Master Prototype Product Development: Key Practices for Success
Explore essential practices for effective prototype product development and ensure...

Effective risk management is essential in the competitive realm of product development, especially within the medical device sector, where the stakes are high and the margin for error is minimal. This article explores four best practices that can significantly improve risk management processes, equipping teams with the necessary tools and strategies to navigate uncertainties confidently. Given the evolving regulations and the complexities inherent in modern development, organizations must ensure they are not only identifying risks but also effectively mitigating them.
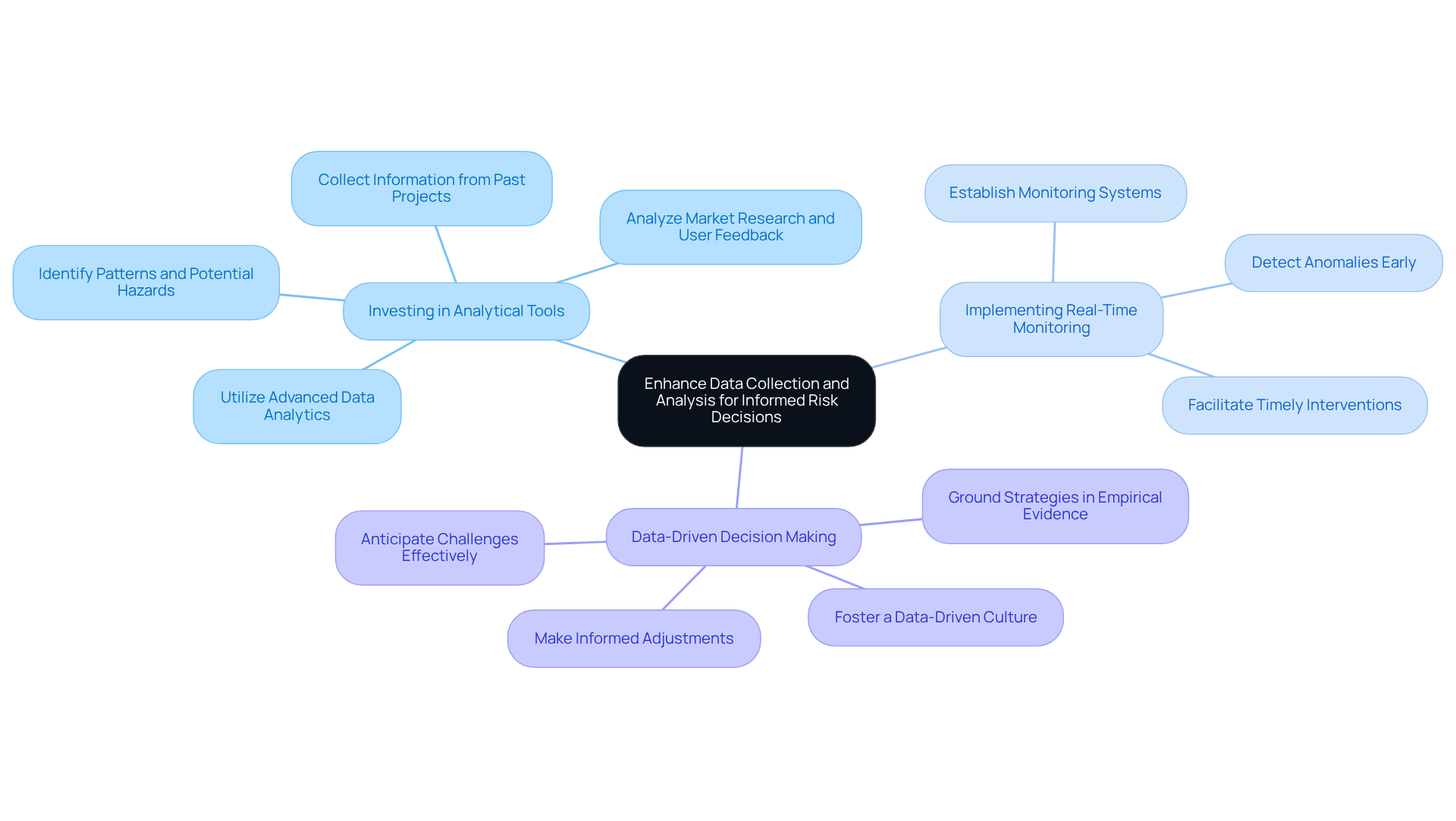
To effectively manage uncertainties in product development, especially within the , it is essential to establish in product development processes. This involves utilizing a range of techniques, including:
Implementing these processes ensures that are identified early, which facilitates that enhance product safety and compliance with rigorous industry standards, thereby improving risk management in product development. By addressing common pitfalls in identifying -such as neglecting ongoing challenges and failing to involve diverse groups-organizations can improve their oversight practices and ultimately increase their chances of success in the competitive medical device market.
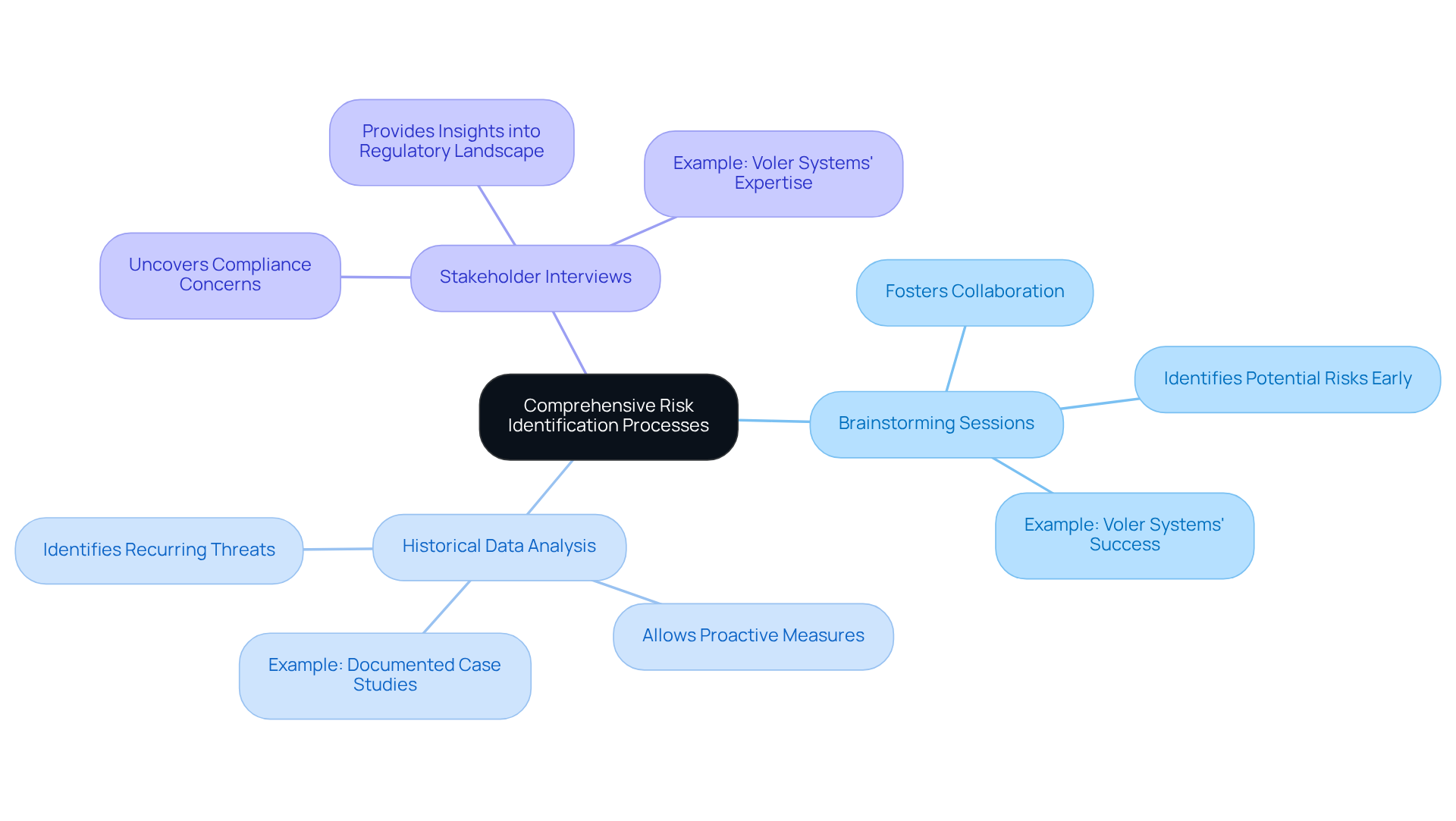
To ensure in , it is essential to define clear ownership and accountability within the group. This can be achieved through several key strategies:
By defining clear ownership and employing organized tools such as the , teams can significantly enhance their responsiveness to threats, ensuring prompt and efficient handling of potential issues. Furthermore, with 57% of participants indicating operational challenges as a primary factor when overseeing subcontractors, it is evident that effective mitigation strategies are essential in today's rapidly evolving sector.
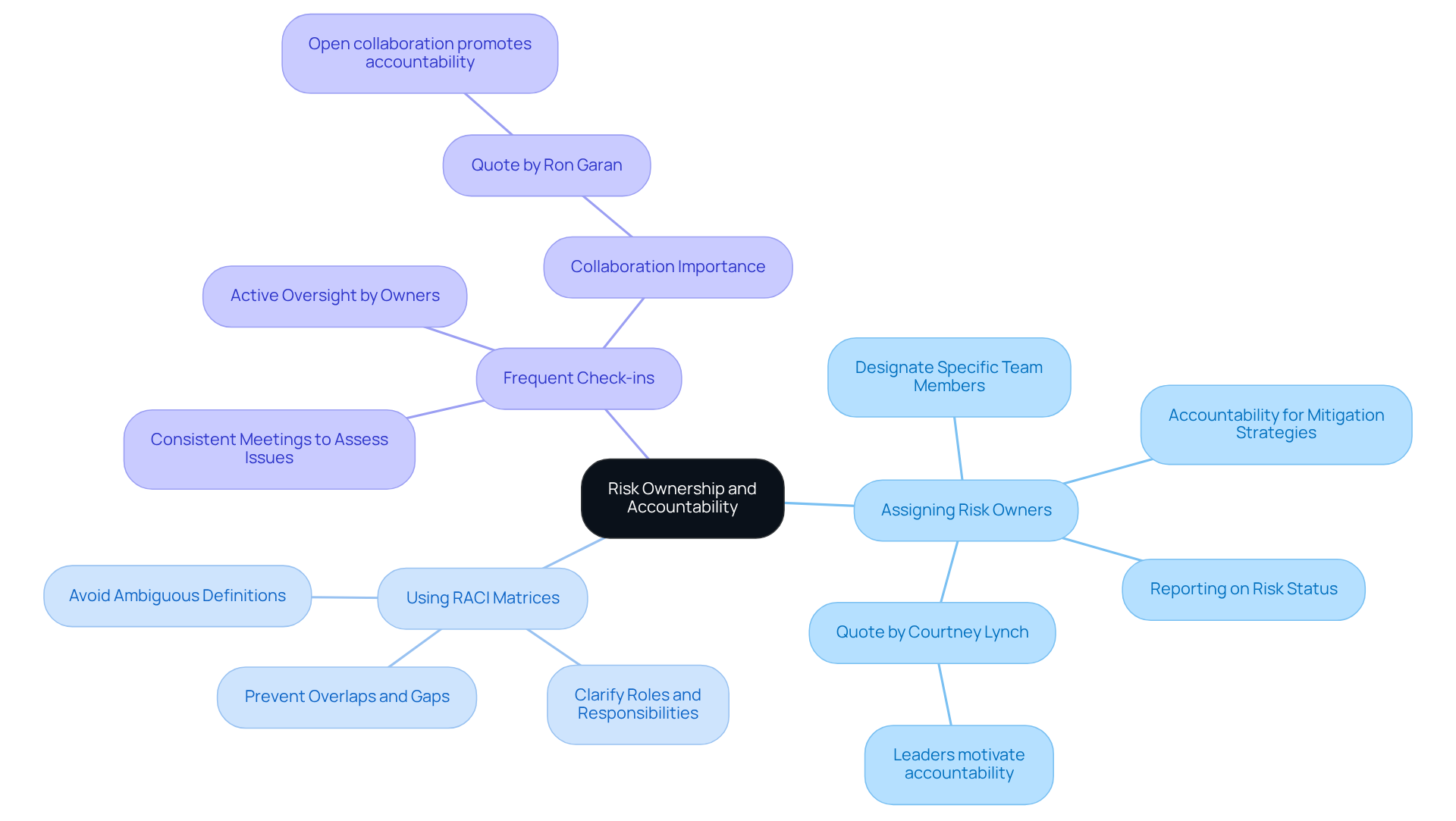
Integrating agile practices into uncertainty handling processes allows teams to effectively address challenges throughout the . Key practices include:
By adopting these agile methodologies, teams can significantly enhance their ability to implement , ensuring proactive management of uncertainties, alignment with project objectives, and adherence to stringent regulatory standards. Data indicates that organizations employing experience higher project success rates, underscoring the effectiveness of these practices within the .
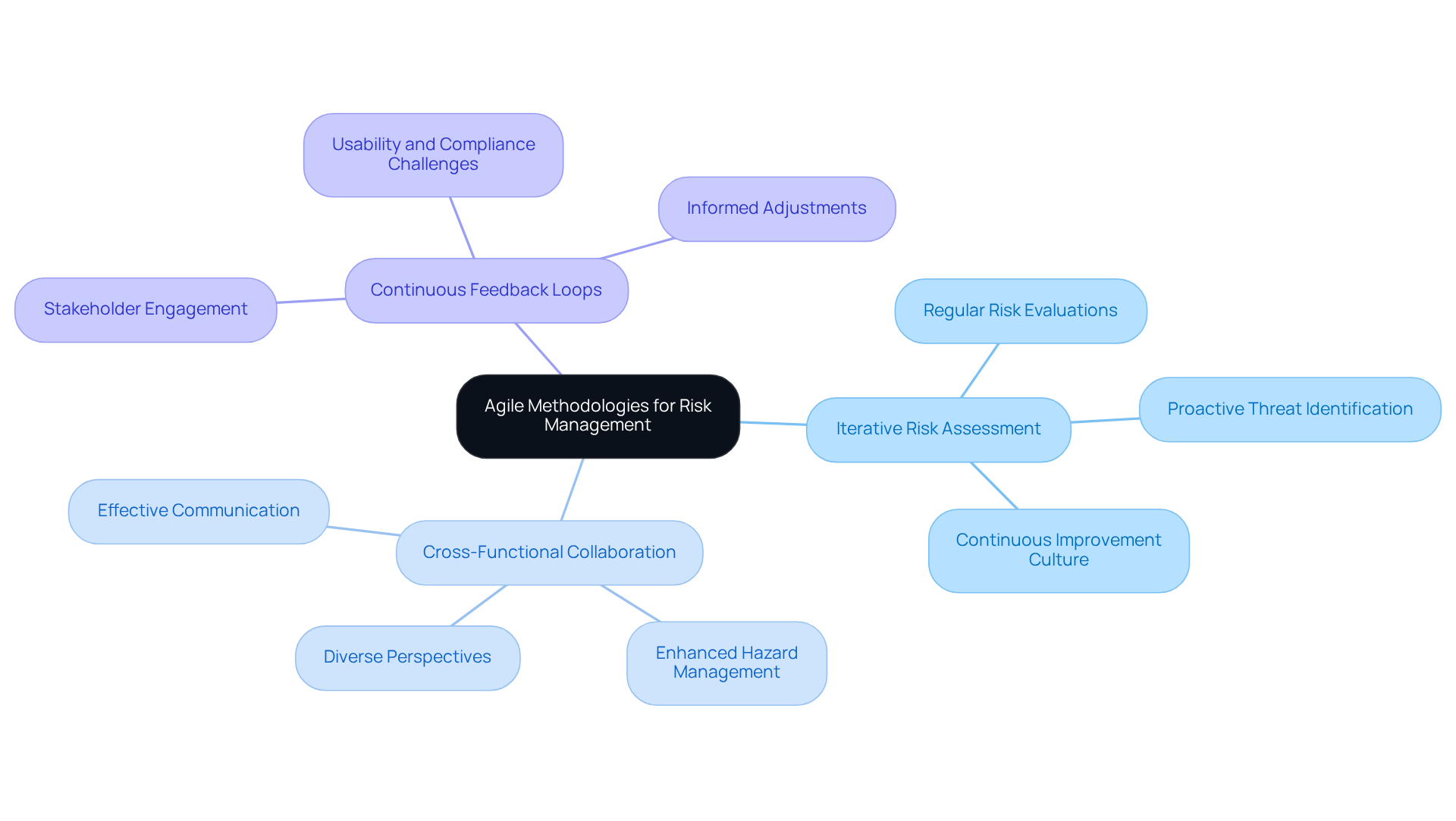
To make informed choices regarding , it is essential to enhance . This can be accomplished through several key strategies:
By improving data collection and analysis, teams can significantly and effectively adapt their risk management approaches.
In product development, establishing effective risk management practices is essential for navigating uncertainties, especially in high-stakes industries such as medical devices. By implementing comprehensive risk identification processes, organizations can proactively address potential challenges, ensuring that safety and compliance are prioritized throughout the development lifecycle.
This article highlights four key best practices:
Each of these practices plays a vital role in fostering a culture of continuous improvement and collaboration, ultimately leading to more informed decision-making and successful project outcomes.
Embracing these strategies not only mitigates risks but also positions organizations to thrive in a competitive market. As the landscape of product development continues to evolve, integrating these best practices will be essential for maintaining compliance, enhancing product safety, and driving innovation. Organizations are encouraged to prioritize these approaches to ensure robust risk management and achieve sustainable success in their product development endeavors.
Why is risk identification important in product development, especially in the medical device sector?
Risk identification is crucial in product development to manage uncertainties effectively, ensuring product safety and compliance with industry standards.
What techniques can be used for comprehensive risk identification?
Techniques include brainstorming sessions, historical data analysis, and stakeholder interviews, which collectively help identify potential risks early in the development process.
How do brainstorming sessions contribute to risk identification?
Brainstorming sessions engage cross-functional teams, fostering collaboration and open dialogue that allows diverse expertise to identify potential risks and challenges.
What role does historical data analysis play in risk management?
Historical data analysis helps teams identify recurring threats and their impacts from previous projects, enabling proactive measures to mitigate similar issues in new projects.
Why are stakeholder interviews important in the risk identification process?
Stakeholder interviews, including discussions with regulatory bodies, provide valuable insights into compliance-related concerns and expectations, ensuring that products meet industry standards.
How can organizations improve their oversight practices in risk management?
Organizations can improve oversight by addressing common pitfalls, such as neglecting ongoing challenges and failing to involve diverse groups, which enhances their risk management strategies.
What benefits do comprehensive risk identification processes offer in product development?
These processes facilitate early identification of potential issues, allowing for timely mitigation strategies that enhance product safety and improve compliance with rigorous industry standards.
