Introduction
Mastering Failure Mode and Effects Analysis (FMEA) is essential for ensuring the safety and effectiveness of medical devices. This structured approach enables manufacturers to identify potential malfunctions early in design and production, fostering enhanced product reliability and compliance with stringent industry standards.
However, manufacturers often face challenges in determining the most effective FMEA method for their specific needs. This article explores the nuances of FMEA in manufacturing, offering insights and practical steps to navigate its implementation while uncovering the substantial benefits it presents for quality and compliance in the medical device sector.
Understand FMEA: Definition and Importance in Manufacturing
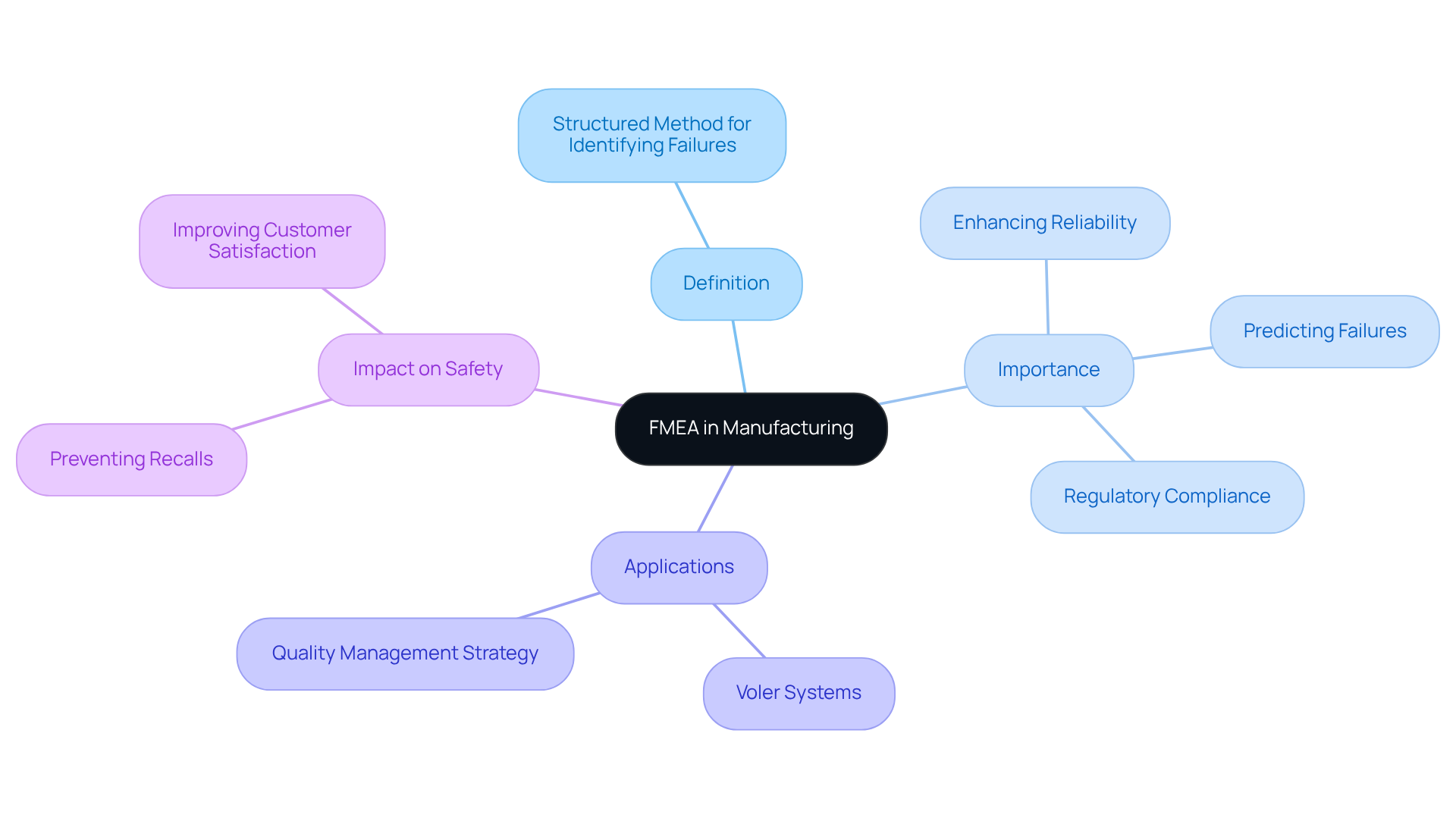
Recognizing potential malfunctions in medical devices is crucial for ensuring safety and effectiveness. Failure Mode and Effects Assessment is a structured method that identifies possible malfunction types within an item or procedure, evaluates their potential consequences, and ranks them according to seriousness and probability. In the field of medical equipment production, FMEA in manufacturing plays a vital role in predicting failures that could threaten safety and effectiveness. By utilizing FMEA in manufacturing, manufacturers, including Voler Systems, can implement corrective actions early in the design and production phases, significantly enhancing product reliability and ensuring compliance with regulatory standards.
FMEA in manufacturing ensures that medical devices meet high standards of precision and reliability through comprehensive evaluation. Addressing potential malfunctions early can prevent costly recalls and enhance overall product quality. Furthermore, this analysis is integral to Advanced Product Quality Planning (APQP) and incorporates FMEA in manufacturing to help manufacturers identify high-severity failure modes and mitigate risks effectively.
Practical applications of FMEA in manufacturing demonstrate its efficacy in enhancing the reliability of items. For instance, at Voler Systems, the execution of FMEA in manufacturing as part of a quality management strategy has resulted in safer items entering the market, thereby improving overall customer satisfaction. Producers that emphasize FMEA in manufacturing not only enhance their adherence to regulatory standards but also improve their reputation by ensuring the creation of dependable and safe medical devices. In an industry where safety is paramount, the structured approach of FMEA in manufacturing is essential for maintaining high standards.
Explore Types of FMEA: Tailoring Approaches for Different Manufacturing Needs
In the realm of manufacturing, understanding the various types of Failure Mode and Effects Analysis (FMEA) is crucial for enhancing product development and production efficiency:
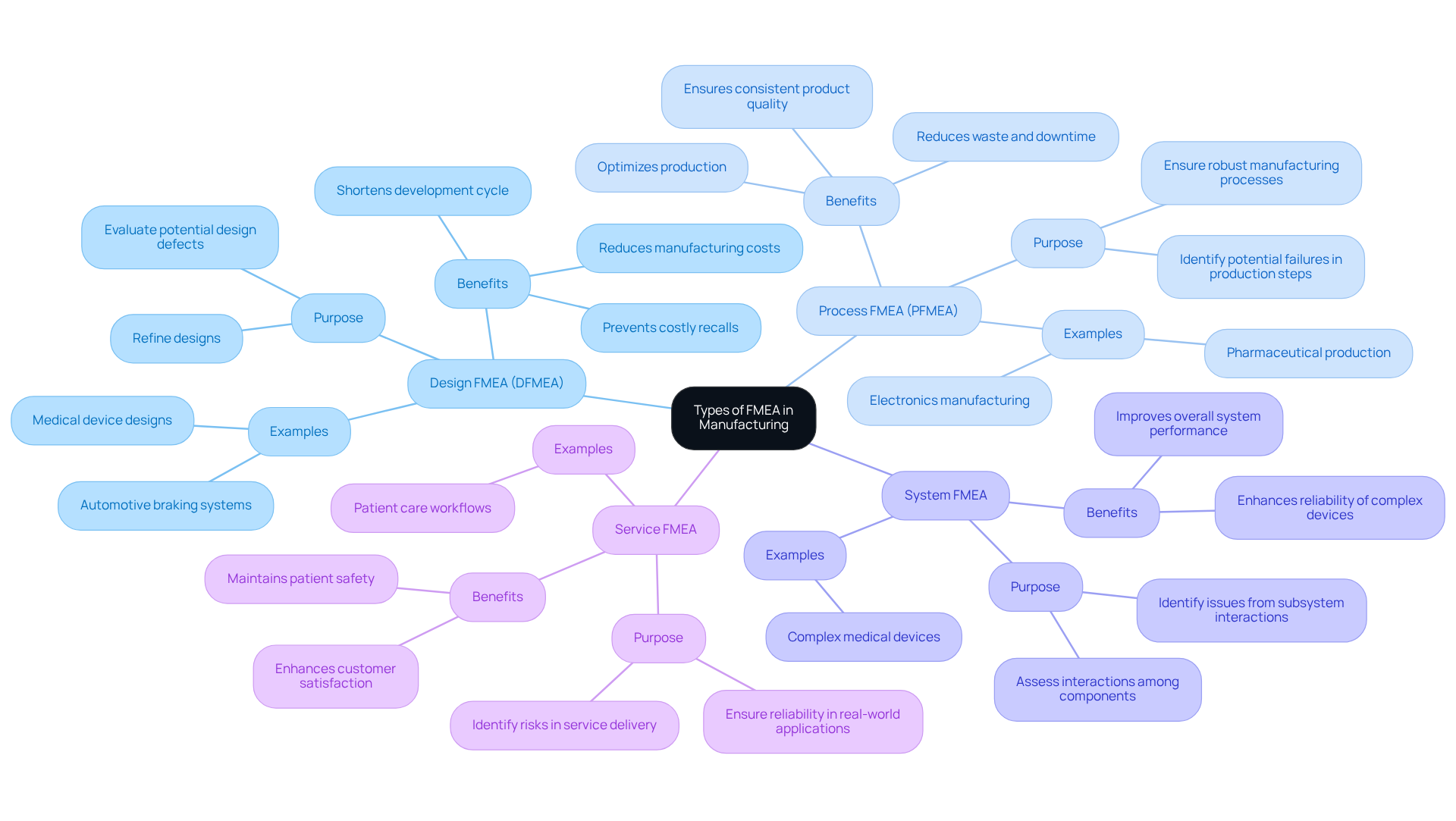
- Design FMEA (DFMEA): This type helps refine designs by evaluating potential design defects that could lead to product shortcomings, ensuring reliability and compliance with safety regulations essential in medical device production. Following best practices in DFMEA can prevent costly recalls and ensure timely, cost-effective delivery by identifying design flaws early, which reduces manufacturing costs and shortens the development cycle.
- Process FMEA (PFMEA): Concentrating on manufacturing processes, PFMEA identifies potential failures in production steps. This analysis is essential for ensuring that manufacturing processes are robust, capable of producing high-quality goods, and compliant with regulatory requirements. For example, PFMEA has been effectively employed in the pharmaceutical sector to sustain contaminant-free production environments, thereby lowering the risk of recalls and improving safety. Additionally, PFMEA helps optimize production by reducing waste, minimizing downtime, and ensuring consistent product quality, which is critical for maintaining high standards in manufacturing processes.
- System Failure Mode and Effects Analysis: This method assesses the interactions among various components within a system, pinpointing issues that may emerge from these interactions. It is particularly useful in complex medical devices where multiple subsystems must work together seamlessly.
- Service Failure Mode and Effects Analysis: Addressing potential failures in service delivery, Service Failure Mode and Effects Analysis ensures that items perform reliably in real-world applications, which is essential for maintaining patient safety and satisfaction.
By comprehending these kinds of FMEA in manufacturing, manufacturers can select the suitable method that aligns with their specific project needs and risk management strategies. The combination of DFMEA and PFMEA not only improves product quality but also aids continuous improvement efforts, ultimately resulting in cost savings and heightened competitiveness in the medical equipment sector. Furthermore, accurate data collection is critical for improving the effectiveness of both DFMEA and PFMEA, ensuring that FMEAs are based on real-world insights. Finally, engaging a multidisciplinary team in the failure mode and effects analysis process is crucial for capturing diverse viewpoints and efficiently identifying potential failures, thus reducing common mistakes in manufacturing tests. Ultimately, the effective implementation of FMEA in manufacturing can significantly mitigate risks and enhance the overall quality of medical devices.
Navigate the FMEA Process: Step-by-Step Guide for Effective Implementation
Implementing FMEA in manufacturing is essential for ensuring risk management in medical device production.
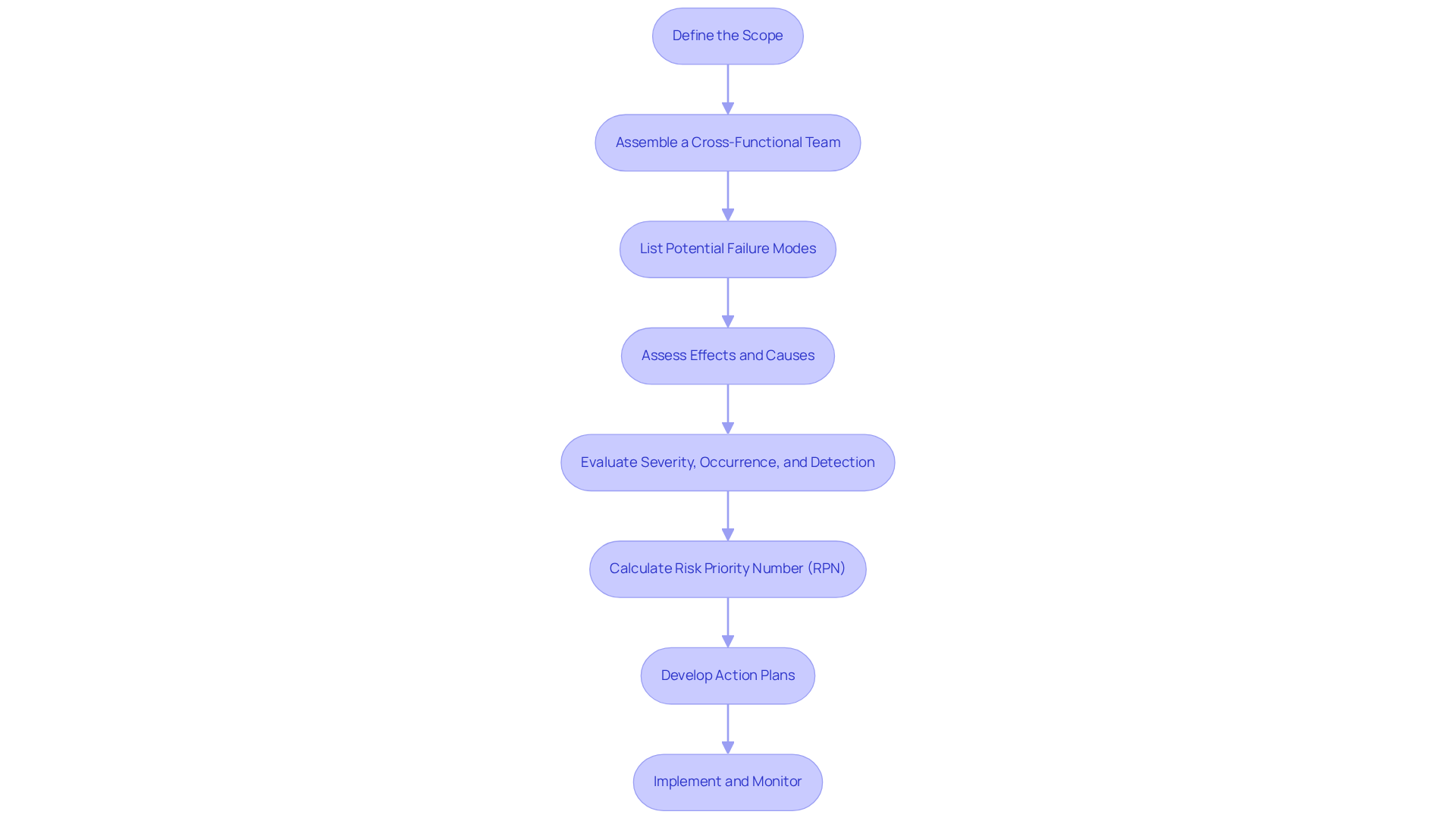
- Define the Scope: Clearly outline the item or process to be analyzed, including boundaries and objectives to focus the FMEA effectively.
- Assemble a Cross-Functional Team: Gather a diverse team with expertise in design, manufacturing, quality assurance, and regulatory compliance. This collaboration is essential for comprehensive analysis and enhances project outcomes significantly.
- List Potential Failure Modes: Conduct brainstorming sessions to identify all possible ways the item or process could fail. Utilizing historical data and user feedback can enrich this process, ensuring a thorough identification of risks.
- Assess Effects and Causes: For each recognized malfunction mode, evaluate the potential effects on the product and determine the underlying causes. This step is crucial for understanding the implications of each mistake.
- Evaluate Severity, Occurrence, and Detection: Rate each defect mode based on its severity (impact), occurrence (likelihood), and detection (ability to identify before it causes harm). High-severity malfunction modes (severity rating of 8 or higher) require immediate attention due to their potential impact on patient safety.
- Calculate Risk Priority Number (RPN): Multiply the severity, occurrence, and detection ratings to prioritize modes based on risk. RPN scores range from 1 to 1,000, guiding teams to focus on the most critical issues.
- Develop Action Plans: For high-priority failure modes, create action plans to mitigate risks. These may include design modifications, process enhancements, or additional testing to improve reliability.
- Implement and Monitor: Execute the action plans and continuously monitor their effectiveness. Regular updates to the failure mode and effects analysis are necessary, especially after significant design modifications or when corrective actions are confirmed as complete.
By systematically following these steps, manufacturers can effectively implement FMEA in manufacturing to identify and address potential failures. This significantly enhances product safety and reliability. Neglecting to implement a robust FMEA process can lead to increased risks and potential harm to patients.
Leverage FMEA Benefits: Enhancing Quality and Compliance in Manufacturing
Implementing FMEA in manufacturing is crucial for manufacturers in the medical device sector, particularly when collaborating with a quality-focused partner like Voler Systems.
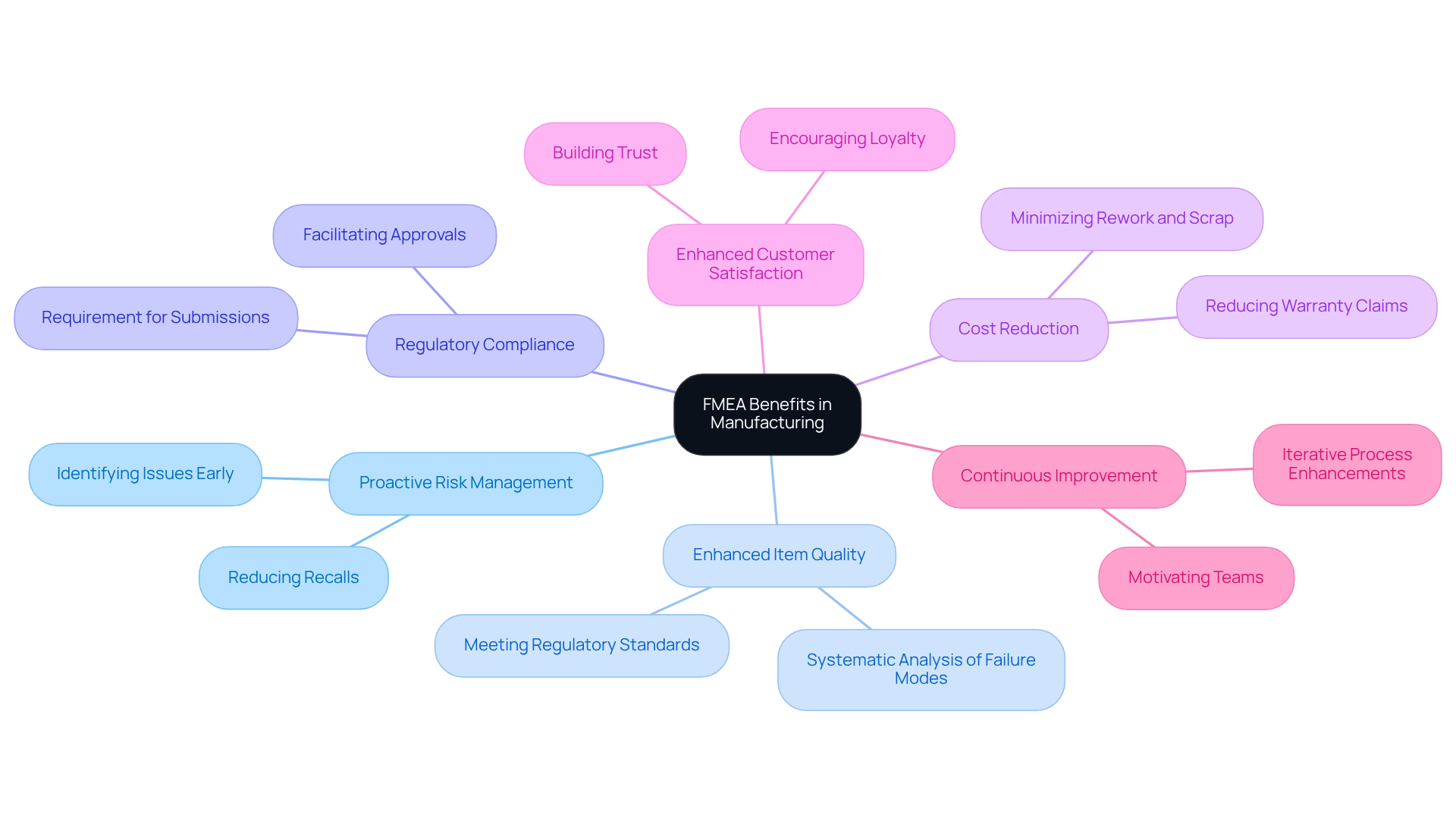
- Proactive Risk Management: FMEA in manufacturing enables manufacturers to recognize and address potential issues before they arise, significantly lowering the risk of expensive recalls and compliance problems. This proactive approach is essential in an industry where regulatory scrutiny is increasing.
- Enhanced Item Quality: Manufacturers can enhance design and manufacturing processes through systematic analysis of failure modes. This leads to the production of higher quality items. Voler Systems exemplifies this commitment to quality, ensuring that enhanced products not only meet regulatory standards but also align with patient safety expectations.
- Regulatory Compliance: FMEA in manufacturing is often a requirement for regulatory submissions in the medical device industry, particularly under standards such as ISO 14971. Its implementation ensures that manufacturers meet compliance obligations, thereby facilitating smoother regulatory approvals. Recent updates, such as the European Commission's adoption of a Delegated Regulation amending the MDR on March 20, 2026, further emphasize the importance of compliance in this evolving landscape.
- Cost Reduction: Early identification of potential failures through failure mode and effects analysis can lead to substantial cost savings by minimizing rework, scrap, and warranty claims. Research shows that efficient FMEA in manufacturing practices can greatly reduce losses associated with time and quality in manufacturing processes. For instance, a study highlighted that implementing FMEA in manufacturing can considerably reduce losses in terms of money, time, and quality.
- Enhanced Customer Satisfaction: Delivering high-quality and reliable products builds customer trust and satisfaction, essential for sustained success in the competitive medical device market. Satisfied customers are more likely to remain loyal and recommend products to peers.
- Continuous Improvement: The failure mode and effects analysis fosters a culture of continuous improvement by motivating teams to frequently review and enhance their assessments based on new information and experiences. This iterative process not only improves quality but also reinforces adherence to evolving regulatory standards.
While FMEA in manufacturing is the most commonly used risk analysis tool in the medical equipment industry, it is important to note that it does not completely satisfy all risk analysis requirements per ISO 14971:2019, especially in addressing safety risks during normal usage. By embracing FMEA, manufacturers not only enhance their product offerings but also solidify their standing in a highly competitive market, especially with the support of a dedicated partner like Voler Systems.
Conclusion
For manufacturers in the medical device sector, ensuring safety and effectiveness from the outset is a complex challenge. Mastering Failure Mode and Effects Analysis (FMEA) is essential, as it systematically identifies and addresses potential failures, making it a crucial tool for enhancing product reliability and ensuring compliance with regulatory standards. This structured approach not only minimizes risks but also promotes ongoing enhancements in product quality, ultimately leading to higher quality medical devices.
The article delves into the various types of FMEA, including:
- Design FMEA
- Process FMEA
These types highlight their specific applications and benefits in manufacturing. Each type plays a vital role in identifying potential malfunctions, optimizing processes, and ensuring that products meet stringent safety regulations. Additionally, the step-by-step guide for implementing FMEA provides manufacturers with a clear roadmap to effectively integrate this analysis into their operations, reinforcing the importance of cross-functional collaboration and data-driven decision-making.
In an industry where patient safety is paramount, the significance of FMEA cannot be overstated. Adopting FMEA practices enables manufacturers to enhance the quality of their medical devices while simultaneously strengthening their reputation and customer satisfaction. Ultimately, the adoption of FMEA practices is not just beneficial; it is a critical factor in sustaining competitive advantage in the medical device industry.
Frequently Asked Questions
What is FMEA and why is it important in manufacturing?
FMEA stands for Failure Mode and Effects Assessment. It is a structured method used to identify potential malfunctions in medical devices, evaluate their consequences, and rank them based on seriousness and probability. Its importance lies in predicting failures that could threaten safety and effectiveness, allowing manufacturers to implement corrective actions early in the design and production phases.
How does FMEA contribute to product reliability in medical device manufacturing?
FMEA contributes to product reliability by enabling manufacturers to address potential malfunctions early in the design process. This proactive approach enhances overall product quality and helps prevent costly recalls, ensuring that medical devices meet high standards of precision and reliability.
What role does FMEA play in compliance with regulatory standards?
FMEA plays a crucial role in ensuring compliance with regulatory standards by helping manufacturers identify high-severity failure modes and mitigate risks effectively. By emphasizing FMEA, manufacturers can enhance their adherence to these standards, thus improving their reputation in the industry.
Can you provide an example of FMEA in practice?
At Voler Systems, the execution of FMEA as part of their quality management strategy has resulted in safer medical devices entering the market. This application of FMEA has improved overall customer satisfaction and demonstrated its efficacy in enhancing the reliability of products.
How does FMEA relate to Advanced Product Quality Planning (APQP)?
FMEA is integral to Advanced Product Quality Planning (APQP) as it helps manufacturers identify potential failure modes early in the development process. Incorporating FMEA into APQP allows for effective risk mitigation and contributes to the overall quality of the manufacturing process.
List of Sources
- Understand FMEA: Definition and Importance in Manufacturing
- FMEAs in Manufacturing: Why They Should Be Your Priority - Cority (https://cority.com/blog/fmea-in-manufacturing)
- Failure Mode and Effects Analysis (FMEA) for Medical Devices (https://vem-medical.com/fmea-for-medical-devices)
- What is FMEA in Manufacturing? Highlighting the Benefits and When to Use It | RB Royal | Turnkey Tube Manufacturers for Hoses, Hardlines, and Hybrid Custom Fluid Transfer Solutions (https://rbroyal.com/about/news/what-is-fmea-in-manufacturing-highlighting-the-benefits-and-when-to-use-it)
- Failure mode effect analysis use and limitations in medical device risk management (https://sciencedirect.com/science/article/pii/S2199853124002336)
- Explore Types of FMEA: Tailoring Approaches for Different Manufacturing Needs
- FMEA, DFMEA, PFMEA, and FMECA: An Overview of FMEA Types (https://relyence.com/2018/08/28/overview-fmea-types)
- DFMEA vs PFMEA: Key Differences, Applications, and Benefits Explained | Tractian (https://tractian.com/en/blog/dfmea-vs-pfmea)
- Types of FMEA: Definitions, Differences, and When to Use Each (https://jamasoftware.com/requirements-management-guide/risk-management/types-of-fmea)
- FMEA Explained: 2023 Guide (https://capvidia.com/blog/fmea-guide)
- FMEA for Medical Devices: Design, Process & Use Risk Analysis Explained (https://meddeviceguide.com/blog/fmea-medical-devices-guide)
- Navigate the FMEA Process: Step-by-Step Guide for Effective Implementation
- Implementing System FMEA: A Step-by-Step Example for Medical Devices (https://volersystems.com/blog/implementing-system-fmea-a-step-by-step-example-for-medical-devices)
- What is FMEA? Failure Mode and Effects Analysis Guide (https://jamasoftware.com/requirements-management-guide/meeting-regulatory-compliance-and-industry-standards/fmea)
- How to do FMEA for Medical Devices: Step-by-Step Guide to Risk Management - SoftComply (https://softcomply.com/fmea-medical-devices-risk-management-guide)
- FMEA in Pharma: Step-by-Step Risk Guide April 2026 | Assyro AI (https://assyro.com/blog/fmea-pharmaceutical-guide)
- Cross-Functional Team Formation in FMEA - Quality Assist (https://quasist.com/fmea/cross-functional-team-formation-in-fmea)
- Leverage FMEA Benefits: Enhancing Quality and Compliance in Manufacturing
- Failure mode effect analysis use and limitations in medical device risk management (https://sciencedirect.com/science/article/pii/S2199853124002336)
- FDA Aligns U.S. Medical Device Rules with Global Standards (https://mddionline.com/regulatory-quality/medical-device-companies-must-navigate-historic-fda-rule-change)
- (PDF) A Case Study: A Process FMEA Tool to Enhance Quality and Efficiency of Manufacturing Industry (https://researchgate.net/publication/287694384_A_Case_Study_A_Process_FMEA_Tool_to_Enhance_Quality_and_Efficiency_of_Manufacturing_Industry)
- April 2026 Regulatory Update: What You Shouldn’t Miss (https://linkedin.com/pulse/april-2026-regulatory-update-what-you-shouldnt-miss-jzsxf)
- Medical Devices Compliance Updates Newsletter – March 2026 (https://sushvin.com/medical-devices-compliance-updates-newsletter-March-2026.html)





