10 FMEA Examples for Medical Device Design Success
Introduction In medical device design, even minor oversights can lead to critical...

In the high-stakes realm of medical device development, ensuring patient safety is of utmost importance. Failure Mode and Effects Analysis (FMEA) stands as a crucial framework for identifying potential risks and enhancing the reliability of devices, ranging from heart pumps to wearable sensors. As the healthcare industry continues to evolve, the integration of advanced methodologies and technologies prompts a significant inquiry: how can manufacturers adeptly navigate the complexities of FMEA to not only meet stringent regulatory requirements but also promote innovation in patient care?
This article explores ten compelling examples of FMEA in action, highlighting its essential role in safeguarding health outcomes across diverse medical technologies.
In heart pump design, a failure mode and effects analysis example plays a critical role in systematically identifying potential failure modes that could compromise patient safety. For instance, a malfunction in the pump's motor could lead to inadequate blood flow, posing a significant risk to patients. By meticulously analyzing the failure modes of each component, engineers at Voler Systems can implement design modifications or redundancies to effectively mitigate these risks. This proactive approach not only of the equipment but also ensures adherence to stringent regulatory standards for medical devices.
Recent advancements in FMEA methodologies, particularly the incorporation of AI-driven risk assessment tools developed by Voler Systems, have further refined this process. These innovations enable more accurate evaluations and contribute to improved patient safety outcomes. Case studies indicate that organizations employing a failure mode and effects analysis example in heart pump development have markedly reduced failure-related incidents, underscoring the importance of this analytical method in safeguarding patient health.
Wearable sensors, such as those used for heart rate monitoring, rely heavily on precise data transmission and processing. Implementing a failure mode and effects analysis example is essential for identifying potential failure modes, including sensor drift and communication errors, which can lead to inaccurate health assessments. By proactively addressing these issues during the design phase, manufacturers can significantly enhance the reliability of wearable technology and ensure compliance with evolving health regulations.
Voler Systems specializes in embedded systems design, leveraging FPGA technology and AI to create intelligent solutions that improve battery life and performance, particularly for wireless medical equipment. These systems offer a flexible, low-risk approach to successful design, integrating hardware acceleration with programmable logic.
The FDA's 2026 guidance clarifies the definition of a regulated device, potentially lowering barriers for certain technologies while emphasizing the necessity for rigorous validation. Recent studies indicate that:
These statistics highlight the importance of comprehensive compliance measures and the role of testing systems in ensuring quality and reliability in the development of wearable health technologies.
However, challenges such as small sample sizes and limited real-world validation persist in the field, underscoring the need for ongoing research and development.
Liquid biopsy platforms utilize advanced technologies to identify cancer biomarkers in blood samples, playing a crucial role in early cancer detection. Implementing a failure mode and effects analysis example is vital for identifying potential failure modes, such as sample contamination and assay inaccuracies, which can significantly compromise diagnostic reliability.
By systematically evaluating these uncertainties, developers can establish rigorous quality controls that improve the accuracy of liquid biopsy tests. This proactive approach not only mitigates challenges but also in oncology, reflecting the industry's commitment to precision medicine.
Notably, CancerIQ data indicates that 40% of patients undergoing comprehensive cancer evaluations and genetic testing require adjustments in their medical management, underscoring the critical need for reliable diagnostic tools in effective cancer care.
As emphasized by the National Cancer Institute, precision prevention has the potential to eradicate cancer as we know it by preventing suffering and death for those at risk, highlighting the significance of dependable diagnostics in achieving this objective.
Furthermore, case studies demonstrate how the failure mode and effects analysis example has been effectively employed to enhance the reliability of liquid biopsy platforms, illustrating its practical implications in improving patient care.
IoT-enabled medical instruments, such as remote monitoring systems, face distinct challenges related to connectivity and data security. Implementing a failure mode and effects analysis example is crucial for identifying potential failure modes, including network outages and data breaches, which can significantly impact performance. Notably, over 70% of healthcare IoT equipment operates on outdated systems, rendering them particularly vulnerable to cyber threats. Addressing these challenges during the design phase allows manufacturers to enhance the robustness and safety of IoT products, ensuring reliable functionality in critical healthcare environments.
Case studies demonstrate that organizations employing a failure mode and effects analysis example can reduce cyberattack risks by as much as 60%, underscoring the importance of robust risk management strategies in the development of IoT health technologies. This proactive approach not only protects but also builds trust between manufacturers and healthcare providers, contributing to a more secure healthcare ecosystem. Moreover, ongoing post-market management and continuous monitoring of deployed equipment are vital for maintaining security throughout their lifecycle.
Engineers at Voler Systems possess the necessary expertise in planning and project assessment, enabling them to identify when a project encounters difficulties and to intervene effectively. Their proficiency in advanced embedded systems design, particularly through AI-driven design and FPGA technology, ensures that wireless healthcare tools are optimized for battery longevity and performance, further enhancing the reliability of these essential health solutions.
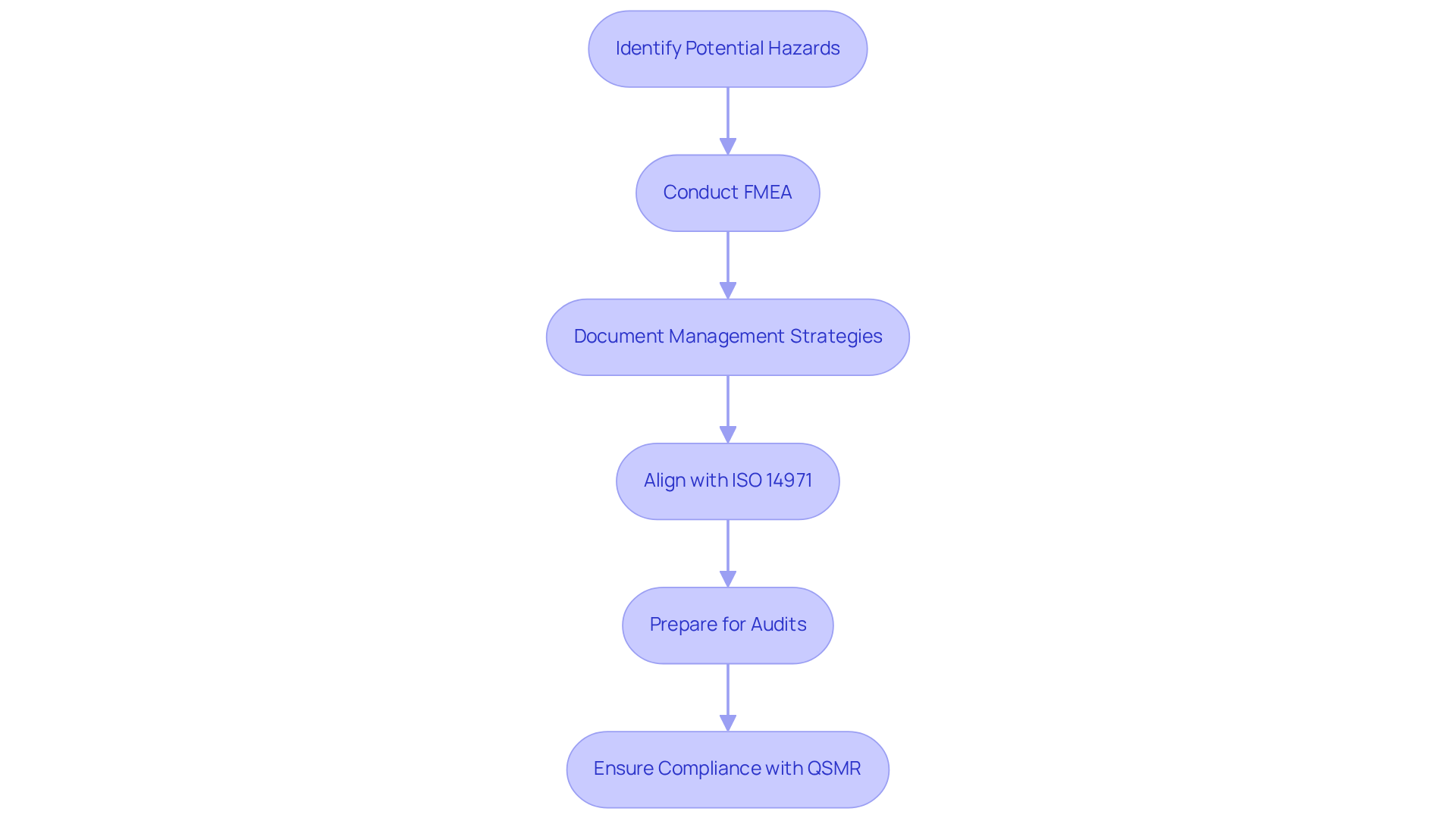
In the medical device industry, adherence to regulations such as ISO 14971 is essential for ensuring product safety and effectiveness. An example of failure mode and effects analysis serves as a critical tool that aids manufacturers in identifying potential hazards during the design and production processes. By systematically analyzing failure modes, companies can document their management strategies, which is vital for demonstrating compliance during audits and inspections.
Recent updates indicate that the FDA is placing increased emphasis on risk management within quality systems, rendering failure mode and effects analysis even more pertinent. For instance, the initial year of implementing the new Quality System Regulation (QSMR) has shown no leniency, compelling companies to fully comply with all requirements from the outset. This proactive approach is necessary to avoid enforcement actions and ensure smooth operations during the transition.
Case studies illustrate that startups often encounter challenges with regulatory compliance due to limited experience and resources. However, those that effectively integrate FMEA into their processes have reported improved alignment with ISO 14971 standards. Established companies are also adapting their quality agreements to reflect the new QSMR language, thereby enhancing their quality systems to meet these updated standards.
Statistics reveal that companies utilizing a failure mode and effects analysis example not only improve their compliance rates but also enhance patient safety outcomes. As the FDA begins to enforce the new regulations, manufacturers must prepare for potential discrepancies in their records and ensure clarity in demonstrating compliance with both the previous and current standards. Thus, this analysis acts as a foundation for achieving , ensuring that medical instruments are designed and produced to meet the highest safety and quality standards.
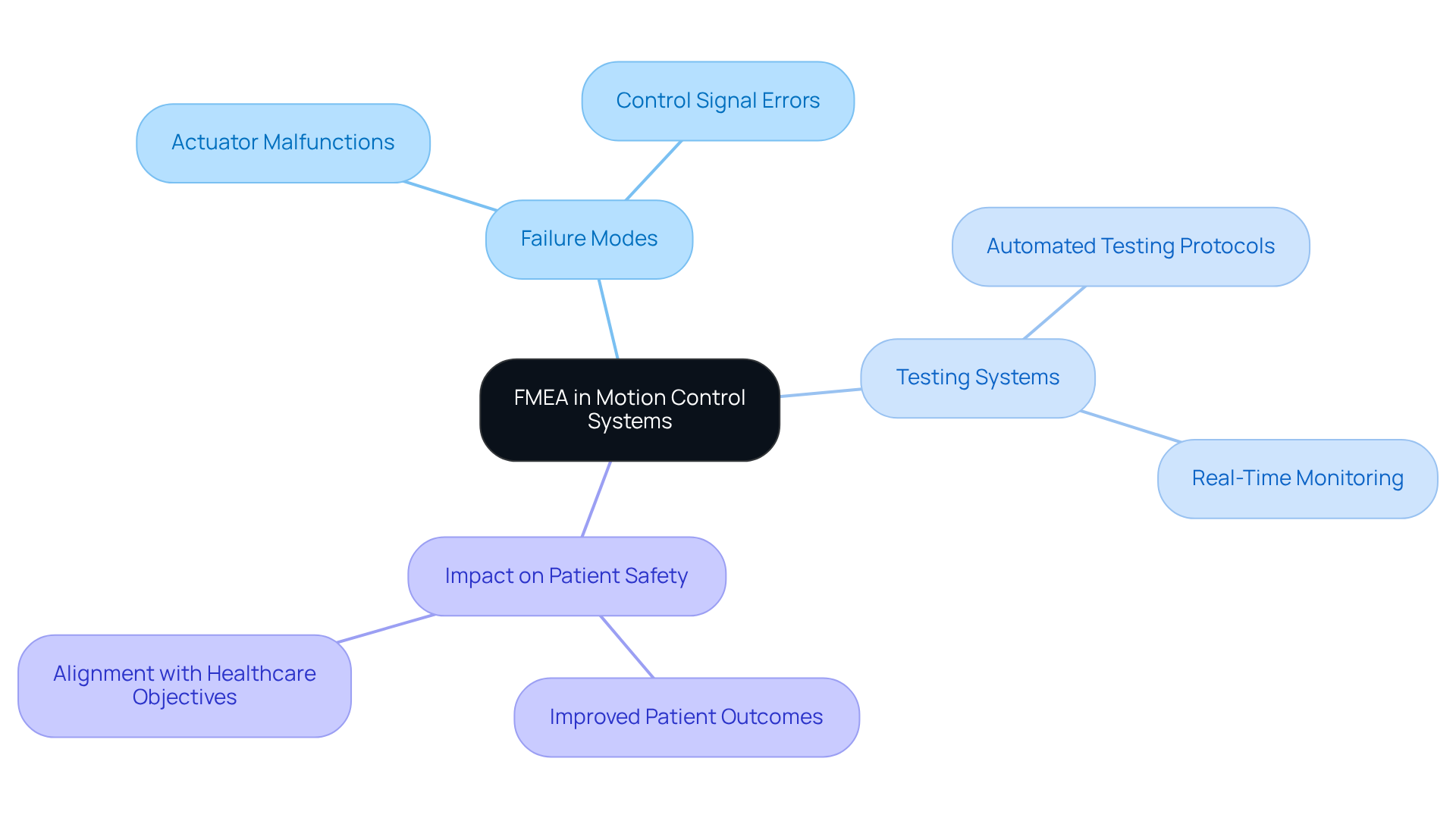
Motion control systems play a crucial role in equipment such as robotic surgical instruments and infusion pumps. A failure mode and effects analysis example is utilized to systematically identify potential failure modes, including actuator malfunctions and control signal errors, which could compromise performance. This proactive approach, recognized since the 1990s as an essential risk assessment tool in healthcare, enables manufacturers to address these risks during the design process, thereby enhancing the precision and safety of motion control systems. Such measures not only improve patient outcomes but also align with the strategic objectives of healthcare organizations, where patient safety is a fundamental quality objective.
Voler Systems underscores the importance of , which incorporate features like automated testing protocols and real-time monitoring, in ensuring that electronic products meet stringent quality and reliability standards. For instance, a failure mode and effects analysis example conducted by two separate groups of pharmacists in Sri Lanka involved using FMEA to analyze the dispensing process, identifying 90 failure modes and prioritizing 66 for corrective actions. This underscores the significance of a failure mode and effects analysis example in ensuring the reliability and safety of health-related tools, especially in critical environments such as surgery. Furthermore, Voler Systems' expertise in supporting legacy test equipment for healthcare companies reflects their commitment to upholding high standards in testing and compliance.
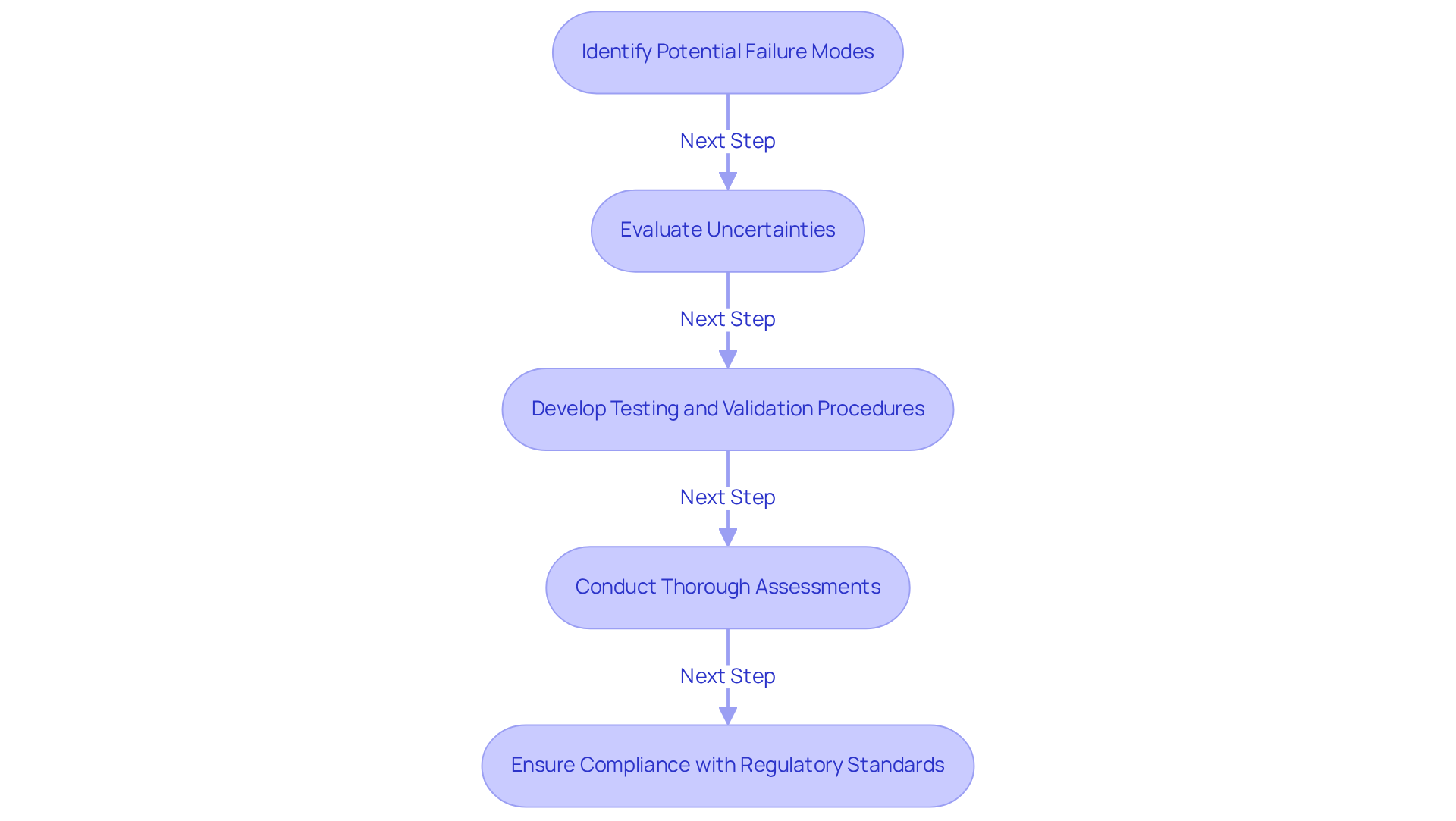
Software and firmware malfunctions in health-related equipment can lead to significant issues, including erroneous data handling and complete equipment breakdown. An example of failure mode and effects analysis is its use as a crucial tool for identifying potential failure modes, such as coding errors and compatibility issues, that may jeopardize the functionality of the equipment. By systematically evaluating these uncertainties, manufacturers can develop comprehensive testing and validation procedures that enhance the reliability of software components in healthcare devices.
Voler Systems emphasizes the importance of , which integrate features like automated testing protocols and real-time monitoring. These systems ensure that products undergo thorough assessments for quality and reliability before they reach the market. Recent statistics indicate that over half of connected healthcare instruments exhibit recognized critical vulnerabilities, highlighting the necessity of using a failure mode and effects analysis example to proactively address these challenges.
Furthermore, case studies demonstrate that using a failure mode and effects analysis example in the development lifecycle not only mitigates risks but also fosters a culture of quality assurance. This approach guarantees that products comply with stringent regulatory standards and prioritize patient safety. Voler Systems also provides documentation compliance assistance, aiding health product startups in effectively navigating regulatory hurdles. As Boris Beizer aptly noted, 'Software never was perfect and won’t get perfect,' highlighting the persistent challenges in achieving software reliability.
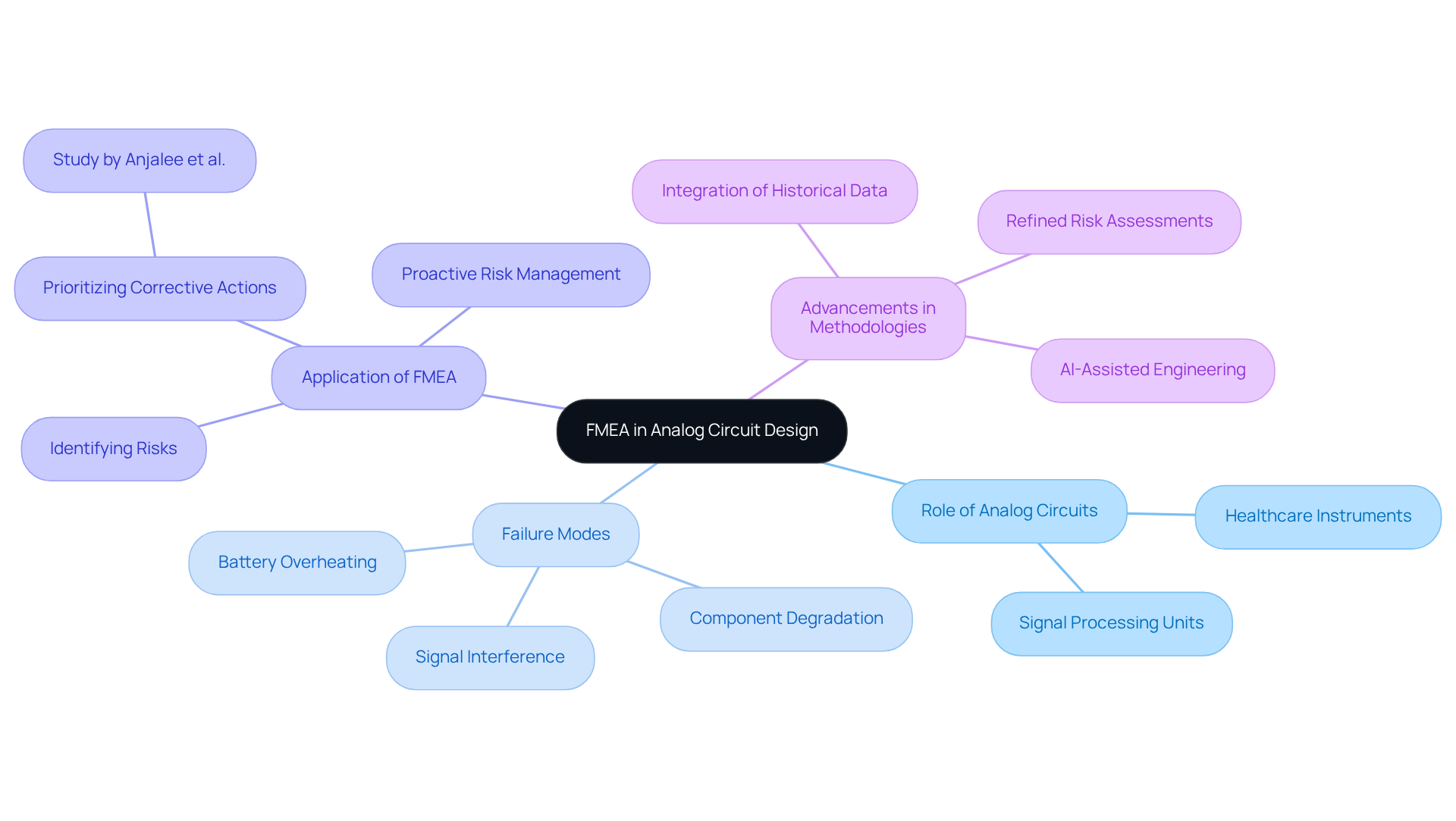
Analog circuits play a crucial role in the functionality of various healthcare instruments, including sensors and signal processing units. The implementation of a failure mode and effects analysis example is vital for identifying potential failure modes, such as component degradation and signal interference, which could jeopardize circuit performance. By addressing these risks during the design phase, manufacturers can enhance the reliability of analog circuits, ensuring that health-related equipment operates efficiently and safely.
Voler Systems, leveraging its expertise in for medical equipment, utilizes AI-assisted engineering to expedite the development of wearable technology, heart pumps, and liquid biopsy platforms. This approach ensures that products are designed for the future of intelligent healthcare. For example, a study conducted by Anjalee et al. identified 90 failure modes and prioritized 66 for corrective actions, demonstrating the effectiveness of FMEA in improving equipment reliability.
Recent advancements in FMEA methodologies have further refined the process, allowing for more precise risk assessments and the integration of historical incident data. This proactive strategy not only adheres to regulatory compliance standards but also protects patient health.
Wireless communication plays a crucial role in , particularly in remote monitoring systems and connected implants. A failure mode and effects analysis example demonstrates how FMEA is employed to identify potential failure modes, such as signal interference and data transmission errors, which can compromise data integrity. Statistics reveal that approximately one-third of healthcare IoT devices have identified significant hazards, underscoring the necessity of this analysis. By systematically assessing these hazards, manufacturers can develop robust communication protocols and error-checking systems, ensuring that health products consistently deliver reliable data transmission.
The integration of specific features in testing systems, including real-time monitoring and automated error detection, is vital for enhancing equipment reliability and managing compliance from prototype to production, particularly in the realm of wearable health devices. Case studies demonstrate that using a failure mode and effects analysis example in the design process not only enhances equipment reliability but also mitigates risks associated with compromised data, ultimately safeguarding patient safety. Moreover, maintaining transparency with customers regarding vulnerabilities is essential for fostering trust and ensuring patient safety.
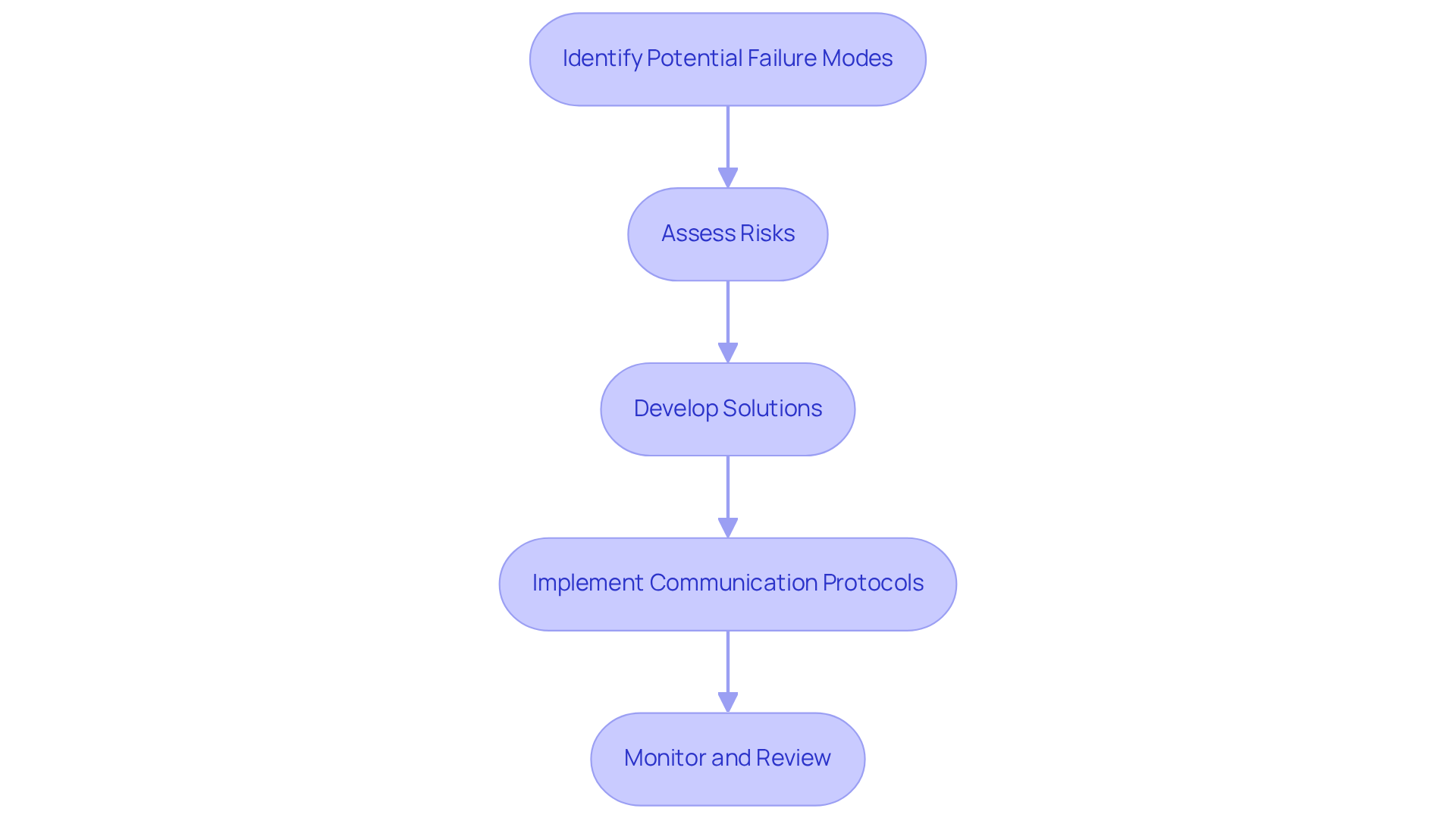
A comprehensive failure mode and effects analysis (FMEA) method necessitates a thorough examination of all components and processes associated with healthcare product design, encompassing hardware, software, and regulatory compliance. This holistic approach is essential for identifying and mitigating throughout the development lifecycle. By integrating FMEA into the design process, manufacturers can significantly improve the safety and reliability of healthcare devices, ultimately enhancing patient outcomes and ensuring adherence to regulatory standards.
For instance, a case study conducted in a teaching hospital showcased a failure mode and effects analysis example, where teams identified 90 failure modes and prioritized corrective actions. This example underscores the effectiveness of FMEA in bolstering safety and reliability. Furthermore, FMEA has been recognized in the healthcare sector since the 1990s as a proactive evaluation tool, emphasizing its role in preventing healthcare errors and maintaining compliance with stringent industry standards. The incorporation of FMEA not only facilitates data collection for informed decision-making but also promotes proactive risk management, establishing it as an indispensable resource for medical device manufacturers committed to delivering high-quality products.
The implementation of failure mode and effects analysis (FMEA) in medical device design is essential for enhancing patient safety and ensuring device reliability. By systematically identifying potential failure modes across various medical technologies - ranging from heart pumps to wearable sensors - FMEA acts as a proactive tool that mitigates risks and complies with regulatory standards. This analytical approach not only strengthens the integrity of healthcare devices but also reinforces the commitment to patient health and safety.
Throughout this article, diverse examples illustrate the critical role of FMEA in various medical applications. For instance, in heart pump design, engineers can proactively address motor malfunctions that could jeopardize blood flow. Similarly, in the realm of wearable sensors, FMEA aids in identifying issues such as sensor drift, thereby ensuring accurate health assessments. The case studies presented highlight the tangible benefits of employing FMEA, including improved compliance with industry regulations and enhanced patient outcomes.
Ultimately, the importance of integrating a comprehensive FMEA approach in medical device development cannot be overstated. As healthcare technology continues to evolve, embracing such proactive risk management strategies will be crucial in fostering innovation while safeguarding patient safety. Manufacturers are encouraged to adopt FMEA as a foundational practice, not only to meet regulatory requirements but also to instill confidence in the quality and reliability of their medical products.
What is the role of Failure Mode and Effects Analysis (FMEA) in heart pump design?
FMEA plays a critical role in systematically identifying potential failure modes in heart pump design that could compromise patient safety, such as motor malfunctions leading to inadequate blood flow.
How do engineers at Voler Systems use FMEA to improve heart pump reliability?
Engineers at Voler Systems analyze failure modes of each component to implement design modifications or redundancies, effectively mitigating risks and enhancing the reliability of the equipment.
What advancements have been made in FMEA methodologies for heart pump design?
Recent advancements include the incorporation of AI-driven risk assessment tools developed by Voler Systems, which enable more accurate evaluations and contribute to improved patient safety outcomes.
What are the benefits of using FMEA in the development of wearable sensors?
FMEA helps identify potential failure modes in wearable sensors, such as sensor drift and communication errors, leading to enhanced reliability and compliance with health regulations.
How does Voler Systems enhance the performance of wearable technology?
Voler Systems leverages FPGA technology and AI to create intelligent solutions that improve battery life and performance, particularly for wireless medical equipment.
What is the significance of the FDA's 2026 guidance regarding regulated devices?
The FDA's 2026 guidance clarifies the definition of a regulated device, potentially lowering barriers for certain technologies while emphasizing the necessity for rigorous validation.
What challenges exist in the field of wearable health technologies?
Challenges include small sample sizes and limited real-world validation, highlighting the need for ongoing research and development to ensure quality and reliability.
Why is FMEA important in liquid biopsy platforms?
FMEA is vital for identifying potential failure modes such as sample contamination and assay inaccuracies, which can compromise diagnostic reliability in liquid biopsy platforms.
How does implementing FMEA improve patient outcomes in oncology?
By systematically evaluating uncertainties and establishing rigorous quality controls, FMEA enhances the accuracy of liquid biopsy tests, ultimately improving patient outcomes in cancer care.
What does CancerIQ data reveal about patients undergoing cancer evaluations?
CancerIQ data indicates that 40% of patients undergoing comprehensive cancer evaluations and genetic testing require adjustments in their medical management, highlighting the need for reliable diagnostic tools.
