Introduction
The rapid evolution of technology has significantly transformed the landscape of medical device development, with 3D prototyping emerging as a pivotal innovation. This approach not only accelerates the creation of functional prototypes but also fosters enhanced collaboration among multidisciplinary teams, ultimately contributing to improved patient care.
However, as the industry embraces these advancements, several challenges persist, including:
- Material limitations
- Regulatory compliance
- Skill gaps
Developers must effectively navigate these hurdles to fully harness the potential of 3D prototypes in medical devices.
Define Rapid Prototyping in Medical Device Development
Quick model creation in healthcare device development involves the rapid production of a physical representation of a healthcare device using computer-aided drafting (CAD) data, primarily through 3D printing technologies. This approach facilitates the swift creation of 3D prototypes that can be tested and refined, significantly accelerating the development process. In the healthcare sector, where accuracy and compliance with regulatory standards are paramount, rapid model development allows teams to produce functional representations that closely resemble the final product. This capability enables early-stage testing and validation, ensuring that concepts meet both clinical and practical requirements.
Voler Systems leverages its expertise in electronic design services to enhance the rapid model development process, particularly for wearable and IoT health products. Their AI-assisted engineering ensures that prototypes are not only functional but also optimized for future intelligent healthcare applications. The integration of 3D prototypes into development processes has proven to be transformative. For example, companies like Medtronic have successfully utilized in-house 3D prototypes to convert surgeon ideas into functional models within a single day, thereby enhancing collaboration and feedback loops. The 3D printed medical devices market is projected to grow from USD 3.41 billion in 2022 to USD 17.76 billion by 2032, driven by the increasing demand for customized solutions tailored to individual patient needs.
Furthermore, rapid modeling facilitates the early identification of errors in the layout, which can prevent costly alterations later in the production process. This iterative approach not only optimizes resources but also fosters innovation, as teams can concurrently test various concepts, gathering real-world insights from clinicians and patients. Moreover, adherence to regulatory standards is critical during the development process, ensuring that all designs conform to necessary guidelines. Consequently, the advancement of healthcare tools becomes more efficient, ultimately leading to improved patient care and outcomes.
Outline Key Steps in the Rapid Prototyping Process
The rapid prototyping process encompasses several key steps that are vital for successful medical device development, particularly in addressing regulatory challenges and ensuring compliance:
- Concept Development: The process begins with brainstorming and sketching ideas that align with user needs and regulatory requirements. This phase is crucial for ensuring that the design meets clinical goals and addresses practical healthcare issues, which is essential for compliance in the medical equipment sector.
- Design Creation: Employ CAD software to develop detailed 3D models of the product. It is imperative to incorporate all necessary features while adhering to applicable industry standards, as this significantly impacts the product's market readiness and regulatory approval.
- Prototype Fabrication: Select the appropriate 3D printing technology, such as Fused Deposition Modeling (FDM) or Stereolithography (SLA), based on the specific material properties required for 3D prototypes. This choice is critical, as it influences the prototype's performance and usability, aligning with established engineering practices.
- Testing and Evaluation: Execute comprehensive functional tests to assess the prototype's performance. Collecting feedback from stakeholders, including clinicians and engineers, is essential for pinpointing areas needing improvement, ensuring that the device meets user expectations and safety standards, which are crucial for regulatory compliance.
- Iteration: Refine the concept based on testing results and stakeholder feedback. This iterative process may necessitate repeating the design steps multiple times to achieve optimal outcomes, which is vital for minimizing costly errors in later production stages and ensuring a successful transition to manufacturing.
- Finalization: Once the prototype meets all requirements, prepare for regulatory submission and production planning. This step is essential for ensuring that the apparatus can be produced efficiently and complies with all necessary standards, demonstrating Voler Systems' commitment to innovating healthcare instrument design through advanced engineering solutions and providing comprehensive documentation compliance assistance.
By adhering to these structured steps, healthcare product developers can enhance their modeling efforts, resulting in reduced time-to-market and improved product quality.
Highlight Benefits of Rapid Prototyping for Medical Devices
The benefits of rapid prototyping in medical device development are substantial:
- Accelerated Time-to-Market: Rapid model creation significantly shortens the development and testing phases, enabling companies to introduce new products to the market more swiftly. This is particularly vital in healthcare, where timely access to innovations can greatly influence patient care. For example, 3D printing enables the rapid creation of 3D prototypes for customized cranial implants based on CT or MRI scans, ensuring an exact fit and reducing complications. Voler Systems has demonstrated this capability by assisting medical device firms in leveraging rapid development to enhance their product offerings.
- Cost Efficiency: By identifying design flaws early, teams can avert costly modifications later in the development cycle. Rapid model development minimizes material waste and eliminates the need for expensive tooling, making it a financially prudent choice for manufacturers. Traditional modeling methods often result in prolonged production times and elevated costs due to expensive tooling, machining, and molding. Voler Systems' expertise in supporting legacy test equipment further contributes to cost optimization throughout the development process.
- Enhanced Design Flexibility: The iterative nature of rapid prototyping allows for swift adjustments based on testing feedback. This flexibility enables teams to explore various development options without incurring significant delays, ultimately leading to improved product outcomes. Innovations in 3D prototypes, including AI-driven algorithms that optimize structures for strength and weight, enhance this flexibility. For instance, Voler Systems has been at the forefront of these innovations, particularly in the creation of wearable medical instruments that continuously monitor vital signs.
- Improved Collaboration: Prototyping fosters collaboration among diverse teams, including engineers, designers, and healthcare professionals. This multidisciplinary approach ensures that all perspectives are integrated into the design process, thereby enhancing the overall quality of the final product. Industry leaders emphasize that incorporating model testing throughout the development lifecycle can expedite the market readiness of innovative products. Voler Systems exemplifies this collaborative spirit by closely partnering with clients to ensure compliance with industry standards.
- Regulatory Compliance: Early-phase modeling facilitates comprehensive testing and validation, ensuring that products meet regulatory standards prior to submission. This proactive approach mitigates the likelihood of delays in the approval process, streamlining the path to market. By employing rapid modeling, manufacturers can efficiently test and validate multiple designs simultaneously, gathering real-world feedback before full-scale production. Voler Systems' commitment to managing compliance in wearable health technology development underscores the importance of this practice.
Identify Challenges in Rapid Prototyping for Medical Devices
Despite its advantages, rapid prototyping in medical device development presents several challenges:
- Material limitations exist as not all materials used in creating 3D prototypes are suitable for medical applications. Selecting the appropriate material that complies with regulatory standards can be complex, requiring a thorough evaluation of the attributes that contribute to successful engineering projects.
- Structure Complexity: Highly intricate designs may necessitate advanced printing methods and extended production times, complicating the modeling process. This complexity can impede the critical steps essential for effective project execution.
- Regulatory Compliance: Navigating the regulatory landscape poses challenges, as prototypes must adhere to stringent safety and efficacy standards before receiving approval for clinical use. A comprehensive understanding of these regulations is vital for ensuring a successful transition to manufacturing.
- The initial investment in high-quality 3D prototypes can be significant, creating a barrier for smaller companies or startups. This financial consideration is a crucial aspect of planning for successful engineering development projects.
- Skill Gaps: There may be a shortage of skilled personnel proficient in both design and 3D printing technologies, which can obstruct the development process. Addressing these skill gaps is essential for improving project outcomes and fostering innovation in healthcare technology.
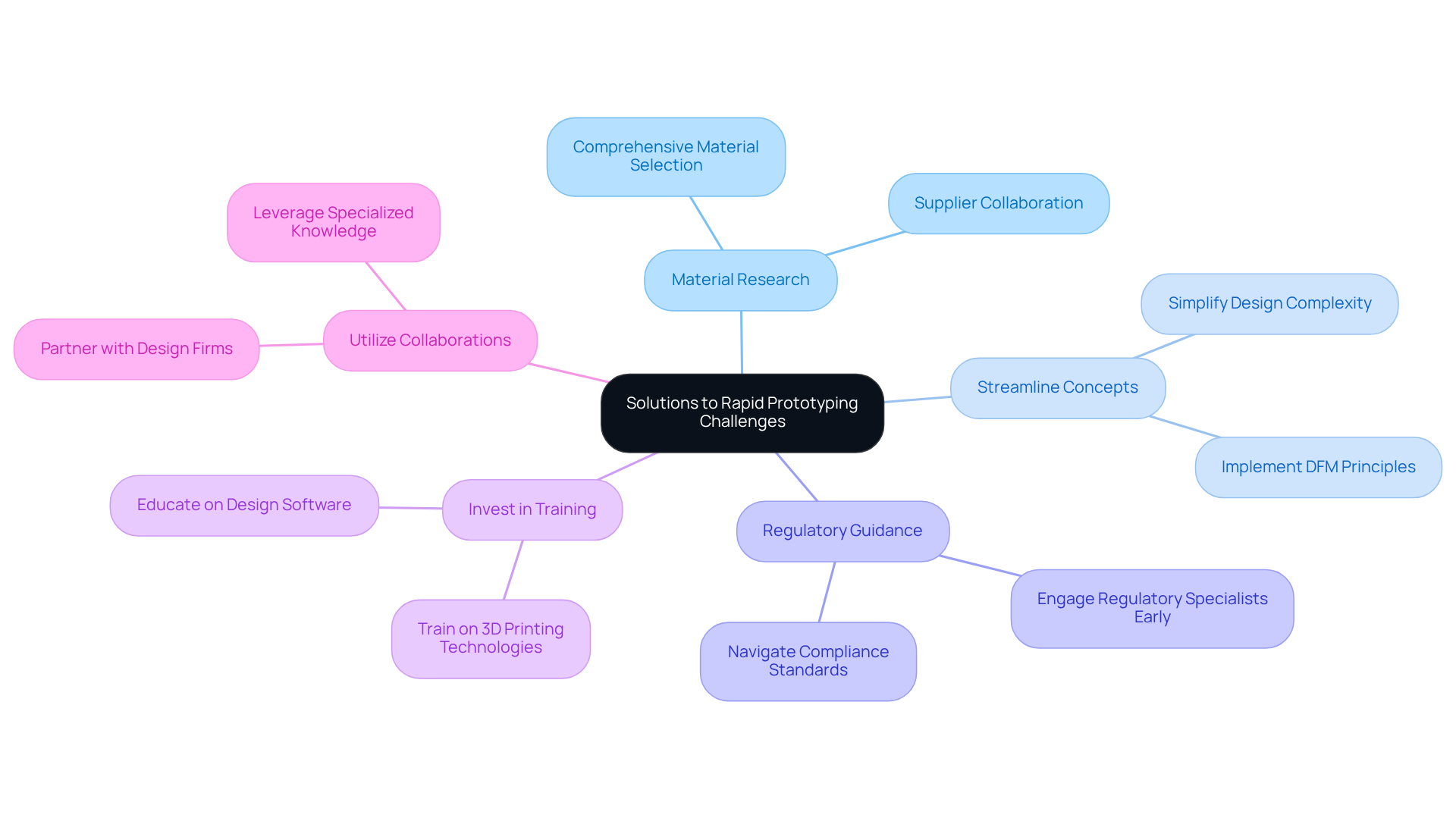
Propose Solutions to Overcome Rapid Prototyping Challenges
To effectively address the challenges associated with rapid prototyping in medical device development, consider the following strategies:
- Material Research: Prioritize comprehensive research and selection of materials that comply with medical standards. Collaborate closely with suppliers to identify the most suitable options tailored to specific applications, ensuring both safety and functionality.
- Streamline Concepts: Aim to simplify concepts wherever possible to reduce complexity, facilitating easier modeling. Implement design for manufacturability (DFM) principles to enhance production efficiency and minimize potential issues during the prototyping phase.
- Regulatory Guidance: Engage regulatory specialists early in the development process to effectively navigate compliance with relevant standards. This proactive approach can significantly streamline the approval process, thereby reducing time-to-market for new devices.
- Invest in training by providing thorough education for team members on the latest design software and 3D printing technologies to create 3D prototypes. This investment addresses skill gaps and enhances overall development capabilities, leading to more successful outcomes.
- Utilize Collaborations: Collaborate with experienced design firms or advisors who can offer specialized knowledge and resources. Such partnerships can help mitigate challenges and accelerate the prototyping process, ensuring that projects remain on track and within budget.
Conclusion
The integration of 3D prototypes in medical device development marks a significant advancement that enhances both innovation and efficiency within the healthcare sector. By utilizing rapid prototyping, teams can quickly create, test, and refine models, ensuring compliance with the stringent requirements of clinical practice and regulatory standards. This method not only accelerates the development timeline but also markedly improves the quality of medical devices, ultimately benefiting patient care.
This article highlights key practices for effective rapid prototyping, including:
- The critical aspects of material selection
- The iterative design process
- The necessity for collaboration among multidisciplinary teams
Each phase, from concept development to finalization, is vital in ensuring that prototypes are functional and adhere to industry standards. Additionally, the discussion of challenges and solutions underscores the importance of strategic planning and investment in technology and talent to navigate barriers in this evolving field.
As the demand for customized medical solutions continues to grow, adopting these best practices in rapid prototyping will be essential for manufacturers striving to maintain competitiveness. The potential for enhanced patient outcomes and improved operational efficiency emphasizes the importance of innovative approaches in medical device development. Stakeholders in the healthcare industry are urged to prioritize these strategies, ensuring they not only address current needs but also lay the groundwork for future advancements in medical technology.
Frequently Asked Questions
What is rapid prototyping in medical device development?
Rapid prototyping in medical device development involves the quick creation of a physical model of a healthcare device using CAD data, primarily through 3D printing. This method allows for the swift production of prototypes that can be tested and refined, accelerating the overall development process.
How does rapid prototyping benefit the healthcare sector?
Rapid prototyping facilitates early-stage testing and validation of healthcare devices, ensuring they meet clinical and practical requirements. It allows teams to produce functional representations that closely resemble the final product, which is crucial for accuracy and compliance with regulatory standards.
What role does Voler Systems play in rapid prototyping?
Voler Systems enhances the rapid model development process by leveraging its expertise in electronic design services, particularly for wearable and IoT health products. Their AI-assisted engineering ensures that prototypes are functional and optimized for future intelligent healthcare applications.
What are the projected market trends for 3D printed medical devices?
The 3D printed medical devices market is projected to grow from USD 3.41 billion in 2022 to USD 17.76 billion by 2032, driven by the increasing demand for customized solutions tailored to individual patient needs.
What are the key steps in the rapid prototyping process?
The key steps in the rapid prototyping process include: 1. Concept Development: Brainstorming and sketching ideas that meet user needs and regulatory requirements. 2. Design Creation: Using CAD software to develop detailed 3D models that adhere to industry standards. 3. Prototype Fabrication: Selecting appropriate 3D printing technology based on required material properties. 4. Testing and Evaluation: Conducting functional tests and collecting feedback from stakeholders. 5. Iteration: Refining the concept based on testing results and feedback, repeating design steps as necessary. 6. Finalization: Preparing for regulatory submission and production planning once all requirements are met.
Why is iteration important in the rapid prototyping process?
Iteration is crucial because it allows for refinement based on testing results and stakeholder feedback, helping to minimize costly errors in later production stages and ensuring a successful transition to manufacturing.
How does adherence to regulatory standards impact the rapid prototyping process?
Adherence to regulatory standards is essential during the rapid prototyping process, as it ensures that all designs conform to necessary guidelines, ultimately leading to more efficient advancement of healthcare tools and improved patient care and outcomes.
List of Sources
- Define Rapid Prototyping in Medical Device Development
- 3D Printed Medical Devices Market Set to Triple by 2032 (https://mymediset.net/blog/the-rise-of-3d-printing-in-medical-devices-revolutionizing-healthcare)
- The Role of Rapid Prototyping in Fast-Tracking New Medical Technologies (https://gcmiatl.org/the-role-of-rapid-prototyping-in-fast-tracking-new-medical-technologies)
- Rapid Prototyping in the Medical Industry: Reducing Time-To-Market with 3D Printing - Medtec China (https://en.medtecchina.com/media/manufacturing/rapid-prototyping-in-the-medical-industry-reducing-time-to-market-with-3d-printing)
- 3D Printing in Healthcare Market to Hit USD 11.22 Billion by 2035, Advancing Personalized Medical Manufacturing – SNS Insider (https://globenewswire.com/news-release/2026/02/13/3238068/0/en/3D-Printing-in-Healthcare-Market-to-Hit-USD-11-22-Billion-by-2035-Advancing-Personalized-Medical-Manufacturing-SNS-Insider.html)
- Global Rapid Prototyping Medical Devices Market Size, Industry Share, Growth Trends & Forecast 2026-2034 (https://verifiedmarketreports.com/product/rapid-prototyping-medical-devices-market)
- Outline Key Steps in the Rapid Prototyping Process
- Rapid Prototyping in the Medical Industry: Reducing Time-To-Market with 3D Printing - Medtec China (https://en.medtecchina.com/media/manufacturing/rapid-prototyping-in-the-medical-industry-reducing-time-to-market-with-3d-printing)
- The Role of Rapid Prototyping in Fast-Tracking New Medical Technologies (https://gcmiatl.org/the-role-of-rapid-prototyping-in-fast-tracking-new-medical-technologies)
- photofabrication.com (https://photofabrication.com/the-role-of-rapid-prototyping-in-medical-device-development)
- What is Rapid Prototyping: Process, Stages, Types and Tools - TechniWaterjet (https://techniwaterjet.com/what-is-rapid-prototyping-process-stages-types-and-tools)
- From Concept to Market: Rapid Prototyping for Medical Devices (https://intech-ind.com/post/from-concept-to-market-rapid-prototyping-for-medical-devices)
- Highlight Benefits of Rapid Prototyping for Medical Devices
- paragonmedical.com (https://paragonmedical.com/blog/saving-development-time-and-costs-with-rapid-prototyping)
- Rapid Prototyping in the Medical Industry: Reducing Time-To-Market with 3D Printing - Medtec China (https://en.medtecchina.com/media/manufacturing/rapid-prototyping-in-the-medical-industry-reducing-time-to-market-with-3d-printing)
- Medical Device Prototyping In Accelerating Time To Market (https://europlaz.co.uk/medical-device-prototyping-in-accelerating-time-to-market)
- Medical Device Manufacturers Accelerate R&D and Speed to Market via Live Prototyping (https://elevarismedical.com/news-media-1/medical-device-manufacturers-accelerate-rampd-and-speed-to-market-via-live-prototyping)
- Orthopedic Implants Manufacturing Expert|YSF Medical (https://ysfbone.com/article_d.php?lang=en&tb=8&id=610)
- Propose Solutions to Overcome Rapid Prototyping Challenges
- mpo-mag.com (https://mpo-mag.com/exclusives/rapid-prototyping-in-the-medical-industry-reducing-time-to-market-with-3d-printing)
- Rapid Prototyping for Medical Devices: 5 Ways to Reduce Cost and Time (https://lsrpf.com/blog/rapid-prototyping-for-medical-devices-5-ways-to-reduce-cost-and-time-to-market)
- The Evolving Role of Additive Manufacturing Services for Medical Devices (https://rapid3devent.com/event/news/the-evolving-role-of-additive-manufacturing-services-for-medical-devices)
- Overcoming Challenges in Medical Prototype Development - Kritikal Solutions (https://kritikalsolutions.com/medical-prototype-development)
- Overcoming Medical Device Manufacturing Regulatory Challenges (https://tessy.com/news-events/navigating-regulatory-hurdles-in-medical-device-manufacturing)






