Best Practices for Industrial Product Development in Medical Devices
Explore best practices in industrial product development for medical devices, ensuring...

In the rapidly evolving landscape of medical device development, effective prototyping serves as a critical bridge between innovative ideas and successful products. Establishing clear objectives, selecting appropriate materials, and embracing iterative testing are essential steps that organizations can take to significantly enhance their prototyping processes.
However, a key challenge persists: how can cross-functional teams collaborate effectively to navigate the complexities of regulatory compliance and user needs?
This article explores best practices for automotive prototyping specifically tailored for the medical sector, providing insights that can transform initial concepts into market-ready solutions.
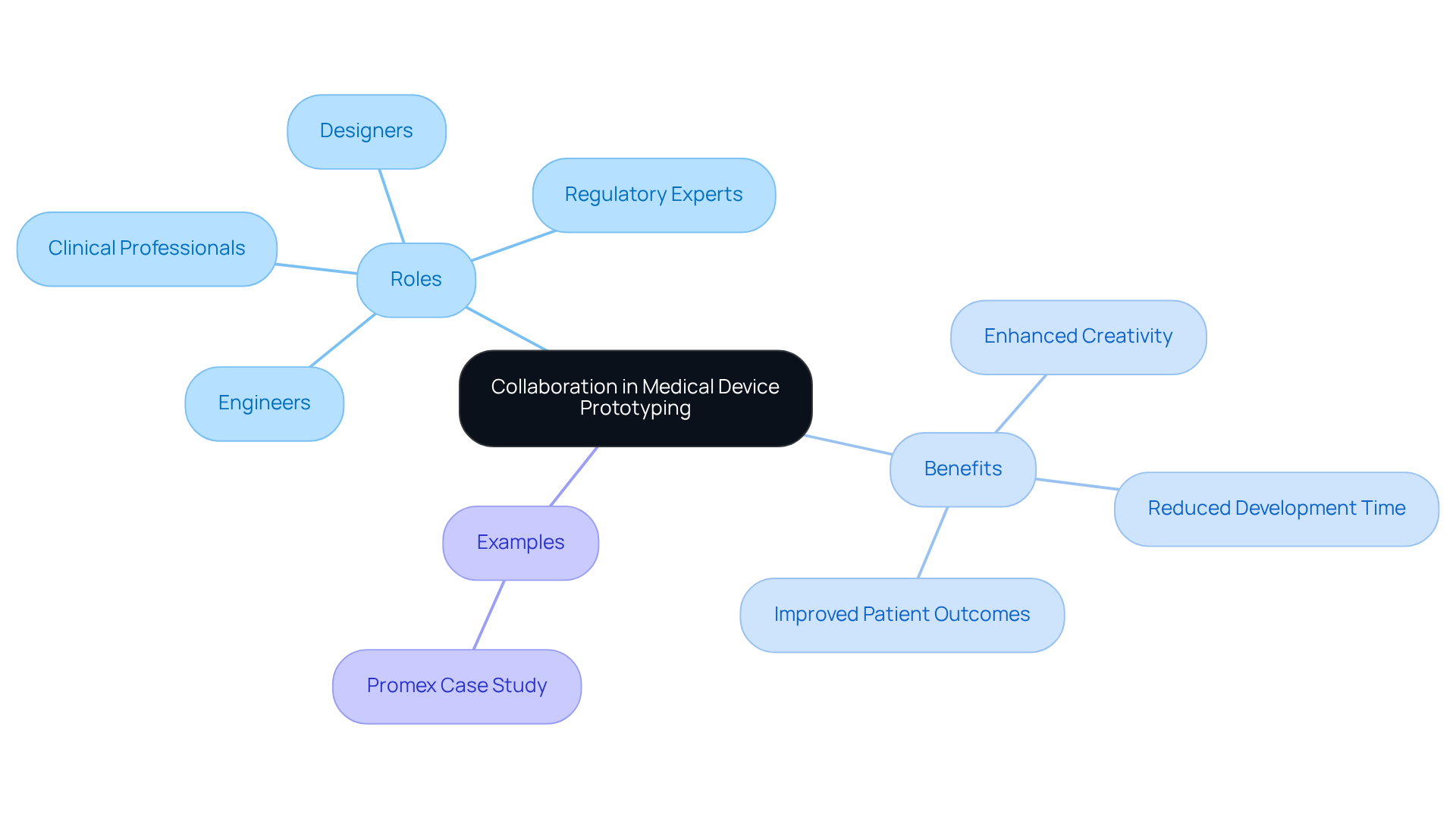
Establishing clear objectives and requirements is essential for initiating an effective in . This begins with outlining specific objectives for the model, such as:
Engaging stakeholders - including engineers, designers, and end-users - ensures that diverse perspectives are considered, leading to a comprehensive understanding of what the prototype should achieve. For instance, if the objective is to evaluate a new , it is crucial to specify the performance metrics that will be assessed during testing.
Thorough documentation of these objectives not only but also aligns all team members toward a common vision. This clarity streamlines the , facilitating focused iterations and enhancing the likelihood of successful outcomes.
Furthermore, adhering to is vital for health technology startups, as it aids in navigating efficiently. Recognizing during the prototyping stage can further ensure quality and efficiency in electronic creation, ultimately resulting in a more .
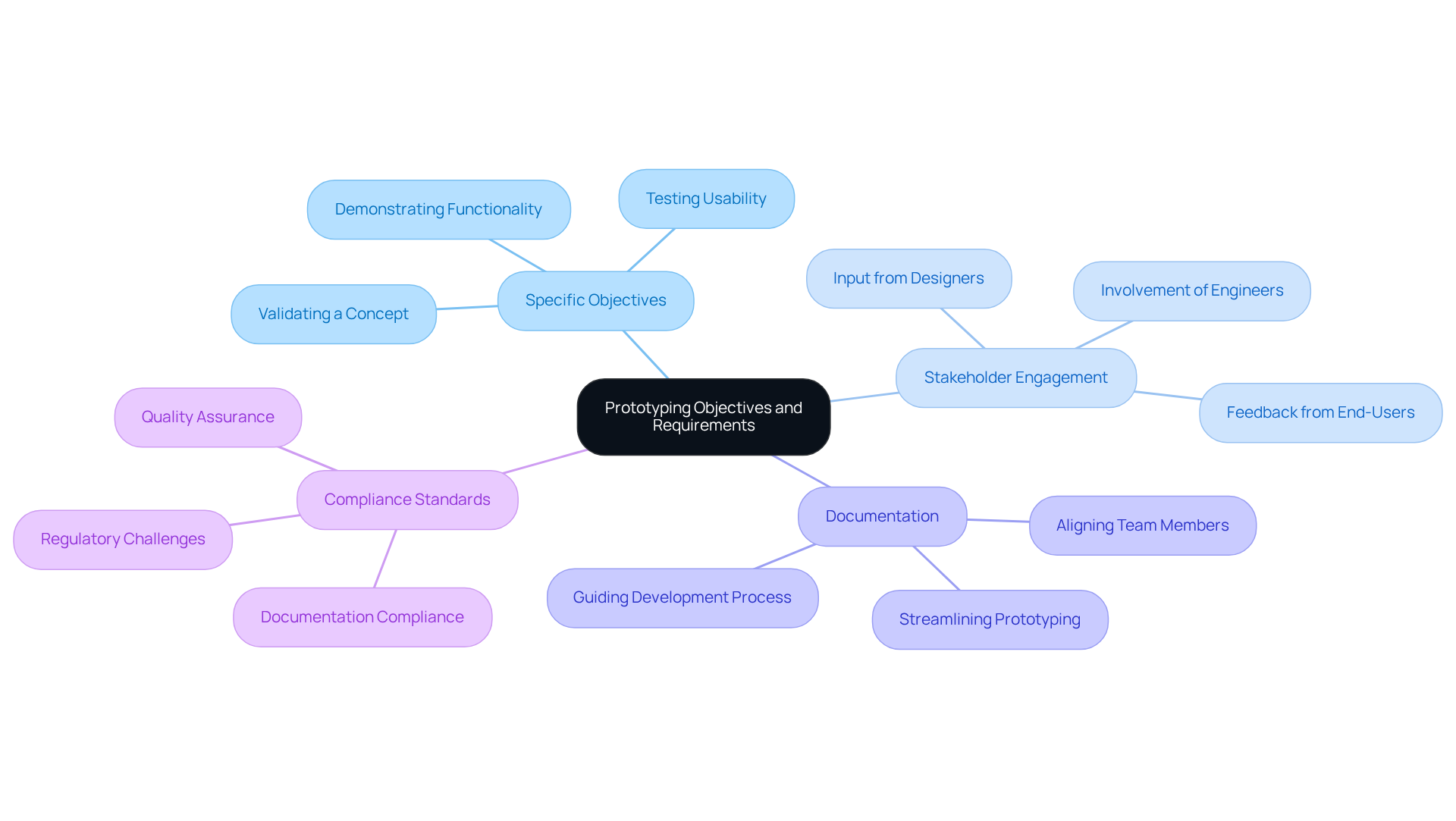
Selecting the right materials and technologies is essential in the for . It is important to consider , including , sterilization needs, and mechanical properties. For wearable devices, materials such as silicone and thermoplastics are often preferred due to their comfort and flexibility.
Advanced technologies like enable the creation of models that closely resemble the final product, allowing for quick iterations and modifications. This adaptability facilitates the rapid evaluation of various concepts, ensuring that models meet both functional and .
Compliance with regulations such as ISO 13485 is crucial, as it streamlines the and increases the likelihood of successful market entry. Current trends in biocompatible materials highlight the use of certified , which are vital for ensuring safety and effectiveness in healthcare applications.
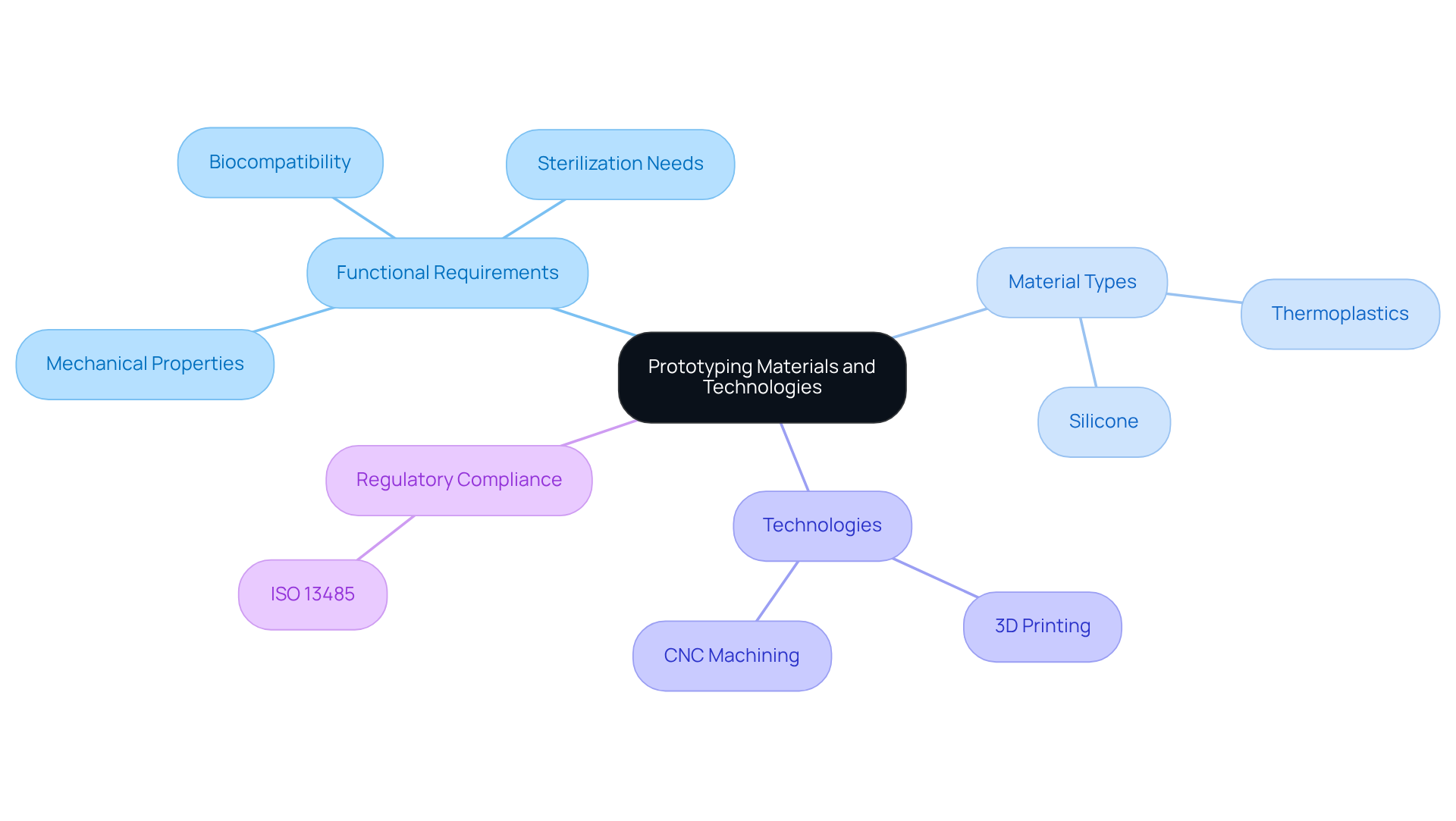
To maximize the effectiveness of the , implementing is essential. Start by conducting preliminary tests with the model to gather insights on its performance and usability. Actively seek feedback from end-users, healthcare professionals, and other stakeholders to identify areas that require enhancement. This feedback should inform necessary alterations, which are then followed by retesting the revised model. This cycle of testing and refinement should continue until the model meets all functional and regulatory requirements.
For instance, if reveals challenges in using a device, adjustments can be made to improve ergonomics or interface layout. , such as the innovative calf-worn device developed by for motion and circumference monitoring in knee replacement rehabilitation, demonstrate that incorporating clinician feedback leads to designs that are both inventive and highly functional, ultimately based on real-world insights.
Furthermore, for by allowing early-stage testing to identify flaws in the concept before full-scale manufacturing. This iterative approach not only refines the prototype but also fosters a culture of collaboration and innovation within the development team. Testimonials from clinical teams highlight how has directly influenced enhancements, showcasing the effectiveness of this approach in real-world applications.
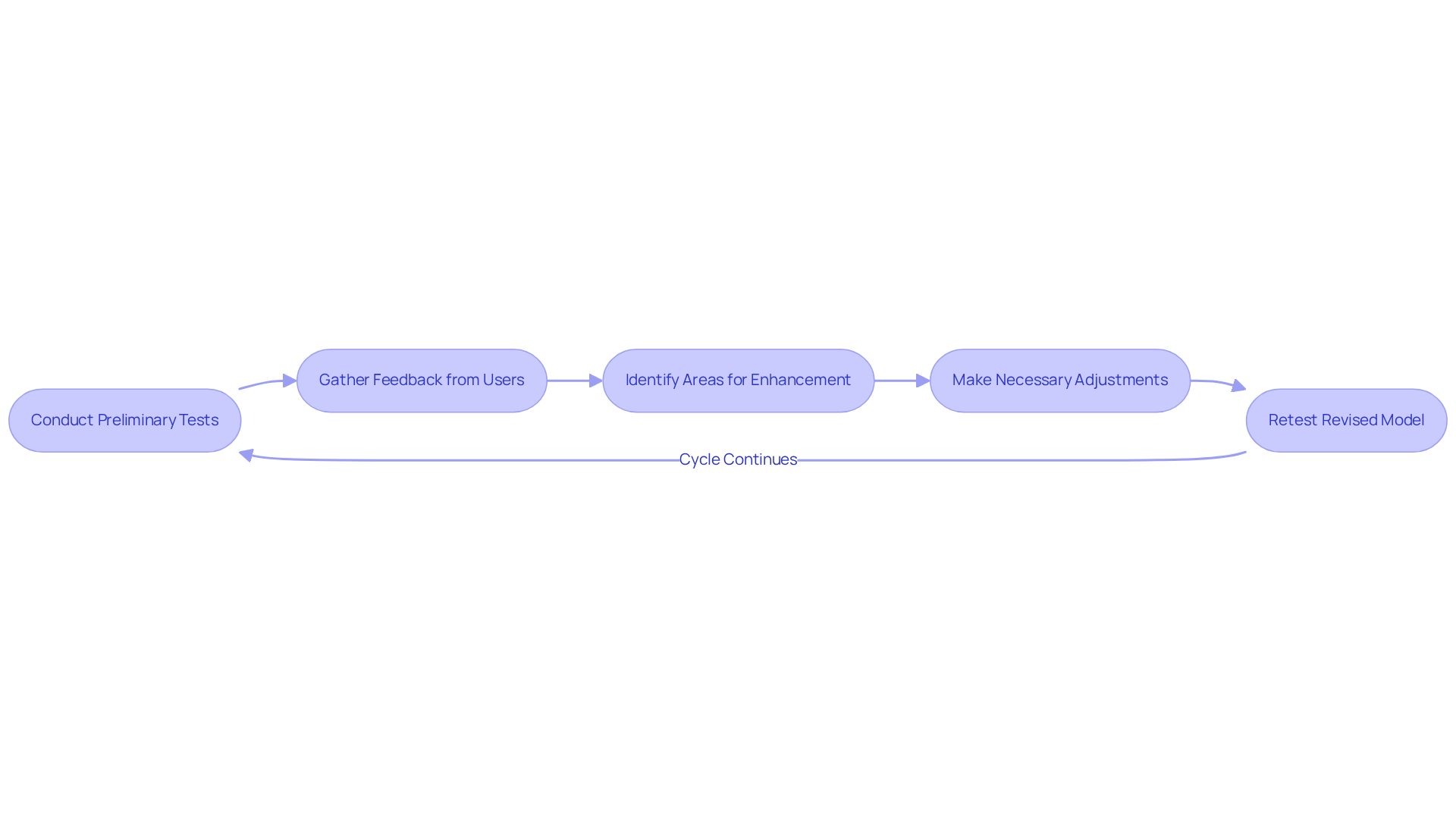
Successful relies heavily on , which include engineers, designers, regulatory experts, and clinical professionals. Establishing regular meetings and effective communication channels is essential for facilitating information sharing and informed decision-making. By encouraging team members to share their unique perspectives, organizations can . For instance, involving clinical professionals early in the prototyping process provides and , ensuring that prototypes are aligned with real-world applications.
Research indicates that and enhance creativity, ultimately leading to more effective . A case study from Promex illustrates this point, demonstrating that mitigated delays and design compromises, successfully achieving product goals related to performance and reliability.
By cultivating a collaborative environment, teams can harness diverse expertise to . This approach results in products that not only meet stringent industry standards but also . As the adage states, 'If you want to go fast, go alone, but if you want to go far, go together.
Establishing an effective prototyping process for medical devices necessitates a strategic approach that includes clear objectives, appropriate material selection, iterative testing, and robust collaboration among cross-functional teams. By adhering to these best practices, developers can significantly enhance the quality and functionality of their prototypes, ultimately leading to successful product launches that comply with regulatory standards and fulfill user needs.
Key insights from the article underscore the importance of:
Engaging stakeholders throughout the process ensures the integration of diverse perspectives, while iterative testing cultivates a culture of continuous improvement. Furthermore, fostering collaboration among various experts accelerates development timelines and enhances innovation, resulting in products that effectively address real-world healthcare challenges.
In the rapidly evolving landscape of medical device development, following these best practices not only streamlines the prototyping process but also positions organizations to better serve patients and healthcare professionals. Embracing these principles can lead to the creation of safer, more effective medical devices that ultimately improve patient outcomes and advance the field of healthcare technology.
Why is it important to establish clear objectives and requirements for prototyping in medical device development?
Establishing clear objectives and requirements is essential as it guides the prototyping process, aligns team members towards a common vision, and enhances the likelihood of successful outcomes.
What specific objectives should be outlined for a prototype?
Specific objectives for a prototype may include validating a concept, testing usability, and demonstrating functionality.
Who should be engaged in the prototyping process?
Stakeholders such as engineers, designers, and end-users should be engaged to ensure diverse perspectives are considered, leading to a comprehensive understanding of the prototype's goals.
How does thorough documentation benefit the prototyping process?
Thorough documentation guides the development process, aligns team members, streamlines the prototyping process, and facilitates focused iterations.
Why is adherence to documentation compliance standards important for health technology startups?
Adhering to documentation compliance standards is vital for navigating regulatory challenges efficiently, which is crucial for health technology startups.
What common mistakes should be recognized during manufacturing tests in the prototyping stage?
Recognizing common mistakes during manufacturing tests can help ensure quality and efficiency in electronic creation, ultimately leading to a more successful product launch.
