5 Best Practices for Effective Product Development Systems in Medical Devices
Introduction In the rapidly evolving field of medical devices, the stakes are higher than...

Navigating the complex realm of medical device development requires not only technical expertise but also a thorough understanding of regulatory standards and best practices. Engineers and developers must implement effective strategies to ensure compliance while simultaneously enhancing product quality and efficiency. However, with the rapid evolution of technology and regulatory landscapes, how can teams effectively balance innovation with the stringent demands of safety and compliance? This article explores four essential best practices in embedded systems engineering that can empower professionals to confront these challenges directly, fostering collaboration and advancement in medical technology.
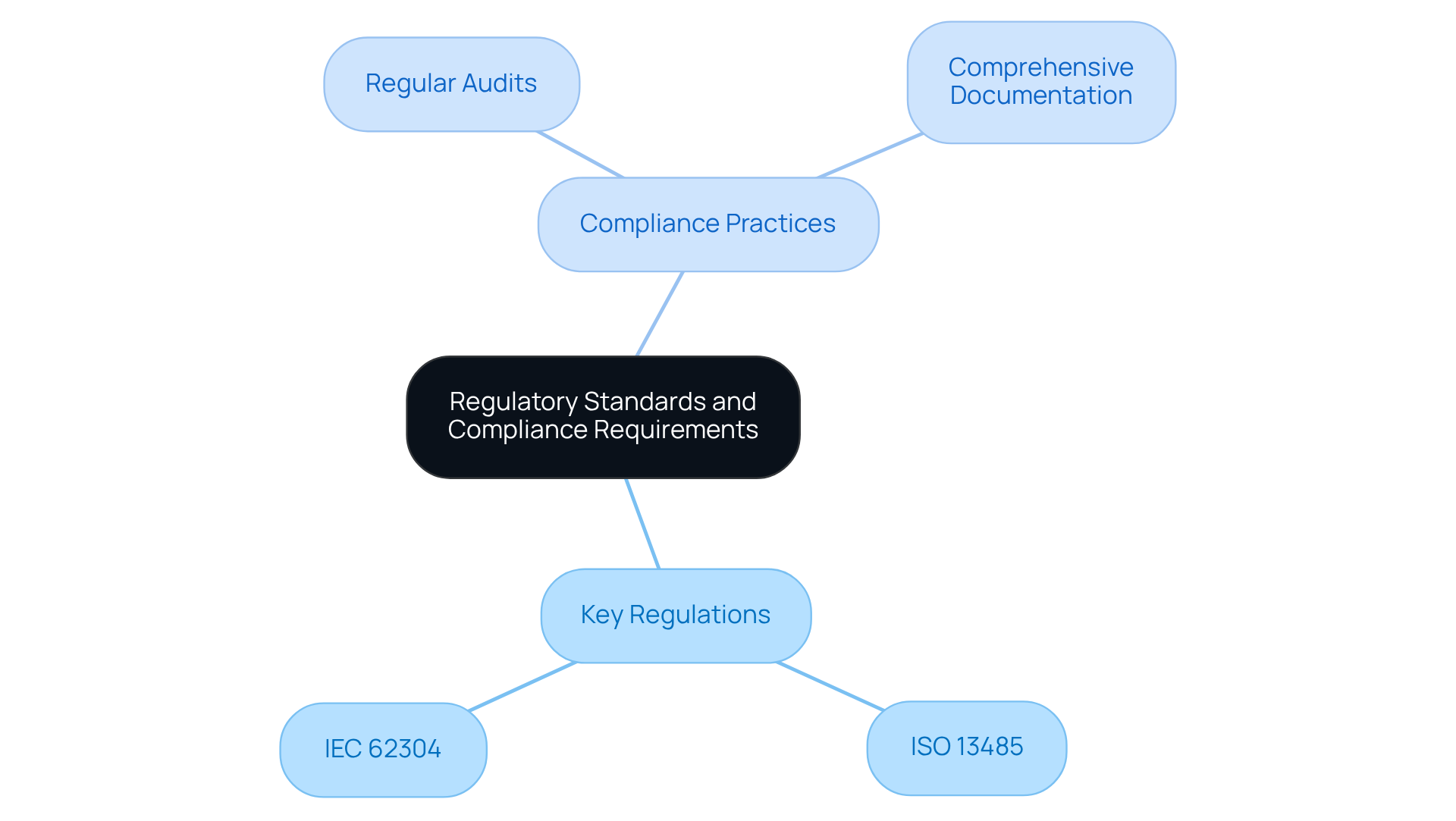
To navigate the complex landscape of healthcare equipment development effectively, engineers must have a thorough understanding of and compliance criteria. Among the key regulations are:
Familiarity with these standards is crucial for ensuring that equipment is safe, effective, and compliant with international regulations. Additionally, integrating early in the development process can mitigate potential compliance challenges. For example:
These practices can facilitate the approval process and enhance product reliability.
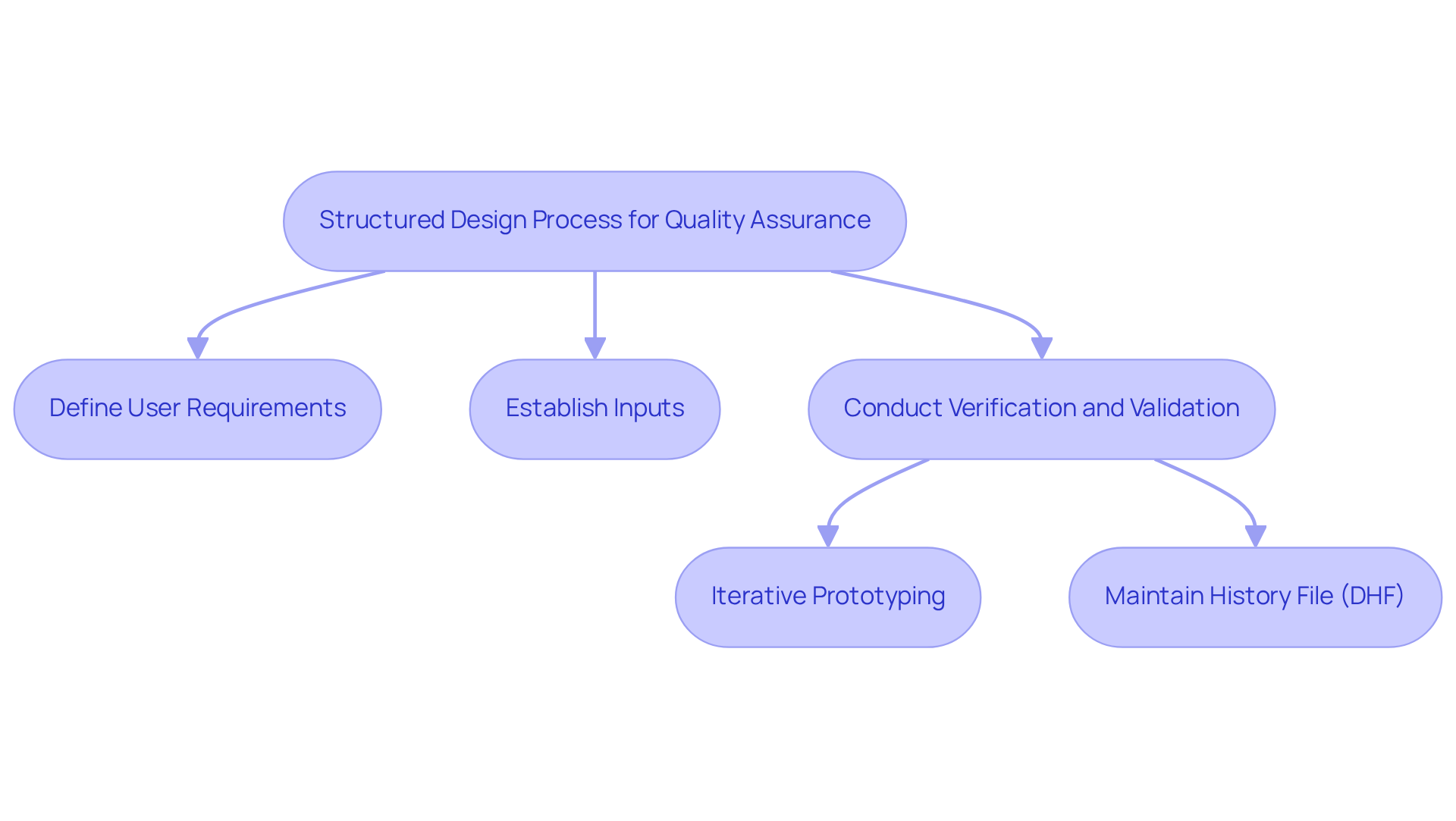
A is crucial for ensuring the . Implementing creates a comprehensive framework for managing the creation and lifecycle of products. This process includes:
For instance, employing by identifying flaws early, thereby minimizing expensive modifications later in the project cycle. Additionally, maintaining a is vital for documenting compliance with regulatory standards, facilitating audits, and ensuring that all development decisions are traceable and thoroughly recorded.
Quality assurance experts emphasize that a well-maintained DHF not only supports but also enhances overall product quality by providing a clear record of the development process and the decisions made throughout.
offers extensive documentation compliance support, including user guides and case studies, assisting startups in navigating the complex regulatory landscape of health technology development. It is also essential to recognize that quality should be a shared responsibility across the entire organization.
Furthermore, early requirement definition, strong traceability, structured review processes, and ongoing risk management are critical components that enhance control over the development process.
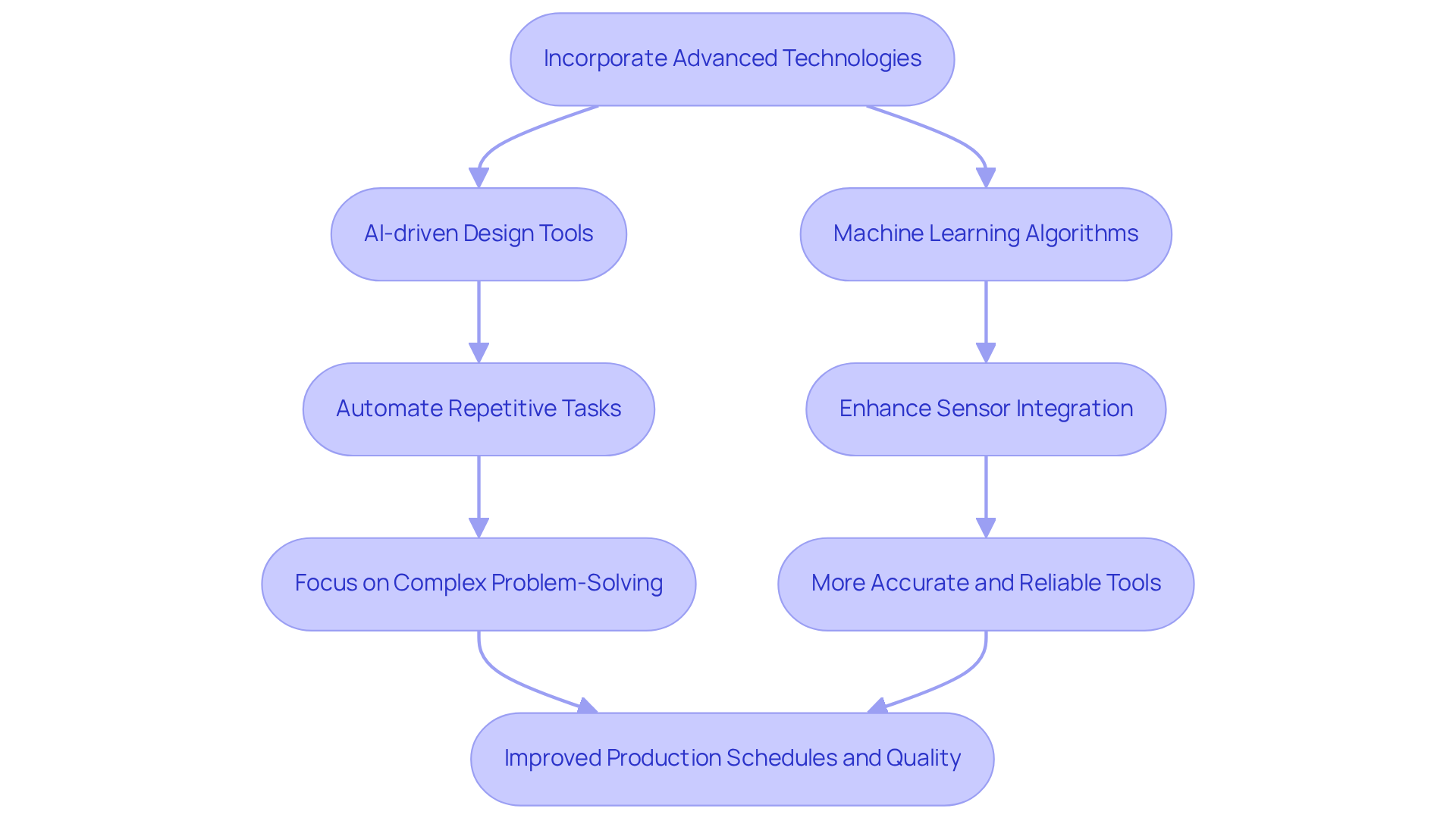
Incorporating significantly enhances efficiency and effectiveness in . At , AI-driven design tools automate repetitive tasks, enabling engineers to concentrate on . For example, our under various conditions, which reduces the need for extensive physical prototyping. This approach not only but also .
Furthermore, , resulting in tools that are both more accurate and reliable. Statistics show that for their revenue cycles, reflecting a broader trend towards automation in the sector.
As industry leaders emphasize, adopting these technologies is crucial for maintaining competitiveness and meeting the stringent demands of regulatory compliance and product safety.
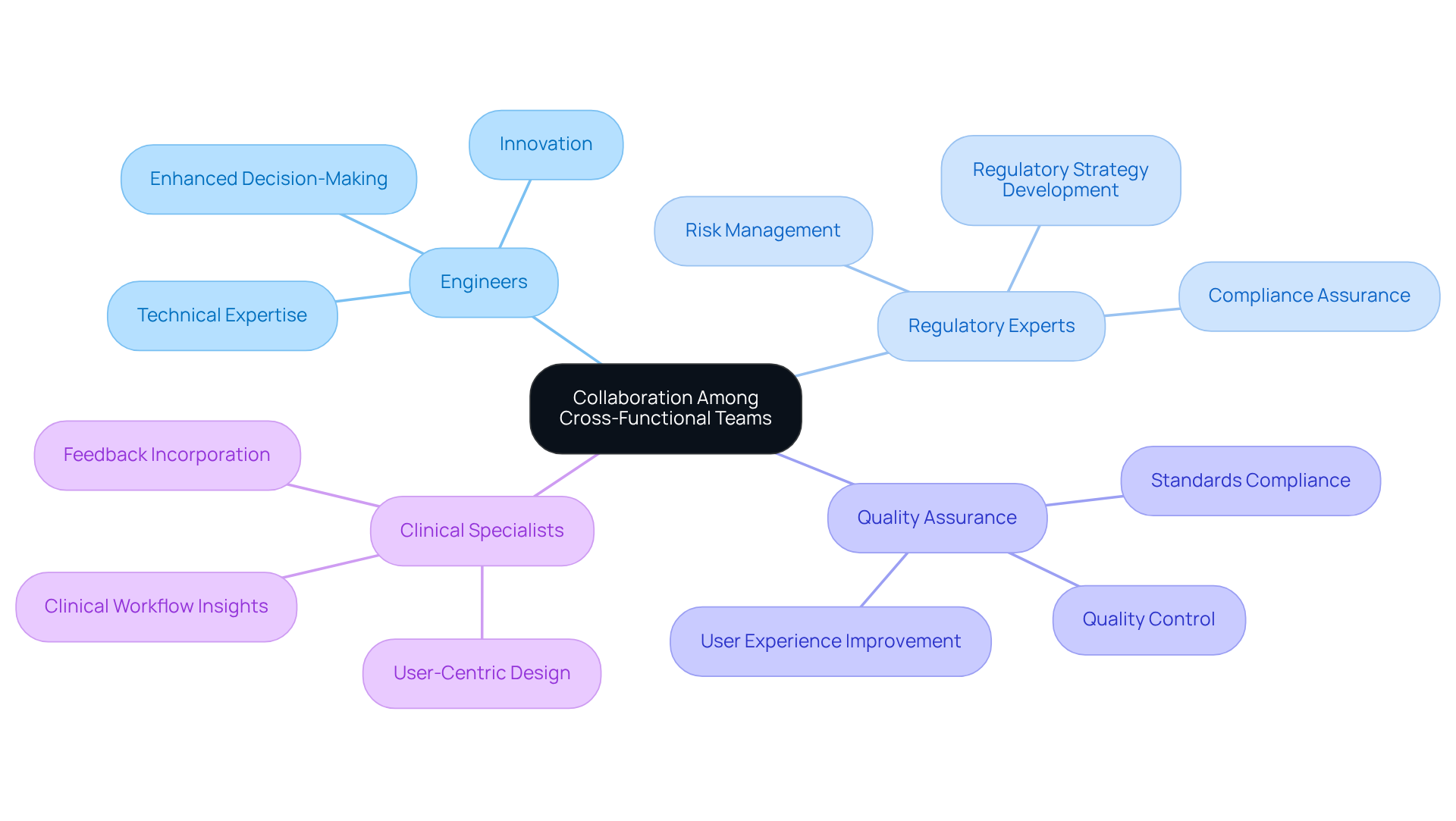
Cooperation among diverse teams is essential in the healthcare product creation process. By integrating engineers, regulatory experts, quality assurance professionals, and clinical specialists, organizations can ensure that a variety of perspectives are included in the planning and development phases. This enhances decision-making and problem-solving while fostering innovation and compliance with industry standards, which is a key focus of ' expertise in .
Research shows that organizations that leverage such collaboration experience that are 34% higher than those employing siloed approaches. Regular meetings and open communication channels are critical for maintaining alignment and nurturing a culture of teamwork. For example, a hospital system successfully reduced implementation delays by 40% by establishing dedicated communication channels for project teams. Furthermore, incorporating feedback from clinical specialists during the design phase can lead to significant improvements in and effectiveness.
The success of is highlighted by the effectiveness of including diverse skill sets, which enhances problem-solving and solution development. Establishing clear roles and shared objectives among team members can streamline processes and improve , as evidenced by various successful medical device engineering projects, including those reviewed by to ensure compliance with emissions and ESD standards.
Understanding and implementing best practices in embedded systems engineering is crucial for the successful development of medical devices. By emphasizing regulatory compliance, structured design processes, advanced technologies, and collaboration, organizations can develop safe, effective, and innovative healthcare solutions that meet the stringent demands of the industry.
Key practices include:
Embracing these best practices not only streamlines the development process but also positions organizations to excel in a competitive landscape. As the medical device industry continues to evolve, prioritizing compliance, quality, and teamwork will be vital for delivering effective healthcare solutions that address the needs of patients and healthcare providers alike. Taking proactive steps now to integrate these principles can pave the way for future success in medical device engineering.
What are the key regulatory standards relevant to healthcare equipment development?
The key regulatory standards include ISO 13485, which focuses on quality management systems for medical devices, and IEC 62304, which outlines software lifecycle processes.
Why is familiarity with regulatory standards important for engineers?
Familiarity with regulatory standards is crucial for ensuring that healthcare equipment is safe, effective, and compliant with international regulations.
How can integrating risk management practices benefit the development process?
Integrating risk management practices early in the development process can mitigate potential compliance challenges and enhance product reliability.
What are some recommended practices to facilitate the approval process of healthcare equipment?
Recommended practices include conducting regular audits and maintaining comprehensive documentation.
