Understanding Prototype Development: Definition and Importance for Medical Devices
Introduction Prototyping serves as a cornerstone in medical device development, where...

In the competitive landscape of medical device development, the success of a product often hinges on the effectiveness of its prototyping phase. Adhering to established best practices enables developers to streamline their processes and ensure compliance with essential regulations, thereby enhancing product safety and efficacy.
However, what critical strategies can transform prototyping from a mere formality into a powerful tool for innovation and market readiness? This article explores four essential practices that can elevate product development prototyping:
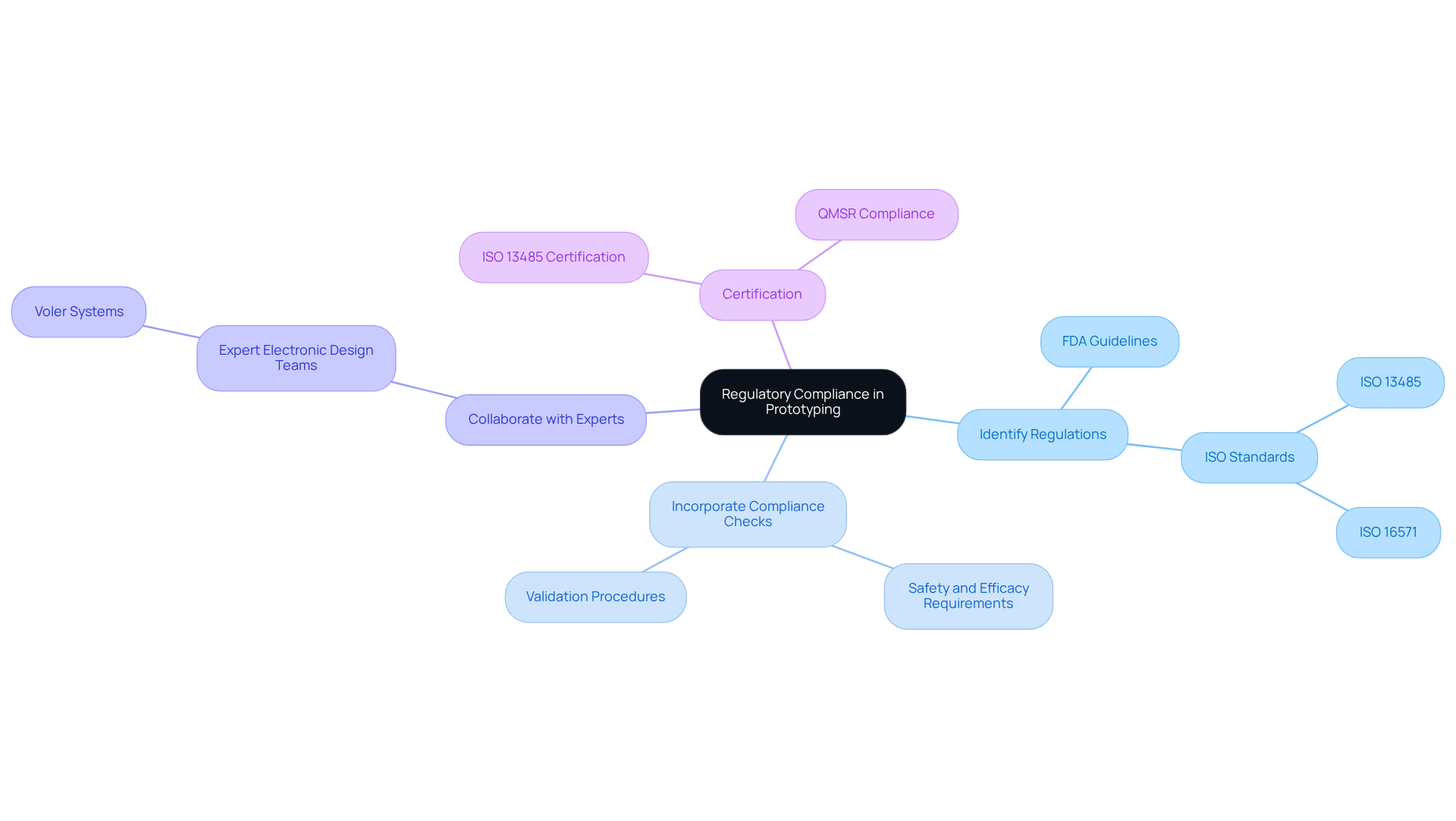
In medical device development, understanding regulatory compliance is essential. In product development prototyping, it is essential to commence with a comprehensive understanding of relevant regulations, such as those established by the FDA or ISO standards. This approach ensures that the prototype not only meets design specifications but also complies with safety and efficacy requirements.
Begin by identifying the specific regulations pertinent to your product category. For example, if you are developing a wearable health monitor, it is crucial to familiarize yourself with the FDA's guidelines on software validation and clinical testing. Incorporating compliance checks during product development prototyping can significantly reduce the risk of costly modifications later in the development cycle.
Moreover, collaborating with expert electronic design teams, such as those at Voler Systems, early in the process can clarify compliance pathways, facilitating a smoother approval process when the product is ready for market entry. Voler Systems has successfully developed hundreds of healthcare instruments up to class III, ensuring that their designs meet stringent regulatory standards.
Statistics indicate that ISO 13485 certification is recognized globally and is vital for companies in the healthcare sector to demonstrate the quality and safety of their products. Furthermore, with the new Quality Management System Regulation (QMSR) set to take effect in February 2026, manufacturers must prioritize aligning their processes with these updated requirements to ensure compliance and maintain a competitive advantage.
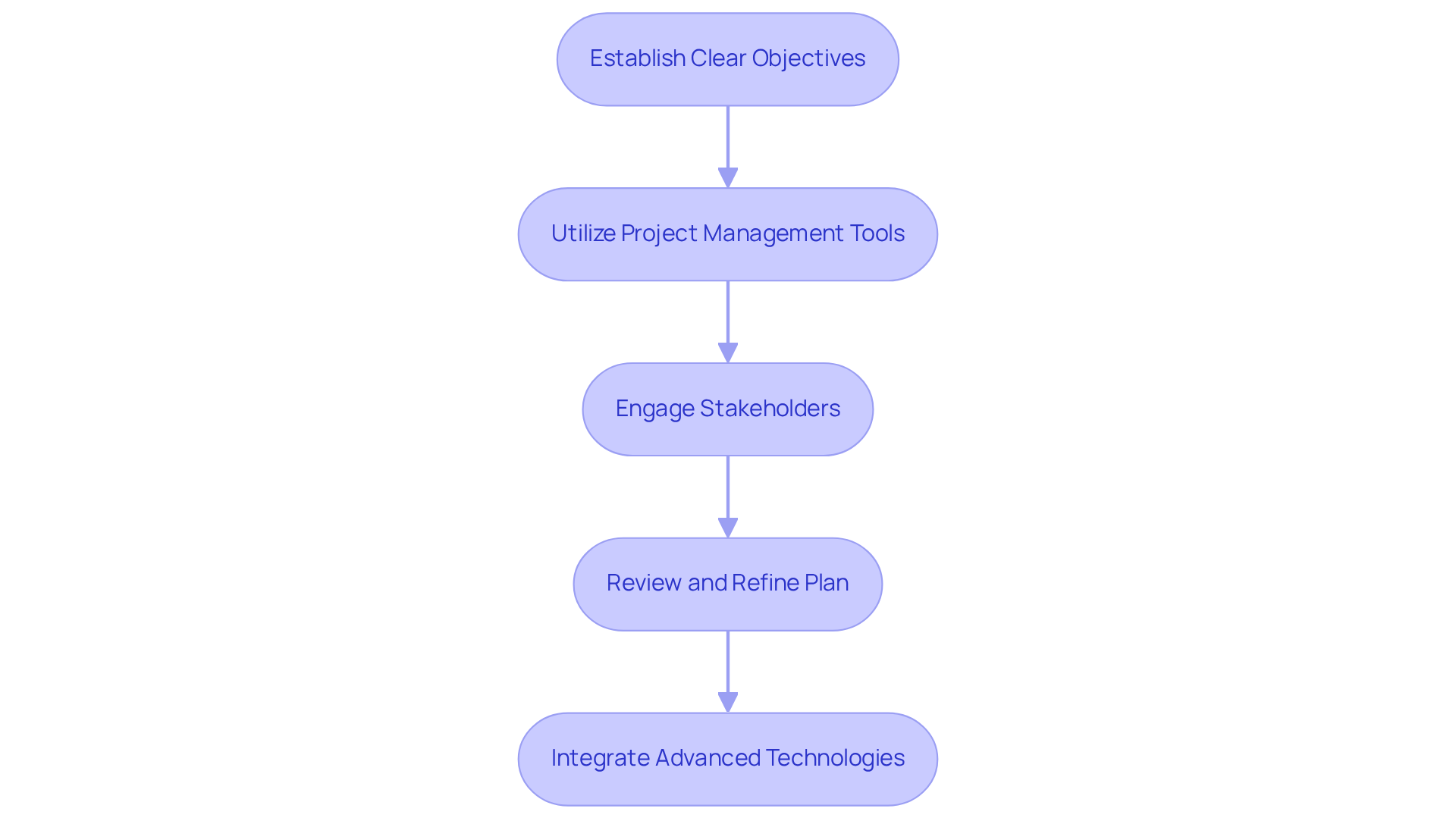
Successful product development prototyping in medical device development hinges on strategic planning that aligns with both business objectives and consumer needs. Begin by establishing clear objectives for your product development prototyping, focusing on aspects such as functionality, user experience, and adherence to regulatory standards. Utilize project management tools, such as Gantt charts, to outline timelines and milestones effectively. Engaging stakeholders early in the product development prototyping process is essential; for example, involving healthcare professionals during the planning phases can yield valuable insights into user requirements and potential challenges. Regularly review and refine your strategic plan for product development prototyping based on stakeholder feedback and testing outcomes to ensure continuous alignment with project goals and market demands. Given that the healthcare equipment sector is projected to grow at a CAGR of 6% from 2025, integrating AI and IoT technologies into your prototypes is vital for maintaining competitiveness. Voler Systems excels in this domain, leveraging innovative embedded systems design with FPGA technology to enhance functionality and optimize battery life in wearable medical devices. Their methodology includes AI-enabled design to create intelligent systems that adapt to complex applications. Furthermore, learning from successful case studies that highlight efficient stakeholder involvement and the integration of advanced technologies can provide practical insights into avoiding common pitfalls in product development prototyping.
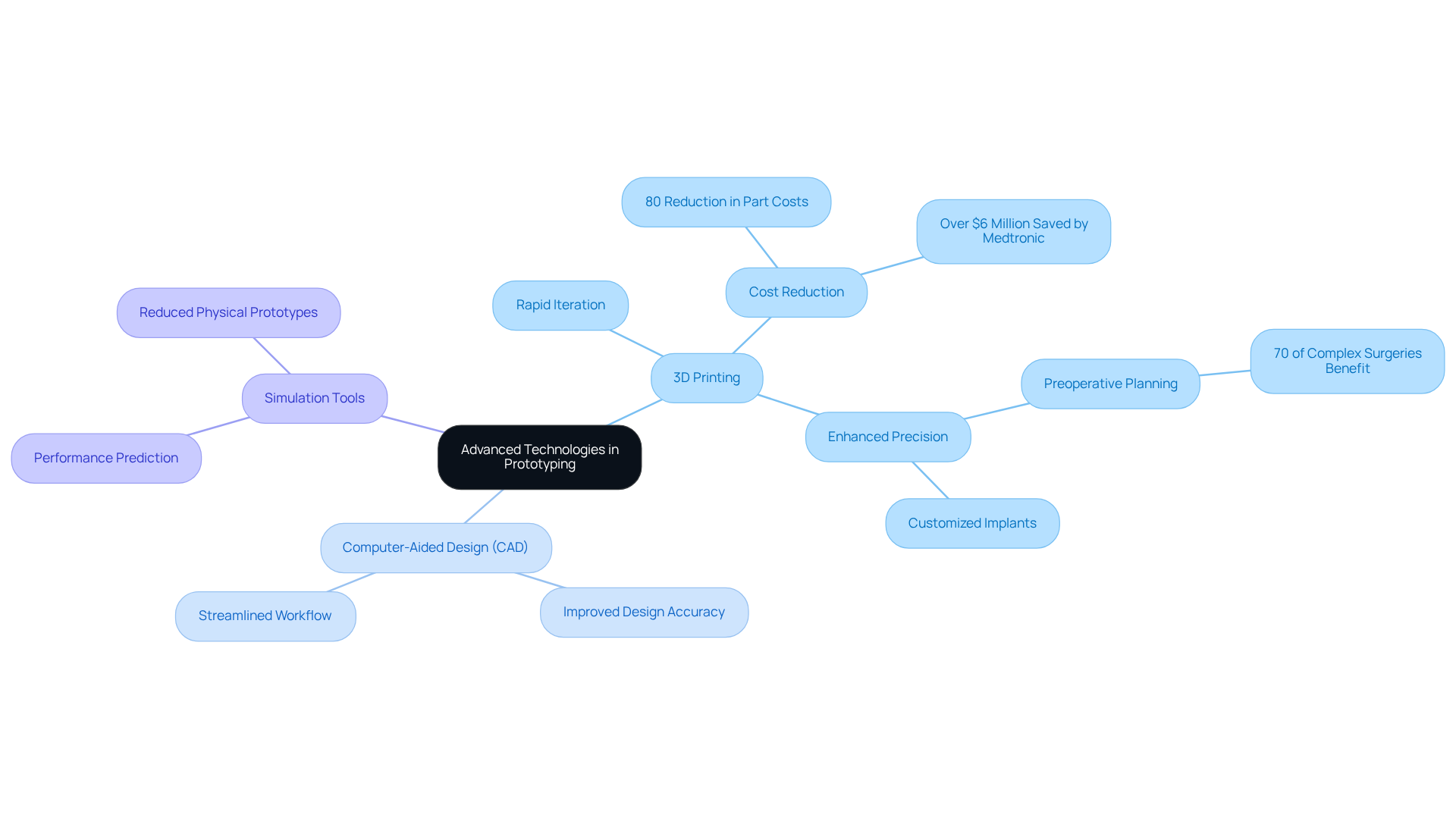
Integrating advanced technologies into the prototyping procedure significantly enhances efficiency and effectiveness. Techniques such as 3D printing and computer-aided design (CAD) enable rapid iteration and testing of prototypes, which is crucial in the medical equipment sector. For instance, 3D printing facilitates the swift production of physical models, allowing for the evaluation of form and fit prior to committing to more expensive manufacturing processes. This capability is particularly advantageous, as nearly 70% of complex surgeries benefit from preoperative planning using 3D-printed models, which enhances precision and minimizes complications.
Moreover, the incorporation of simulation tools can predict performance under various conditions, thereby reducing the need for multiple physical prototypes. This strategy not only accelerates the design cycle but also improves product reliability. Adopting collaborative platforms that enable real-time feedback and communication among team members ensures alignment and informed decision-making throughout the prototyping phase. Medtronic's experience illustrates this, as bringing prototyping in-house has led to an 80% reduction in average part costs and significant savings, underscoring the financial and operational advantages of these technologies in healthcare product development. Additionally, Voler Systems utilizes artificial intelligence (AI) to enhance prototyping processes, resulting in smarter, more efficient solutions tailored to patient needs. 3D printing also allows for the creation of implants and prosthetics specifically designed to fit a patient's unique anatomy, further highlighting its transformative impact on the industry.
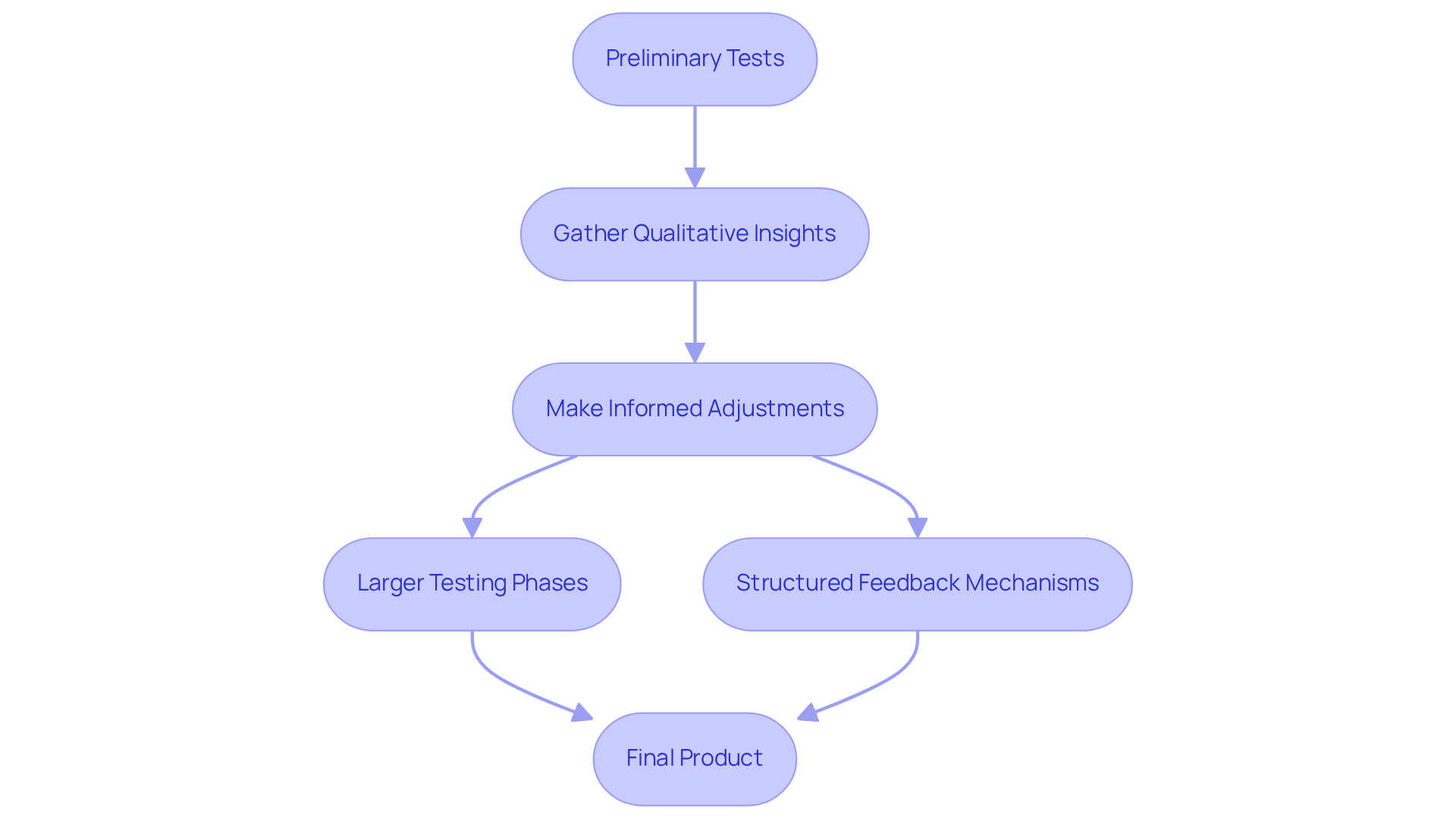
Iterative testing and feedback loops are crucial for a successful prototyping strategy in medical device development. It is essential to begin with preliminary tests involving a selected group of participants to gather qualitative insights regarding the prototype's functionality and usability. This feedback is vital for making informed adjustments prior to advancing to larger testing phases. Structured feedback mechanisms, such as surveys and focus groups, should be implemented to effectively capture insights from participants.
For instance, when assessing the innovative calf-worn tool designed for motion and circumference monitoring in knee replacement rehabilitation, collecting feedback from healthcare professionals who will utilize the tool in practical scenarios is imperative. A systematic review indicates that participant involvement was reported in the development process of 13 commercially available products for epilepsy management, underscoring the significance of integrating feedback from end-users.
This iterative approach in product development prototyping not only identifies potential issues early but also cultivates a culture of continuous improvement, ensuring that the final product aligns closely with client expectations and adheres to stringent regulatory requirements. Furthermore, it is essential to recognize common errors in establishing manufacturing tests, as these can considerably affect the quality and efficiency of the final product. By addressing these challenges and leveraging user insights, manufacturers can enhance the quality and safety of their medical devices, thereby ensuring compliance with industry standards.
In the field of product development prototyping, especially in the medical device sector, a comprehensive approach is essential for success. By emphasizing regulatory compliance, strategic planning, advanced technologies, and iterative testing, companies can establish a robust framework that not only meets industry standards but also aligns with consumer needs. Prioritizing these practices enables effective navigation through the complexities of product development, ensuring that prototypes are both innovative and compliant.
Key insights from this article underscore the necessity of:
Addressing regulatory requirements from the outset helps minimize costly modifications later in the development process. Strategic planning enhances alignment with business objectives and user expectations, while advanced technologies such as 3D printing and AI streamline prototyping and improve product functionality. Lastly, iterative testing promotes continuous improvement, resulting in products that genuinely meet market demands.
The significance of these practices transcends mere compliance or efficiency; they embody a commitment to quality and safety in medical device development. As the industry evolves, integrating these best practices will not only encourage innovation but also provide a competitive advantage. Companies are urged to adopt these strategies proactively, ensuring their prototypes comply with regulations and resonate with users, ultimately facilitating successful market entries and enhancing patient outcomes.
Why is understanding regulatory compliance important in medical device prototyping?
Understanding regulatory compliance is essential in medical device prototyping to ensure that the prototype meets design specifications and complies with safety and efficacy requirements established by regulations such as those from the FDA or ISO standards.
How should one start to ensure regulatory compliance in product development prototyping?
One should begin by identifying the specific regulations pertinent to their product category, such as familiarizing themselves with the FDA's guidelines on software validation and clinical testing for products like wearable health monitors.
What are the benefits of incorporating compliance checks during product development?
Incorporating compliance checks during product development can significantly reduce the risk of costly modifications later in the development cycle.
How can collaborating with electronic design teams aid in regulatory compliance?
Collaborating with expert electronic design teams, like those at Voler Systems, early in the process can clarify compliance pathways and facilitate a smoother approval process when the product is ready for market entry.
What experience does Voler Systems have in developing healthcare instruments?
Voler Systems has successfully developed hundreds of healthcare instruments up to class III, ensuring that their designs meet stringent regulatory standards.
Why is ISO 13485 certification important for companies in the healthcare sector?
ISO 13485 certification is recognized globally and is vital for companies in the healthcare sector to demonstrate the quality and safety of their products.
What upcoming regulation should manufacturers be aware of, and when does it take effect?
Manufacturers should be aware of the new Quality Management System Regulation (QMSR), which is set to take effect in February 2026, and they must prioritize aligning their processes with these updated requirements to ensure compliance and maintain a competitive advantage.
