence but also stimulates innovation and improves the overall quality of healthcare software.
Recent trends reveal that approximately 80% of healthcare equipment firms are adopting to comply with the , acknowledging its vital role in upholding high standards of safety and effectiveness. For instance, a case study involving illustrated how the introduction of a in a small healthcare startup led to significant improvements in and operational efficiency.
By prioritizing , medical device manufacturers can more effectively navigate the complexities of while advancing product development.
The significance of the IEC 62304 checklist in the design of medical devices is paramount. This vital framework not only guarantees compliance with regulatory standards but also bolsters the safety and efficacy of healthcare products. By comprehensively understanding and implementing the key components of this checklist, manufacturers can adeptly navigate the complexities of medical device development while cultivating a culture of quality and accountability.
This article has delved into essential aspects of the IEC 62304 checklist, including:
Furthermore, the importance of:
has been underscored as foundational elements for achieving and sustaining compliance.
Given the rapidly evolving landscape of healthcare technology, it is crucial for manufacturers to prioritize adherence to the IEC 62304 checklist. By adopting continuous improvement practices and utilizing expert guidance, such as that provided by Voler Systems, organizations can not only ensure compliance but also foster innovation and enhance product quality. A steadfast commitment to these practices will ultimately lead to the development of safer, more effective medical devices that fulfill the needs of both patients and healthcare providers.
What is Voler Systems and what expertise do they offer?
Voler Systems is a leader in electronic product design, particularly in the healthcare sector, providing expert guidance for IEC 62304 compliance. Their team consists of seasoned engineers with over 20 years of experience in navigating healthcare product development.
Why is the IEC 62304 checklist important?
The IEC 62304 checklist is a consensus standard recognized by regulatory bodies like the FDA. It ensures that software used in healthcare products is developed through a comprehensive, risk-oriented process, enhancing product safety and efficacy.
How does Voler Systems assist clients with IEC 62304 compliance?
Voler Systems leverages its knowledge of regulatory standards to help clients streamline their compliance efforts, ensuring their products are safe and market-ready. They provide support in documentation adherence and navigating regulatory challenges.
What does the IEC 62304 checklist outline regarding medical device applications?
The IEC 62304 checklist outlines life cycle processes for medical device applications, including development, maintenance, and risk management. It emphasizes the importance of defining the scope of adherence early to align development practices with regulatory expectations.
What is the significance of defining the scope of IEC 62304 compliance?
Defining the scope early clarifies the software's intended use, classification, and associated risks, allowing manufacturers to streamline regulatory efforts, mitigate costly revisions, and enhance overall product safety and effectiveness.
What role does risk management play in IEC 62304 compliance?
Risk management is a cornerstone of IEC 62304 compliance, requiring manufacturers to systematically identify, assess, and mitigate risks associated with their software, aligning their strategies with ISO 14971.
What are the key components of a risk management strategy under IEC 62304?
Key components include defining the scope of the healthcare instrument, establishing criteria for risk acceptability, and detailing the risk assessment process.
Why are regular risk evaluations important during the development lifecycle?
Regular risk evaluations are essential for promptly identifying and managing potential hazards, enhancing product safety, and supporting compliance with regulatory standards for successful market entry.
How does post-market data contribute to risk management?
Integrating post-market data into risk assessments helps validate initial assumptions and uncover new hazards, reinforcing the ongoing nature of effective risk management.
