Overview: Wearable Device Sensors Presentation | Voler Systems
Wearable fitness devices, once primarily focused on tracking steps and physical activity,...

Navigating the intricate landscape of medical device development presents significant challenges, particularly concerning regulatory compliance and market readiness. Consultants specializing in medical devices, such as those from Voler Systems, provide critical expertise that can streamline this process. Their involvement ensures that products not only adhere to safety standards but also achieve market entry efficiently.
However, the real challenge lies in effectively utilizing this expertise. How can companies forge productive partnerships that enhance collaboration and lead to successful outcomes? This article delves into best practices for engaging with medical device consultants, offering insights into:
These practices can significantly improve the development journey.
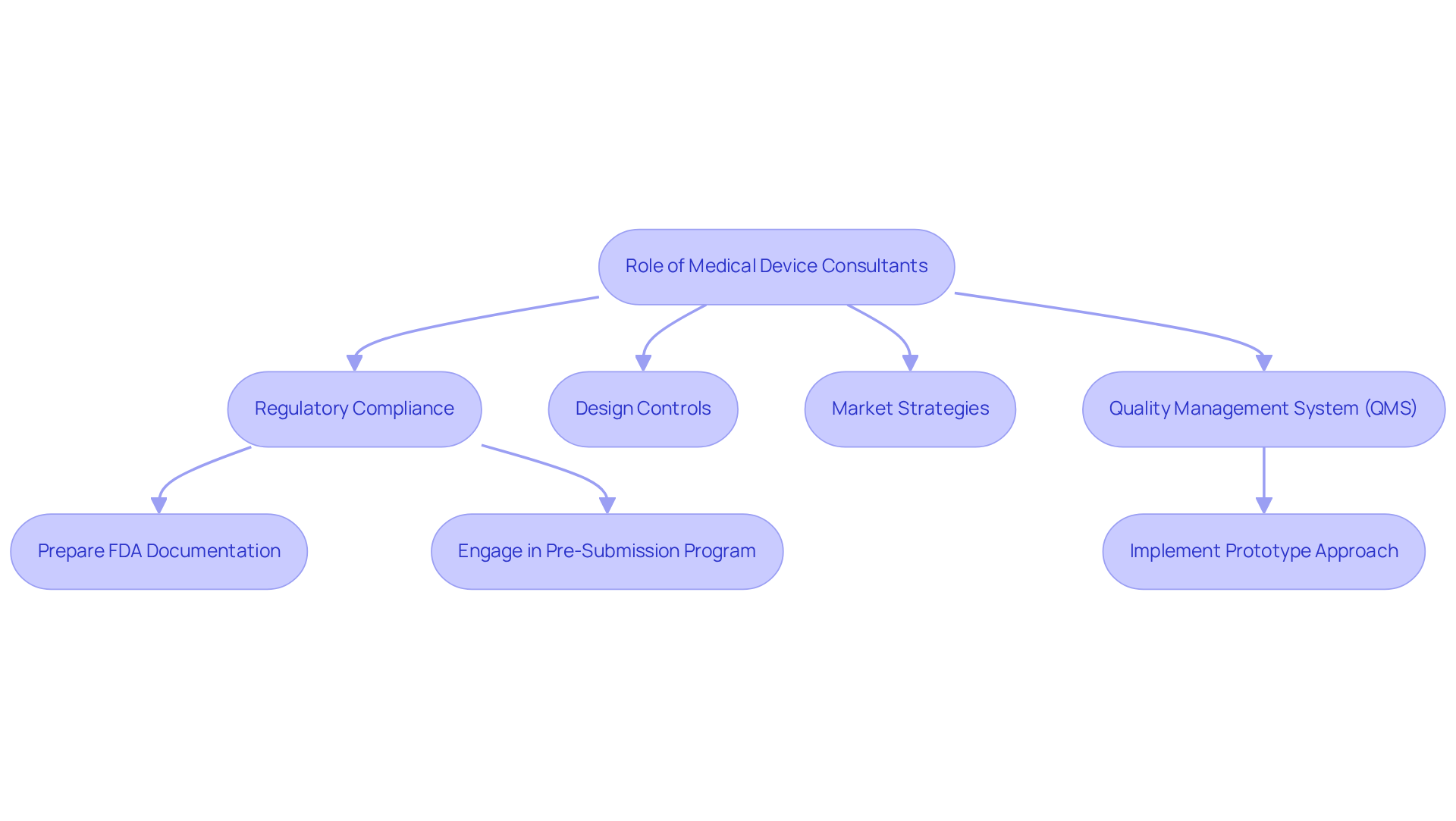
Healthcare equipment advisors, including like Voler Systems, play a pivotal role in the successful development and commercialization of healthcare products. They provide specialized expertise in , design controls, and market strategies. Their deep understanding of the intricate landscape of is vital for ensuring that products meet safety and efficacy standards.
For example, consultants are instrumental in preparing documentation for , a critical step for securing . The FDA Pre-Submission Program facilitates early engagement with the agency, which can streamline the submission process and potentially shorten the typical 90-day wait for feedback associated with traditional 510(k) submissions. By leveraging the expertise of , companies can enhance the efficiency of , improve compliance success rates, and ultimately reduce time to market, establishing these consultants as invaluable partners in the healthcare equipment sector.
A robust (QMS) is essential for the success of , further underscoring the consultant's role in ensuring adherence to regulatory standards. Additionally, Voler Systems promotes a that simplifies hardware development and design modifications, ensuring compliance is maintained from prototype to production in wearable healthcare products.
For instance, in a recent initiative, Voler Systems successfully guided a startup through the FDA submission process, illustrating the effectiveness of their consulting services in practical applications.
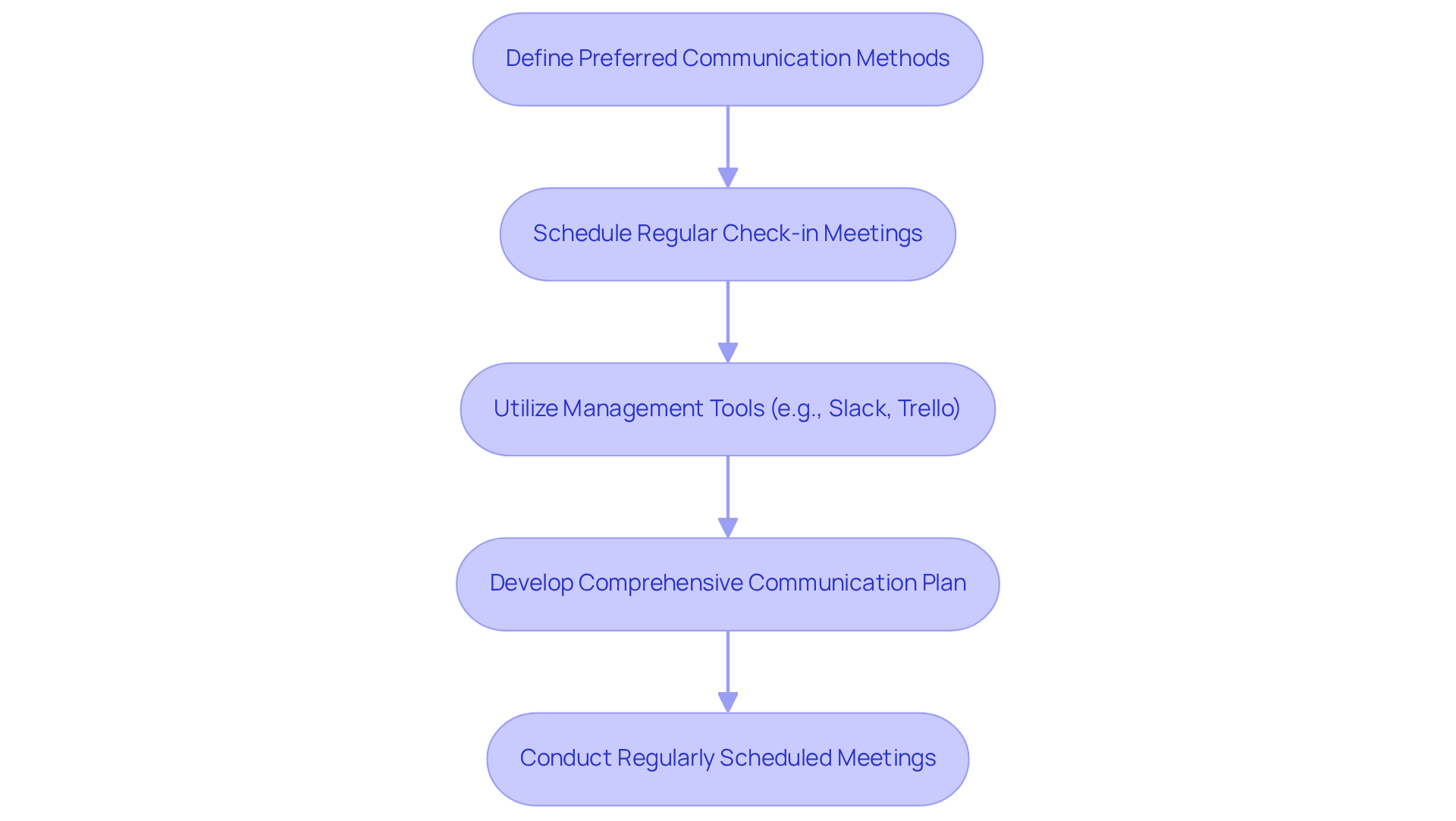
Establishing clear is essential for a successful partnership with . This process begins with and scheduling to ensure alignment. Utilizing such as Slack or Trello facilitates real-time updates and feedback, thereby enhancing collaboration.
Furthermore, developing a that delineates roles, responsibilities, and expectations is crucial to prevent misunderstandings. Regularly scheduled meetings provide opportunities to address concerns and adjust scopes, ensuring that all parties remain aligned throughout the project lifecycle.
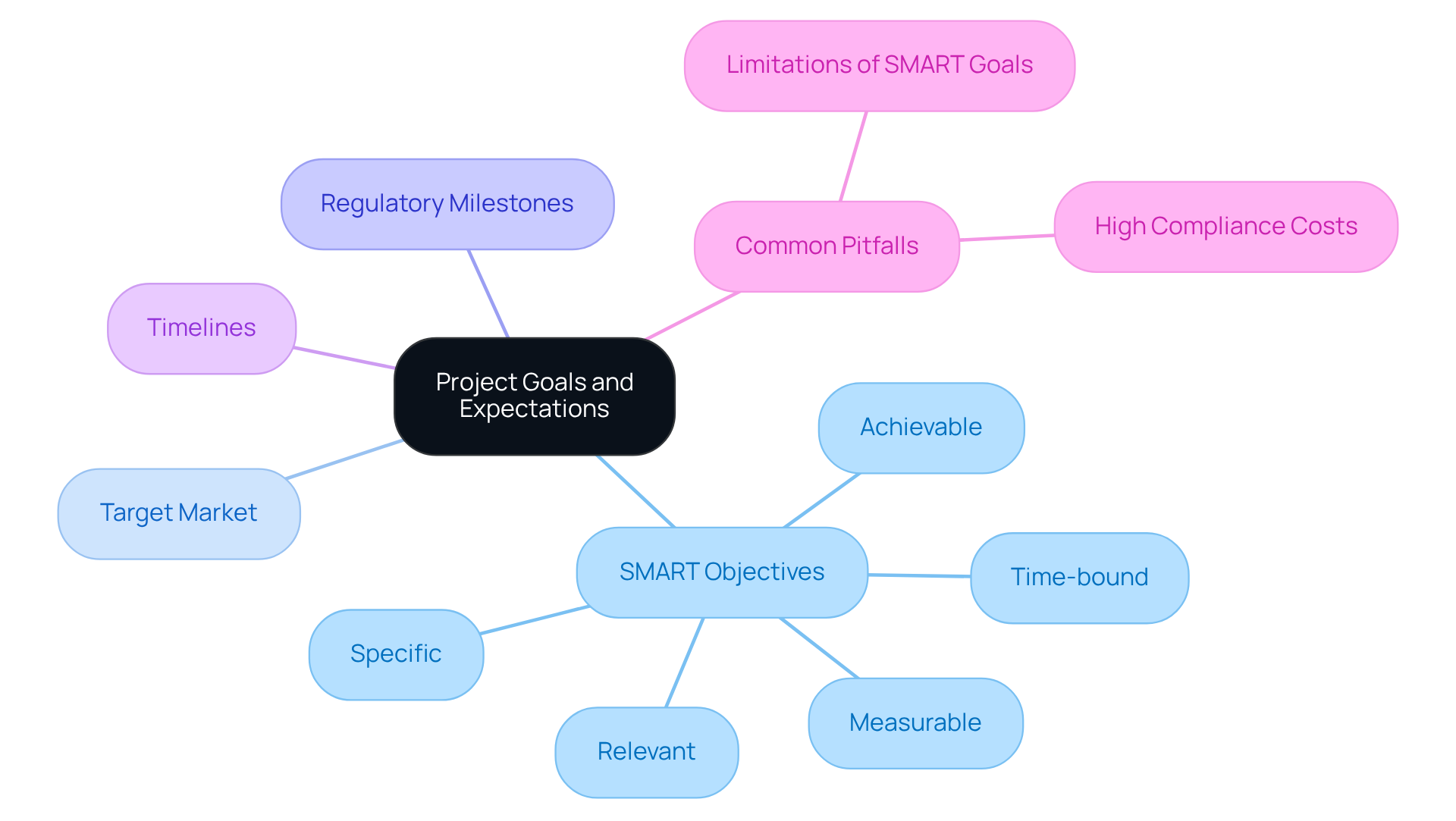
Setting clear objectives and expectations is crucial for effective collaboration with . Start by defining specific, measurable, achievable, relevant, and time-bound (SMART) objectives. For example, if the goal is to , outline the target market, regulatory milestones, and timelines for each phase of development. This clarity not only guides the consultant's efforts but also establishes a . Regularly revisiting these goals throughout the project ensures the team remains focused and aligned, allowing for .
Research indicates that healthcare equipment initiatives with tend to achieve higher success rates, highlighting the importance of . However, it is vital to recognize common pitfalls in goal-setting, such as the limitations of SMART goals, which may hinder creativity and overlook unforeseen opportunities. Additionally, the in healthcare equipment management should be acknowledged when setting objectives, as these factors can greatly impact timelines and resource allocation. By leveraging ' , these challenges can be effectively addressed, facilitating a smoother development process.
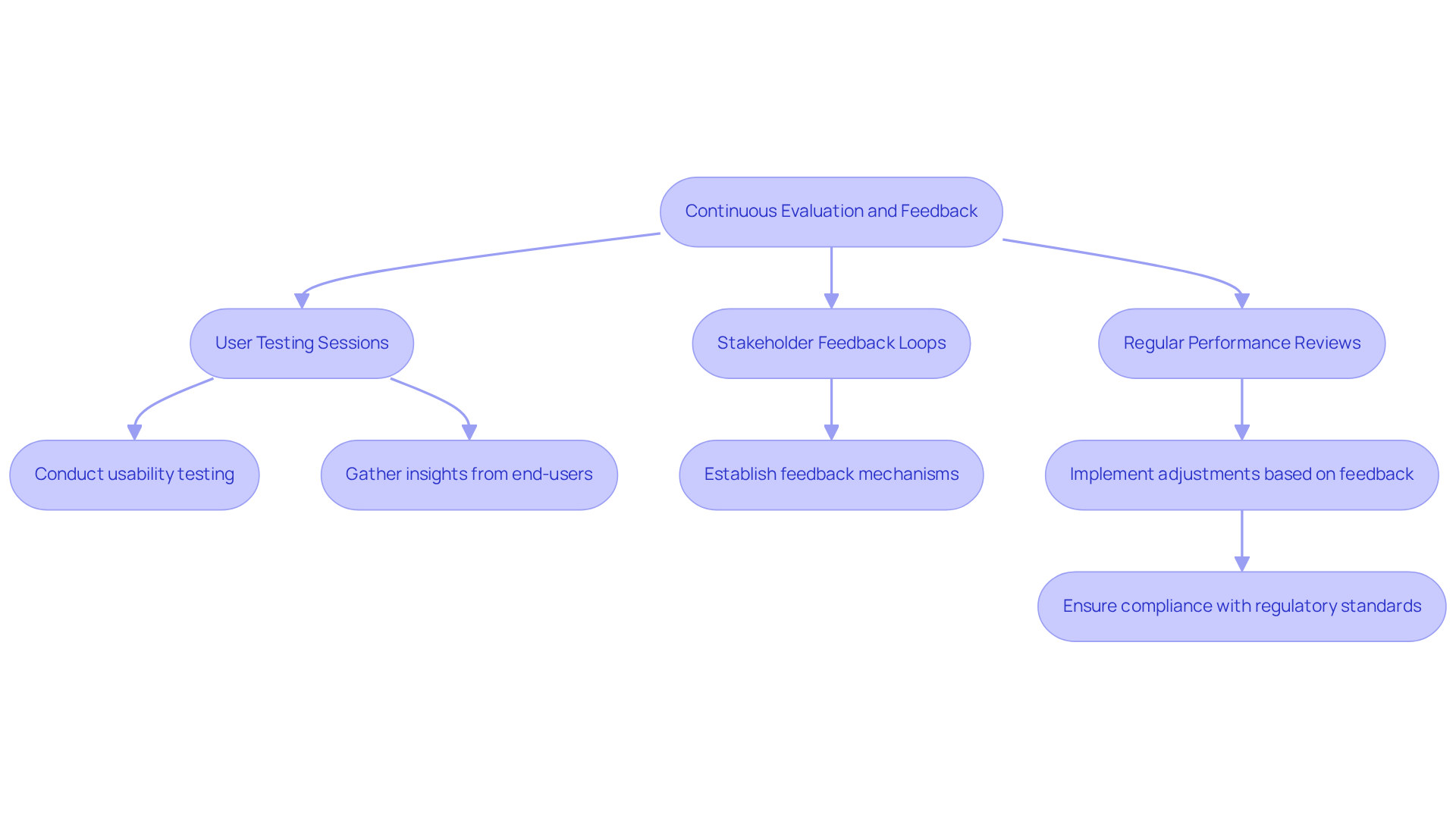
To maximize the effectiveness of medical devices consultants, particularly in , implementing and feedback mechanisms throughout the lifecycle is essential. This approach includes:
For example, conducting with end-users yields valuable insights into product design and functionality, enabling timely adjustments that enhance user experience.
understands how user behavior and environmental factors can impact the efficiency and longevity of , making crucial in their development process. Furthermore, establishing a feedback loop with medical devices consultants facilitates ongoing improvements and ensures that projects remain aligned with regulatory requirements and market needs. The FDA emphasizes that manufacturers must oversee the of their products in the field, reinforcing the necessity of incorporating feedback mechanisms into the creation process.
A significant case study from Bentley's User Experience Center involved approximately 80 simulated surgeries to evaluate a health instrument's design performance, illustrating the impact of user testing on product quality. By fostering a culture of continuous improvement and leveraging AI-driven engineering, which supports precise sensing and seamless data communication, companies like can significantly enhance the quality and reliability of their medical devices. As Jodi Scott, a partner in the industry, notes, early engagement with monitoring strategies can clarify expectations and prevent delays, further underscoring the importance of in product development.
Engaging effectively with medical device consultants is crucial for the successful development and commercialization of healthcare products. Understanding their role and leveraging their expertise in regulatory compliance, design controls, and market strategies allows healthcare companies to significantly enhance their product development processes. This partnership not only improves compliance success rates but also accelerates time to market, positioning these consultants as essential allies in navigating the complexities of the healthcare landscape.
Key practices for optimizing collaboration with medical device consultants include:
By fostering open lines of communication and setting SMART objectives, stakeholders can ensure alignment throughout the project lifecycle. Furthermore, regular feedback loops and performance reviews are vital for making timely adjustments, enhancing product quality, and adhering to regulatory requirements.
Ultimately, collaboration with medical device consultants should be viewed as a strategic investment in innovation and compliance. By embracing these best practices, healthcare companies can streamline their development processes and enhance the overall quality and reliability of their medical devices. A commitment to continuous improvement and effective partnership with consultants is a cornerstone for success in the ever-evolving healthcare sector, paving the way for safer and more effective healthcare solutions.
What is the role of medical device consultants?
Medical device consultants, such as those at Voler Systems, provide specialized expertise in regulatory compliance, design controls, and market strategies to aid in the development and commercialization of healthcare products.
How do medical device consultants assist with regulatory compliance?
They have a deep understanding of healthcare equipment regulations and help prepare documentation for FDA submissions, ensuring that products meet safety and efficacy standards.
What is the FDA Pre-Submission Program?
The FDA Pre-Submission Program allows for early engagement with the agency, which can streamline the submission process and potentially shorten the typical 90-day wait for feedback associated with traditional 510(k) submissions.
How can medical device consultants improve product development efficiency?
By leveraging their expertise, consultants can enhance compliance success rates and reduce time to market, making them invaluable partners in the healthcare equipment sector.
Why is a Quality Management System (QMS) important in healthcare product marketing?
A robust QMS is essential for ensuring adherence to regulatory standards, which is a critical aspect of the consultant's role in supporting healthcare product marketers and manufacturers.
What approach does Voler Systems promote for hardware development?
Voler Systems promotes a prototype approach that simplifies hardware development and design modifications, ensuring compliance is maintained from prototype to production in wearable healthcare products.
Can you provide an example of Voler Systems' consulting effectiveness?
Voler Systems successfully guided a startup through the FDA submission process, demonstrating the practical effectiveness of their consulting services.
