4 Benefits of Low Power VLSI Design for Medical Devices
Introduction Low power VLSI design is transforming the medical device sector, where the...

The healthcare sector is undergoing a significant transformation as advanced technologies reshape the landscape of medical devices. The global AI healthcare market is projected to exceed $187 billion by the end of the decade, underscoring the necessity for engineers to integrate VLSI design flows to enhance patient care and comply with stringent regulatory standards. However, the complexities inherent in VLSI design introduce unique challenges. This raises an important question: what critical considerations must medical device engineers prioritize to achieve successful outcomes in this rapidly evolving field?
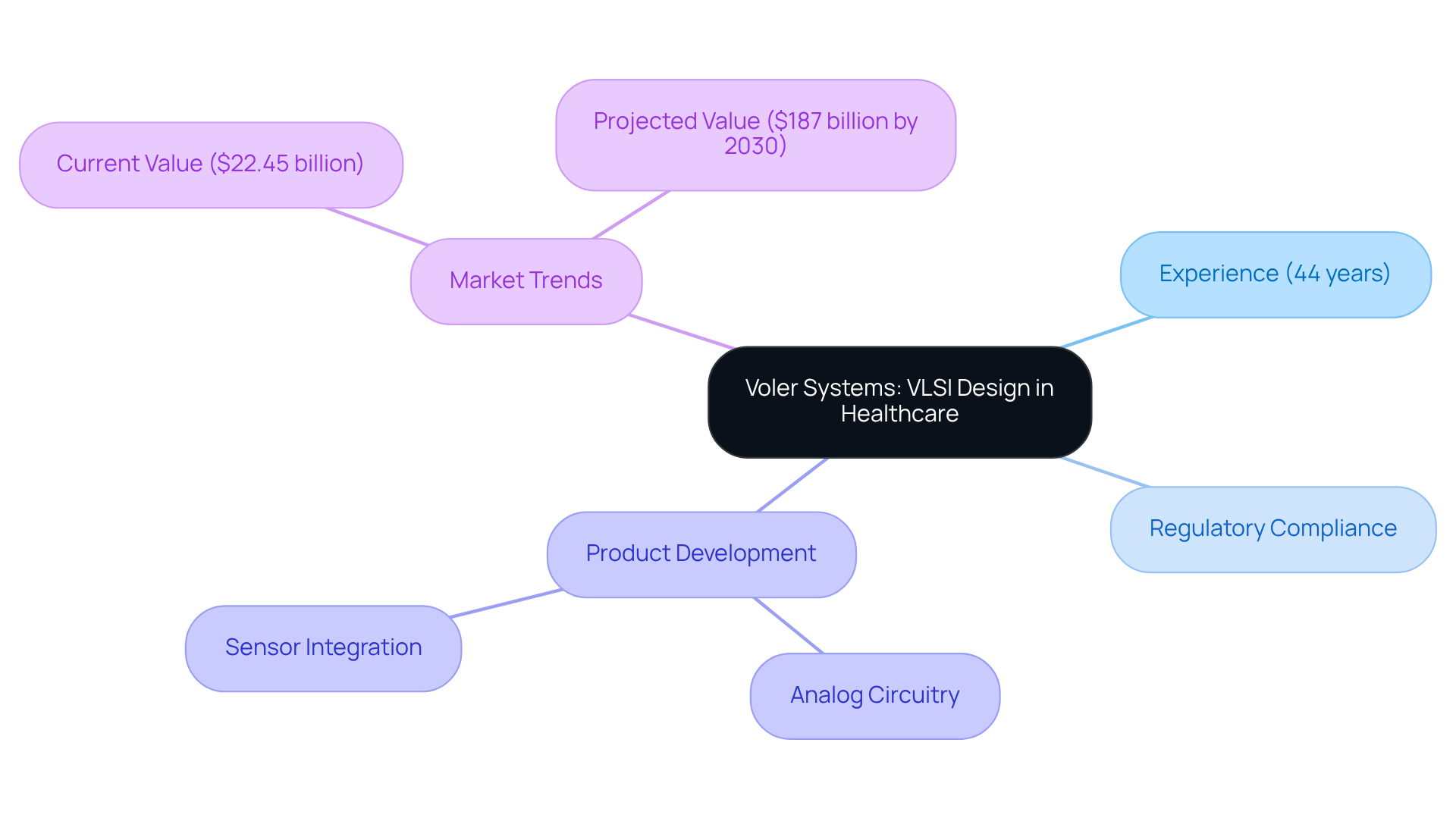
The company leads in VLSI development within the healthcare sector, leveraging over 44 years of experience to integrate advanced techniques that drive innovation. This approach not only guarantees functionality but also complies with stringent regulatory standards, which are essential in healthcare technology. By applying expertise in analog circuitry and sensor integration, the company develops products that significantly enhance patient care and streamline healthcare processes.
The global AI healthcare market, valued at $22.45 billion in 2023, highlights the increasing significance of such innovations, with forecasts suggesting it will surpass $187 billion by the decade's end. This trend underscores the growing reliance on the VLSI design flow notes and advanced development processes to meet the evolving demands of the healthcare sector. As we approach 2026, the focus on real-time integrated frameworks and ultra-low power solutions continues to shape the industry landscape, positioning Voler Systems as a leader in delivering high-quality, compliant healthcare products that improve health outcomes.
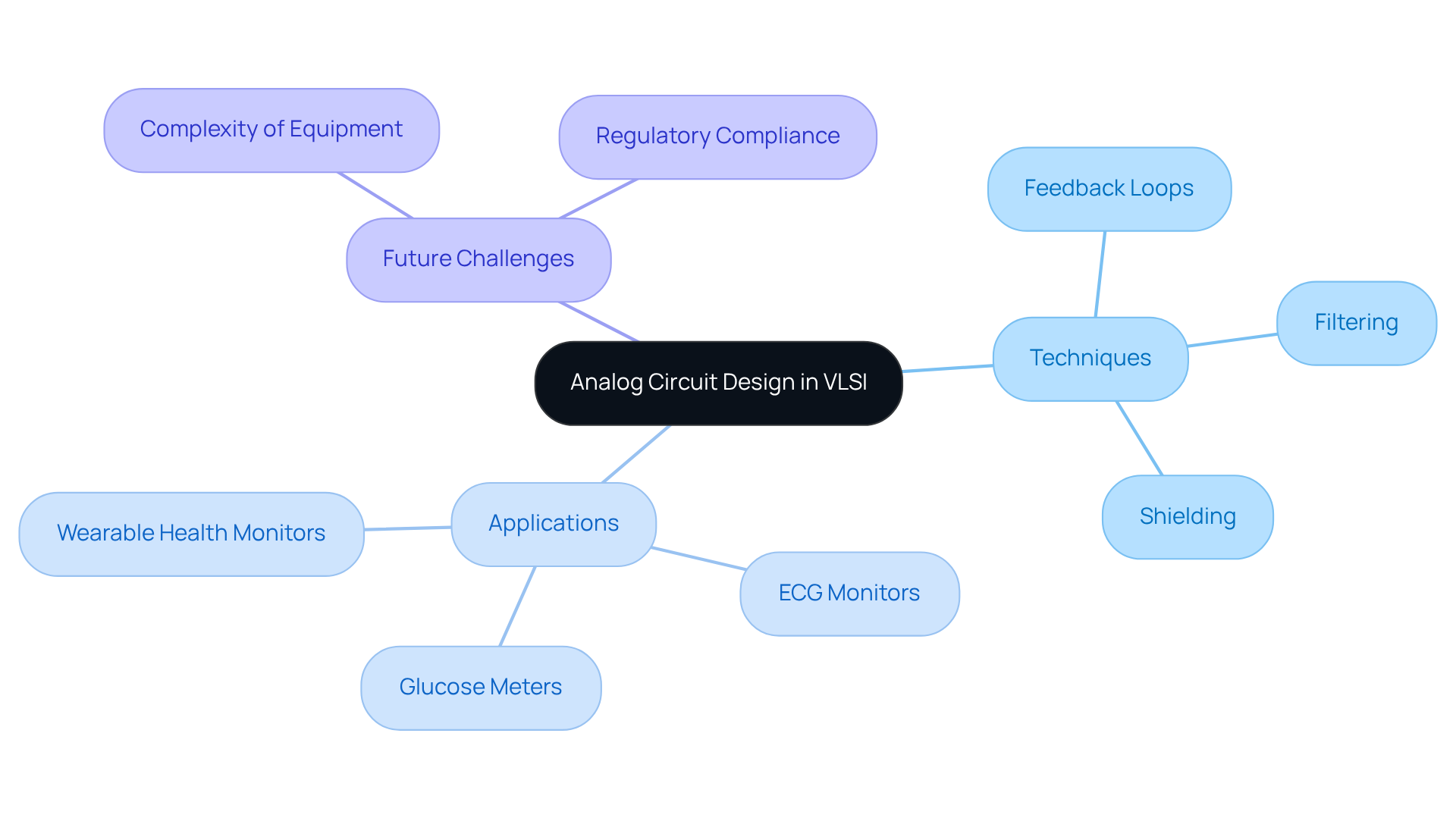
Analog circuitry serves as a cornerstone of VLSI systems, particularly within medical apparatus that demand high precision in signal processing. The integrity of sensor readings is contingent upon minimizing noise and distortion, factors that can significantly affect diagnostic accuracy. Engineers utilize various techniques, including:
to enhance signal fidelity and mitigate unwanted interference. For instance, in wearable health monitors, analog circuits are pivotal as they convert physiological signals into digital data, enabling real-time monitoring and diagnostics. This capability is crucial for ensuring that instruments such as ECG monitors and glucose meters deliver reliable and precise health information, ultimately improving patient outcomes.
As we look toward 2026, challenges in analog circuitry will include addressing the increasing complexity of equipment and ensuring compliance with stringent regulatory standards. This necessitates robust engineering practices to minimize the risks of equipment failure or malfunction. By implementing strategies to enhance signal integrity, similar to those used in handheld HVAC testers, engineers can further augment the functionality of health-related equipment while adeptly navigating the regulatory landscape.
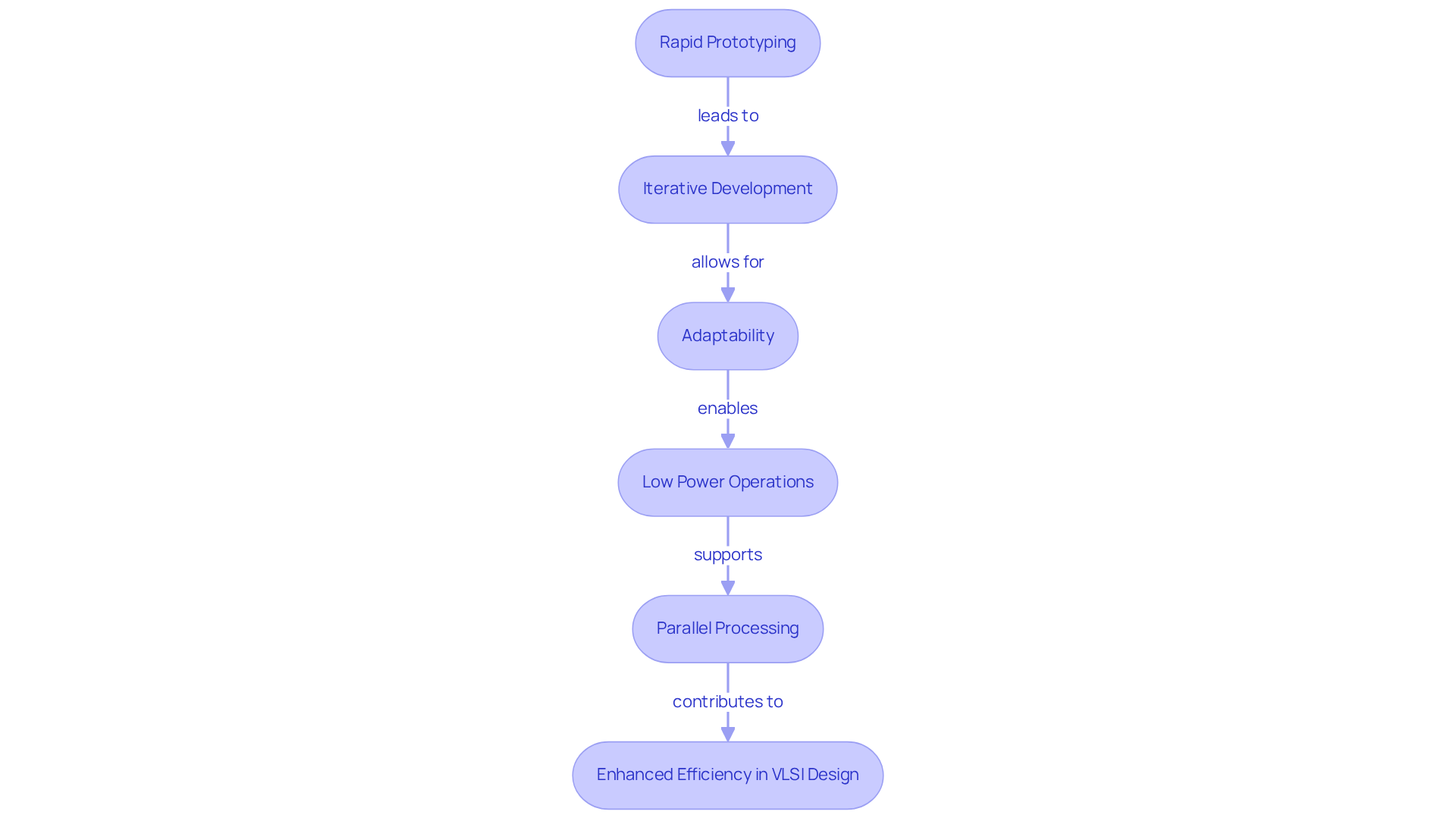
Field Programmable Gate Arrays (FPGAs) are a vital resource in VLSI development, particularly within the medical equipment sector, as they enable rapid prototyping and testing. Their reconfigurable nature allows engineers to modify hardware even after manufacturing, which is crucial for iterative development processes. For example, in the creation of a cardiac monitoring system, engineers can quickly adjust the FPGA to assess different algorithms for signal processing. This adaptability ensures that the device achieves optimal performance prior to finalizing the layout, thereby significantly enhancing the efficiency of the development cycle.
Moreover, FPGAs support low power operations, making them ideal for battery-powered devices, which increases their relevance in critical healthcare applications. Voler Systems employs a prototype approach to refine hardware development and design modifications, ensuring that healthcare products not only meet regulatory standards but also adapt to the evolving needs of healthcare professionals. Additionally, the parallel processing capabilities of FPGAs provide high throughput and low latency for specialized tasks, further augmenting their utility in time-sensitive healthcare environments.
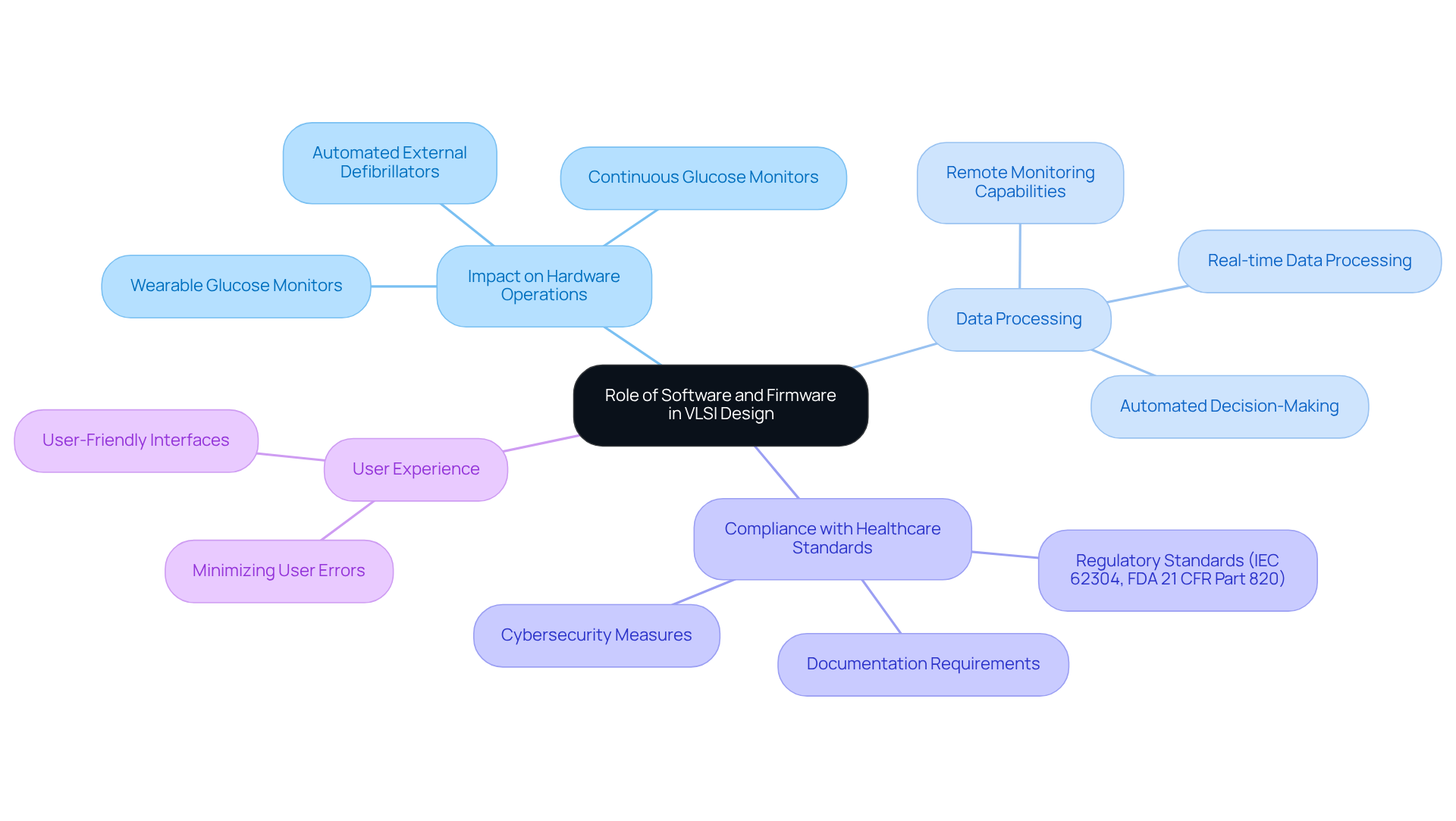
Software and firmware are essential components in the functionality of VLSI-designed health equipment, directly impacting hardware operations, data processing, and compliance with healthcare standards. For instance, in wearable glucose monitors, firmware is pivotal for processing sensor data and enabling communication with mobile applications. This integration not only enhances user experience but also significantly boosts the reliability of the equipment.
Engineers must prioritize effective software architecture during the VLSI design flow notes to ensure that these products meet the stringent standards required for healthcare applications. Successful implementations of firmware in wearable glucose monitors demonstrate that meticulous planning can lead to improved patient outcomes, underscoring the necessity for engineers to focus on this aspect throughout the development cycle.
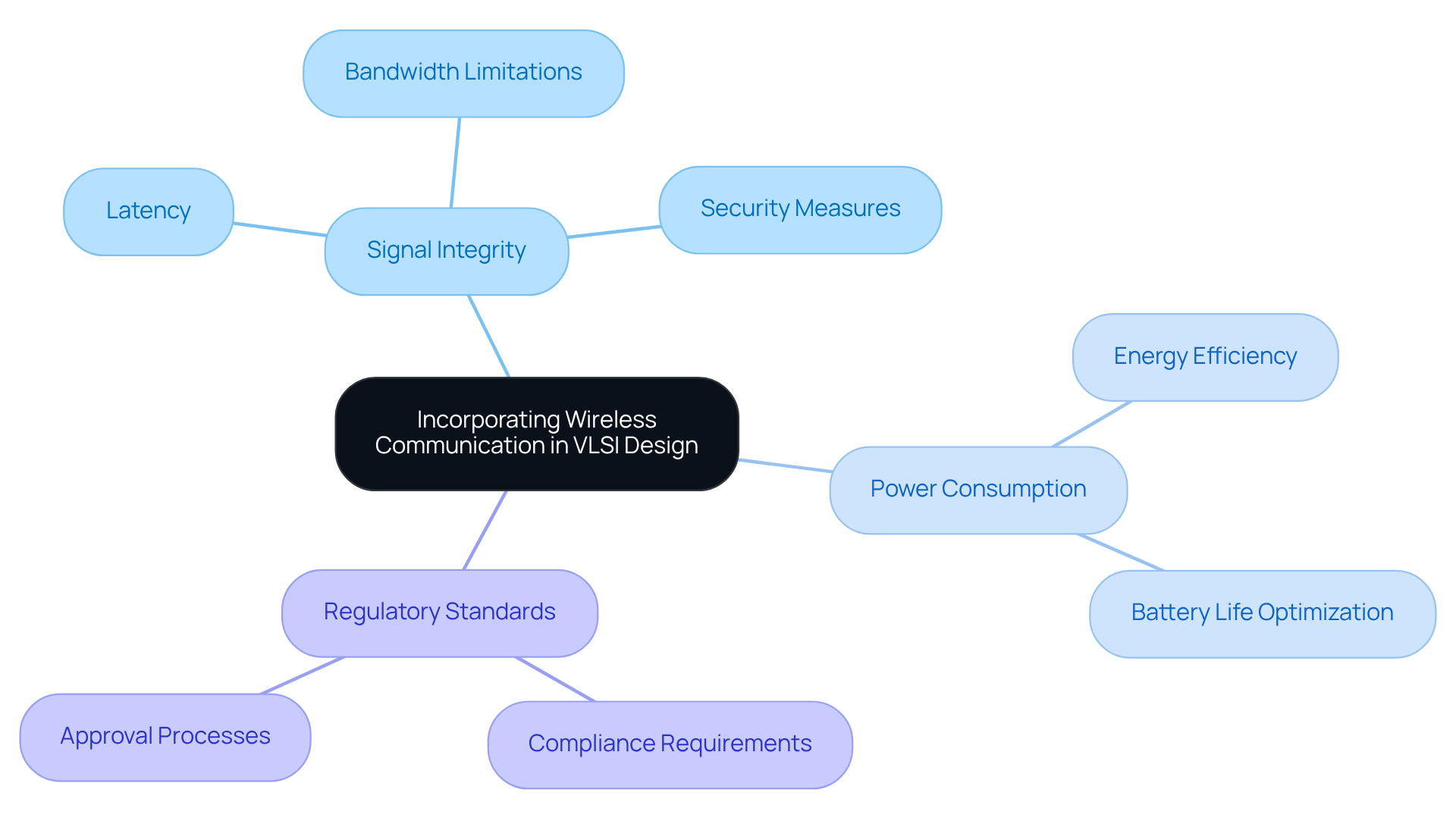
Wireless communication serves as a cornerstone of contemporary medical technology, facilitating real-time data transmission and remote patient monitoring. Engineers at Voler Systems must prioritize several key factors outlined in the VLSI design flow notes when integrating wireless technologies into VLSI designs. These factors include:
In telehealth applications, for example, maintaining reliable communication between systems and healthcare providers is crucial for ensuring patient safety and delivering effective treatment.
The incorporation of wireless technology enhances the functionality of medical equipment and promotes proactive patient care through continuous monitoring. This is particularly evident in advancements such as the transition from tethered systems to genuinely wireless cardiac monitoring solutions. However, challenges such as latency, bandwidth limitations, and the necessity for robust security measures can complicate the integration process. These issues may transform network infrastructure into a bottleneck for AI in healthcare, with projected spending rising from $6.5 billion in 2024 to $30.9 billion by 2029.
Addressing these challenges is essential for optimizing the performance of wireless-enabled VLSI design flow notes in healthcare settings. Furthermore, linking equipment through the Internet of Medical Things (IoMT) is vital for facilitating proactive, data-informed patient care.
Effective sensor integration is crucial for the performance of VLSI-designed medical equipment, as outlined in the VLSI design flow notes. Engineers at the company ensure that sensors are accurately calibrated and properly interfaced, following the guidelines outlined in the VLSI design flow notes.
For example, in a wearable heart rate monitor, the integration of photoplethysmography sensors necessitates careful consideration of signal processing algorithms to guarantee accurate heart rate readings. This integration not only enhances the overall functionality and reliability of the device but also utilizes AI-assisted engineering to optimize performance.
By addressing environmental challenges and employing ultra-low-power approaches, Voler Systems is committed to transforming modern healthcare through innovative wearable technology.
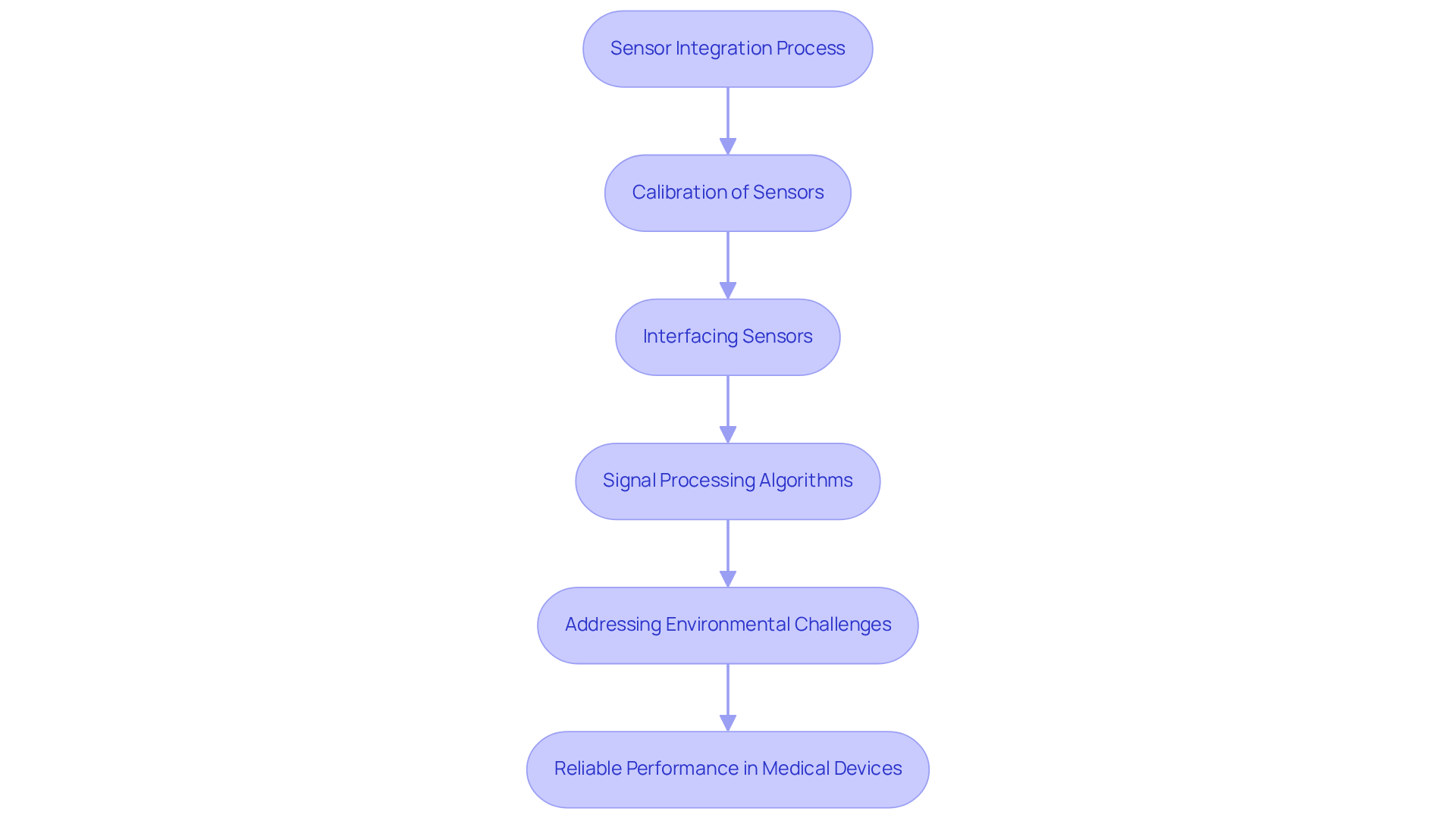
Motion control systems play a vital role in the development of healthcare instruments, particularly in ensuring precise movements and positioning. In the context of VLSI design flow notes, engineers must emphasize the integration of actuators and feedback systems to achieve optimal motion characteristics. For example, in robotic surgical systems, precise motion control is essential for performing delicate procedures with accuracy. The implementation of robust algorithms is critical to guarantee that these motion control systems operate reliably and safely, ultimately improving patient outcomes and operational efficiency. Recent advancements in motion control technology have significantly enhanced the capabilities of robotic systems, enabling more sophisticated and adaptable surgical solutions.
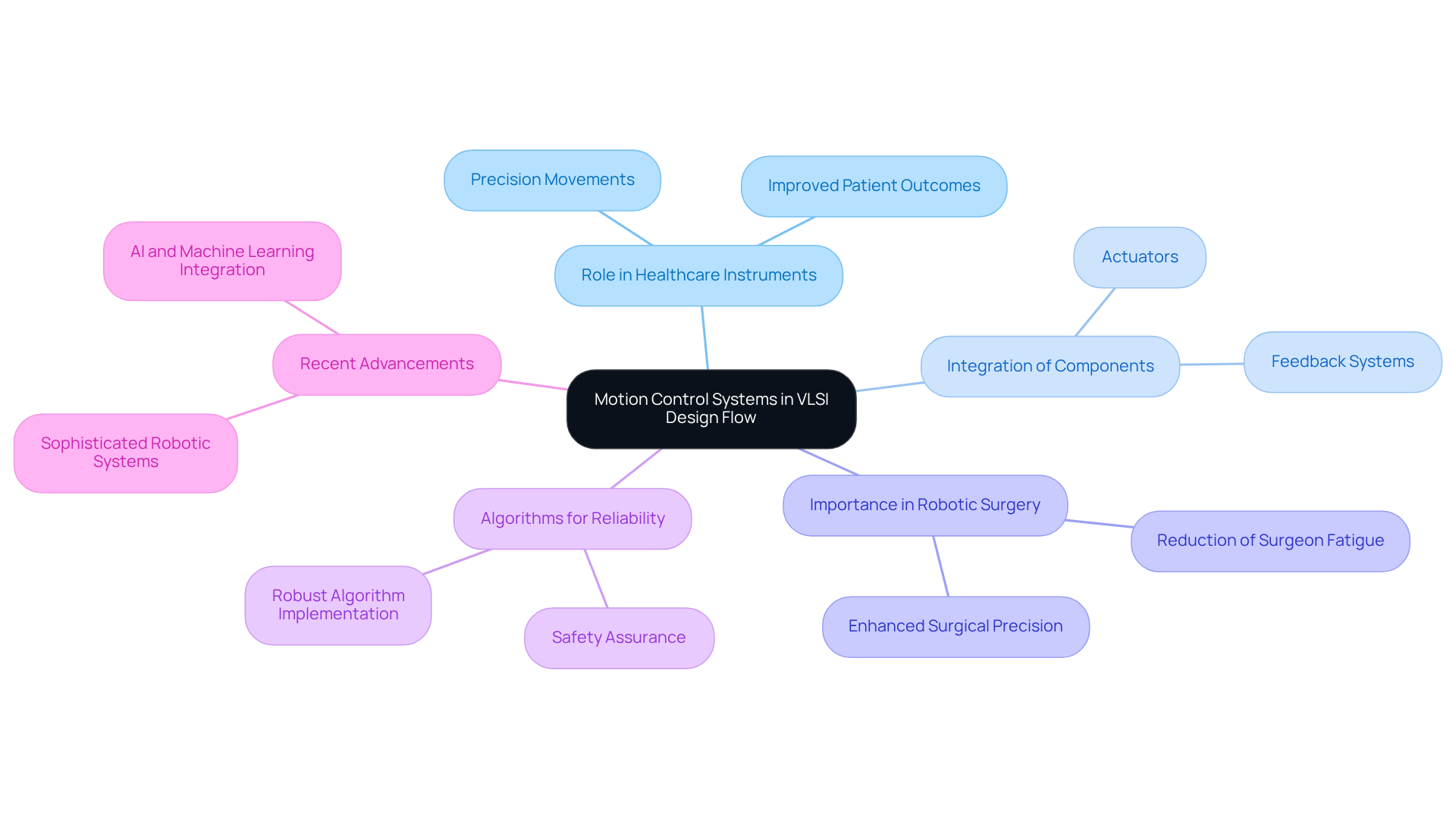
Design for manufacturability (DFM) is a critical component of VLSI development, especially within the medical equipment sector, where adherence to stringent quality standards is paramount. At Voler Systems, our experienced engineers prioritize the optimization of concepts, layouts, and material selection to enhance manufacturability. For instance, in the development of wearable technologies, heart pumps, and liquid biopsy platforms, meticulous planning not only reduces manufacturing costs but also significantly improves product reliability.
The successful implementation of DFM can streamline the manufacturing process, which is vital considering that the Medical Device Manufacturing industry in the United States is anticipated to reach a market size of $58.0 billion by 2026, with a compound annual growth rate (CAGR) of 1.7% from 2021 to 2026. Early integration of DFM principles during the planning phase is essential for achieving optimal outcomes, as it fosters improved communication with suppliers and mitigates risks associated with manufacturability challenges. Companies that adopt DFM strategies are better equipped to navigate the complexities of the healthcare equipment sector, ensuring compliance and enhancing operational efficiency.
Ultra-low power architecture is crucial for medical equipment, particularly those reliant on battery operation. Engineers at a technology firm implement strategies such as:
to minimize energy consumption. These strategies take into account user behavior and environmental factors that affect the effectiveness and durability of wearable sensors. For instance, in wearable health monitors, optimizing power usage significantly extends battery life, enabling continuous monitoring without the need for frequent recharging. This capability is vital for enhancing patient adherence and the overall effectiveness of the equipment.
By leveraging AI-enabled electronic design, Voler Systems develops ultra-low-power wearable sensors that effectively address environmental challenges, including moisture and temperature variations. This ensures reliable performance across diverse settings. We work closely with clients to create customized electronic and embedded software solutions tailored to their wearable devices.
AI-driven engineering is fundamentally transforming the creation of Very Large Scale Integration (VLSI) by automating complex tasks and streamlining workflows. Machine learning algorithms empower engineers to predict outcomes, enhance performance, and significantly reduce time-to-market. For instance, in developing innovative wearable health devices, such as a calf-worn gadget designed for motion and circumference tracking during knee replacement rehabilitation, AI can analyze extensive datasets to identify optimal specifications, resulting in more efficient and effective solutions.
As the healthcare equipment industry faces rapid technological advancements and regulatory scrutiny in 2026, the adoption of AI technologies becomes essential for engineers aiming to maintain a competitive edge and foster innovation. Furthermore, cross-disciplinary collaboration among engineers, regulatory experts, and clinicians is vital for successful healthcare product development, ensuring compliance with evolving regulatory requirements. The growing demand for compact healthcare instruments and a focus on user-centered design further underscore the importance of integrating AI into the development process.
However, engineers must remain cognizant of the potential risks and uncertainties introduced by AI, including issues related to data quality and algorithm transparency. To effectively implement AI-driven engineering, medical device manufacturers, including those collaborating with Voler Systems, should prioritize regulatory-ready design from the outset, aligning their processes with standards such as FDA and ISO to mitigate risks and enhance product quality.
The exploration of VLSI design flow in medical devices underscores its critical role in advancing healthcare technology. By integrating advanced techniques and adhering to stringent regulatory standards, engineers can develop innovative solutions that enhance patient care while ensuring compliance and functionality. The insights presented throughout this article highlight the necessity of a comprehensive approach, where each component - from analog circuitry to AI-driven engineering - contributes to the overall success of medical device development.
Key arguments emphasize the importance of:
Furthermore, the incorporation of wireless communication and effective sensor integration significantly enhances device functionality and user experience. Challenges such as manufacturability and power management are addressed, highlighting the need for engineers to adopt best practices and innovative strategies to navigate these complexities.
As the healthcare sector continues to evolve, the demand for cutting-edge VLSI design becomes increasingly essential. Embracing these critical design considerations not only fosters innovation but also drives improvements in patient outcomes. Medical device engineers are encouraged to leverage these insights and adopt a proactive approach in implementing VLSI design flow notes, ensuring they remain at the forefront of technological advancements while meeting the ever-changing needs of the healthcare industry.
What is Voler Systems known for in the healthcare sector?
Voler Systems is known for pioneering VLSI (Very Large Scale Integration) design flow in medical devices, leveraging over 44 years of experience to integrate advanced techniques that drive innovation and ensure compliance with regulatory standards.
How does Voler Systems enhance patient care through its products?
The company develops products that significantly enhance patient care by applying expertise in analog circuitry and sensor integration, streamlining healthcare processes and improving health outcomes.
What is the current state of the global AI healthcare market?
The global AI healthcare market was valued at $22.45 billion in 2023, with forecasts suggesting it will surpass $187 billion by the end of the decade, indicating a growing reliance on VLSI design and advanced development processes in healthcare.
What are the challenges facing analog circuitry in VLSI design as we approach 2026?
Challenges include addressing the increasing complexity of medical equipment and ensuring compliance with stringent regulatory standards, which necessitates robust engineering practices to minimize risks of equipment failure or malfunction.
Why is analog circuitry important in medical devices?
Analog circuitry is crucial for high precision in signal processing, as it minimizes noise and distortion, which can significantly affect diagnostic accuracy in devices like ECG monitors and glucose meters.
How do engineers enhance signal fidelity in analog circuits?
Engineers enhance signal fidelity using techniques such as feedback loops, filtering, and shielding to mitigate unwanted interference and improve the integrity of sensor readings.
What role do FPGAs play in VLSI development for medical equipment?
FPGAs (Field Programmable Gate Arrays) are vital for rapid prototyping and testing in VLSI development, allowing engineers to modify hardware even after manufacturing, which is essential for iterative development processes.
How do FPGAs contribute to the efficiency of healthcare product development?
FPGAs allow for quick adjustments in hardware for different algorithms, ensuring optimal performance before finalizing the layout, thus enhancing the efficiency of the development cycle.
What advantages do FPGAs offer for battery-powered medical devices?
FPGAs support low power operations, making them ideal for battery-powered devices, which increases their relevance in critical healthcare applications.
How does Voler Systems ensure that its healthcare products meet regulatory standards?
Voler Systems employs a prototype approach to refine hardware development and design modifications, ensuring that products not only meet regulatory standards but also adapt to the evolving needs of healthcare professionals.
