Understanding Prototype Services in Medical Device Development
Introduction In the rapidly evolving landscape of medical technology, prototype services...

Prototyping is a fundamental aspect of medical device development, serving as a crucial link between innovative concepts and actual products. By implementing effective prototyping practices, developers can significantly improve their prospects for regulatory approval and market success, which ultimately contributes to enhanced patient care. Nevertheless, this process presents challenges. Key considerations must be addressed when selecting prototype design services, and teams must ensure that their prototypes not only comply with regulatory standards but also resonate with end-users.
Prototyping serves as a vital phase in the lifecycle, acting as a crucial link between initial concepts and the final product. This process allows designers to visualize and rigorously test their ideas, . Early-stage prototypes are instrumental in identifying potential , enabling teams to implement necessary modifications before progressing to more costly production stages.
For instance, effective can significantly enhance success rates during by identifying issues early, thereby conserving valuable time and resources. Furthermore, the fosters innovation, permitting teams to explore various materials and technologies, which can lead to the creation of more efficient and user-friendly health products, such as and .
By prioritizing , developers can markedly improve the likelihood of and market success, ultimately enhancing patient care and healthcare outcomes.
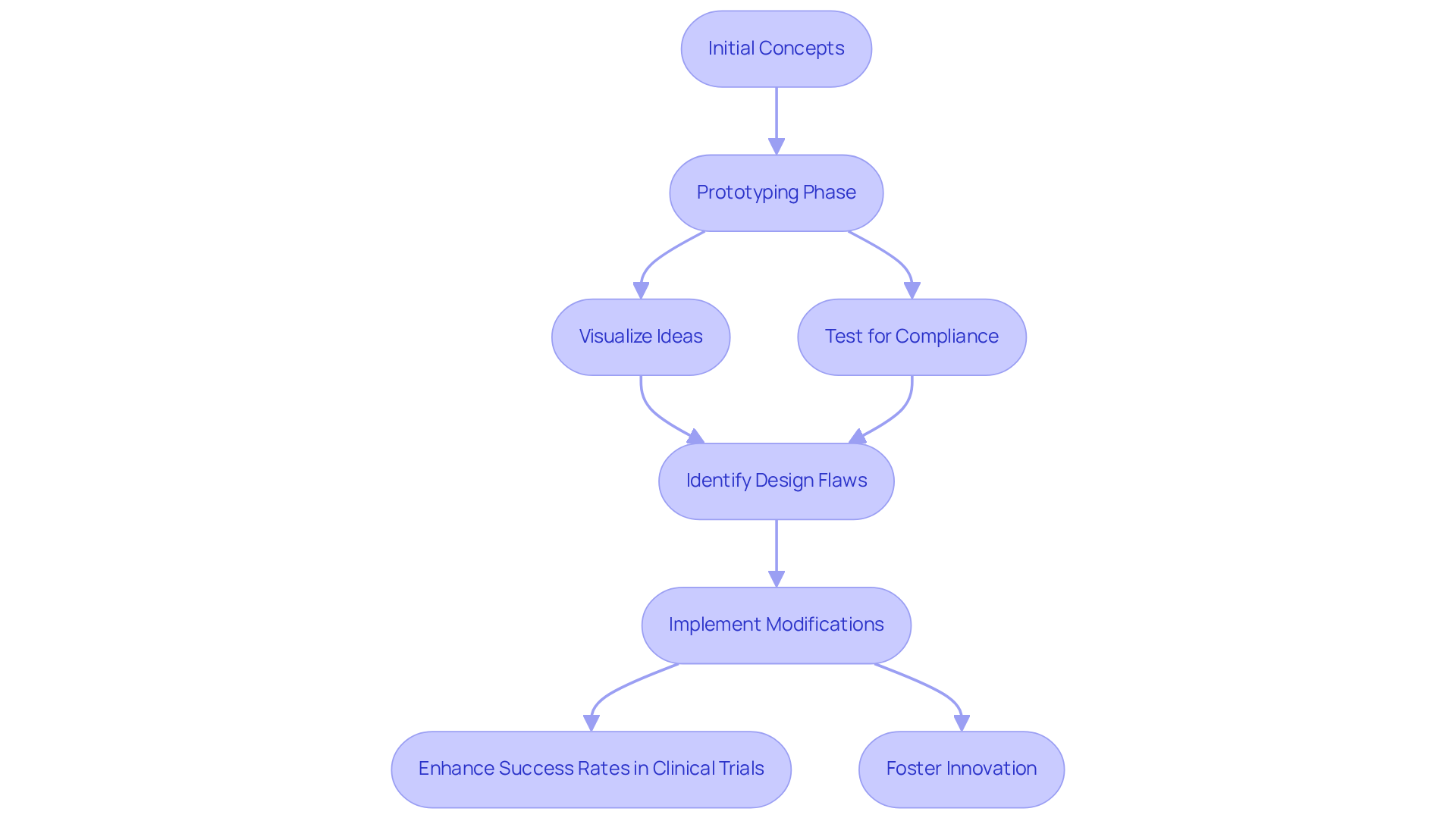
When selecting a , several key criteria should guide your decision-making process:
By carefully evaluating these criteria, you can select prototype design services that not only meet your technical requirements but also align with your strategic objectives in the competitive healthcare landscape.
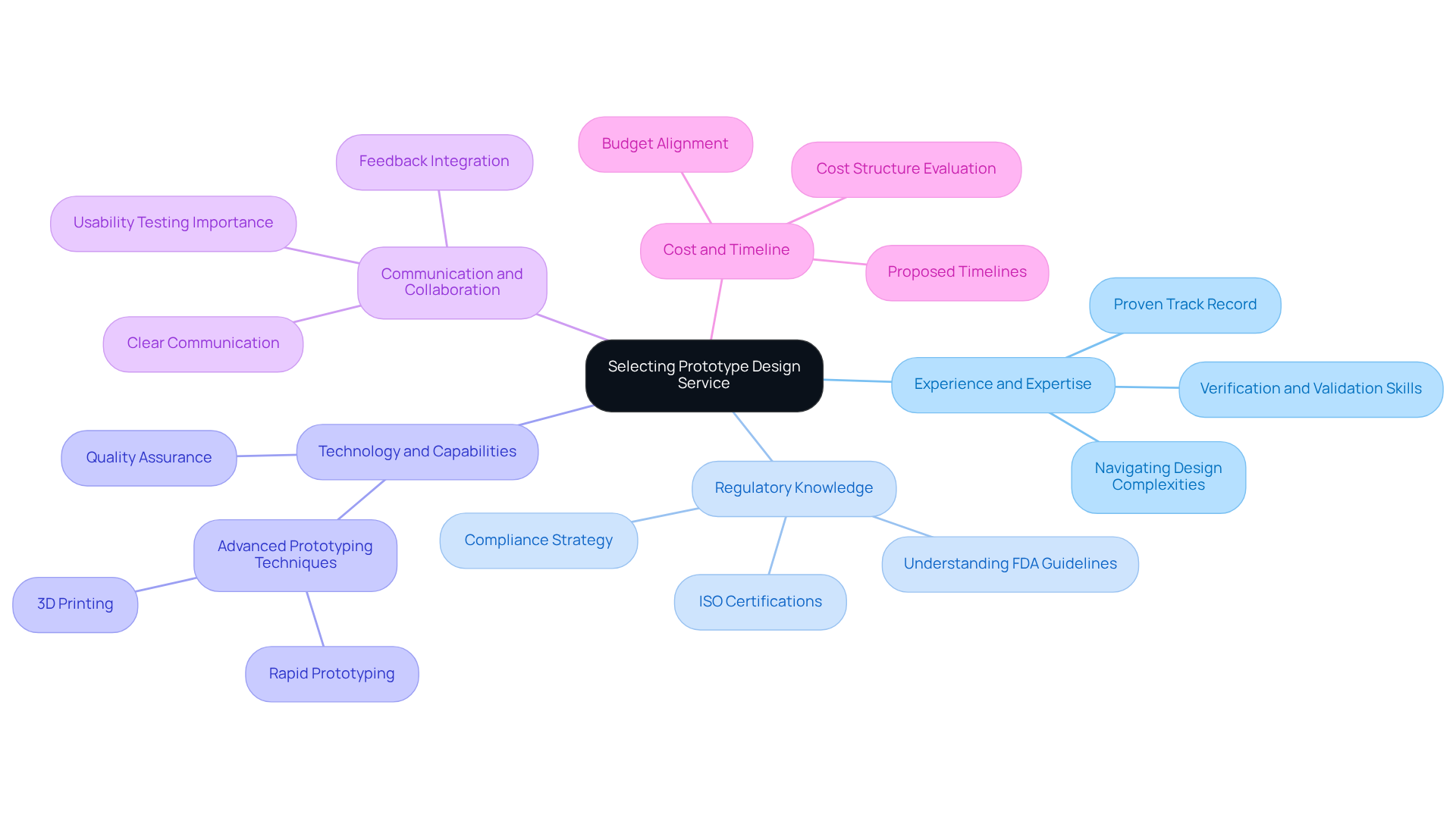
To ensure compliance and quality in , the following techniques should be implemented:
By incorporating these best practices, manufacturers of healthcare equipment can enhance their model development efforts, resulting in safer, more effective products that comply with regulatory standards and meet user requirements.
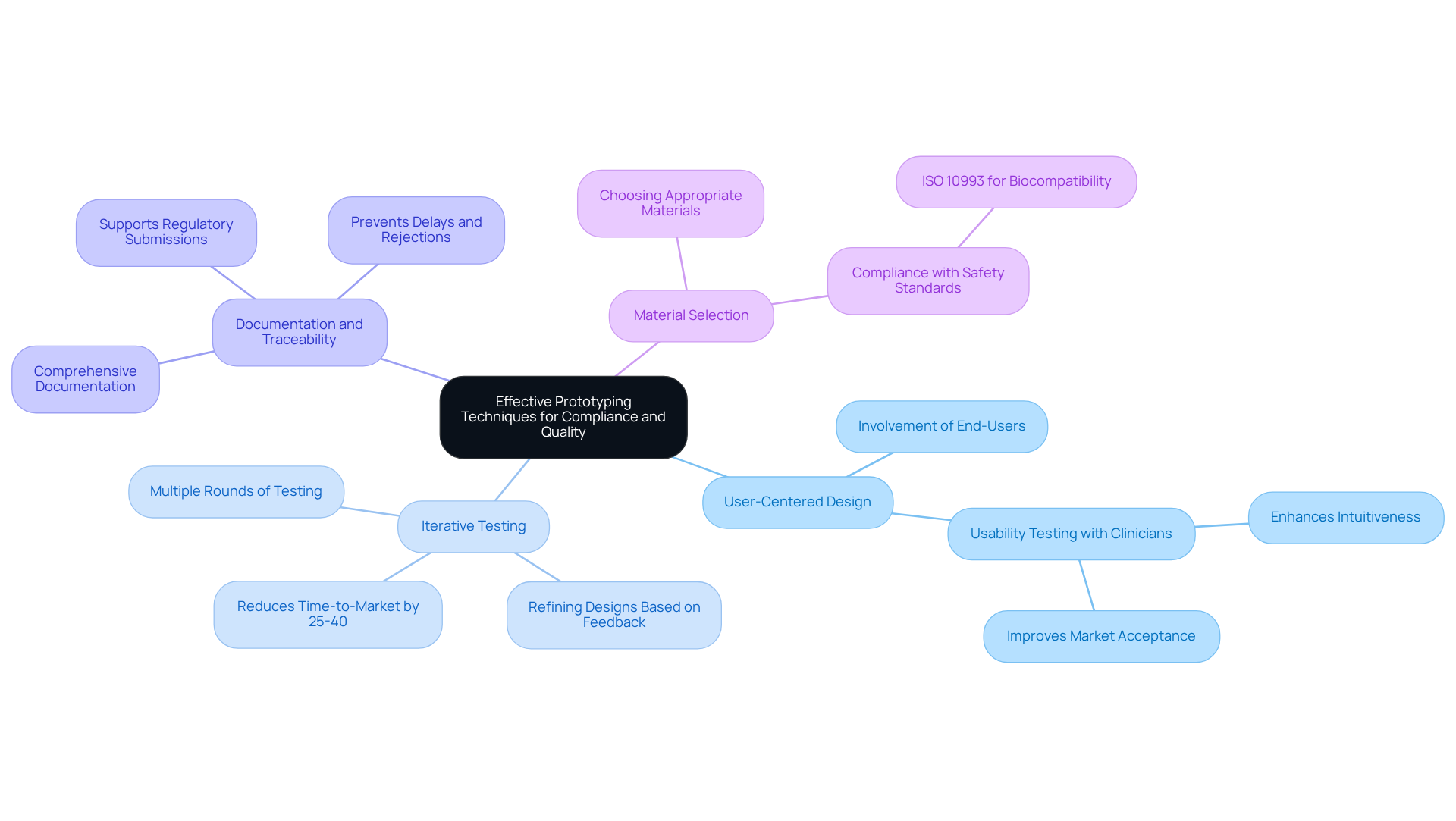
Iterative testing and feedback are critical components of the medical equipment development cycle. To effectively implement this approach, consider the following steps:
This cycle of testing and feedback promotes a culture of , ultimately leading to the development of more successful using .
Prototyping is a critical component in the medical device development process, serving as a vital link that converts initial concepts into viable products. By dedicating time and resources to effective prototype design, developers can significantly improve the quality and compliance of their medical devices, ultimately leading to enhanced patient outcomes and increased market success.
This article highlights key practices such as:
Recognizing the importance of regulatory knowledge, advanced technology, and clear communication further equips teams to navigate the complexities of medical device development with efficiency. By adhering to these best practices, organizations can mitigate risks and improve their likelihood of achieving regulatory approval.
In the rapidly evolving healthcare landscape, the importance of effective prototyping cannot be overstated. It is not merely a step in the development process but a crucial investment in innovation and quality. By embracing these best practices, organizations will not only streamline the development cycle but also cultivate a culture of continuous improvement, ultimately resulting in safer, more effective medical devices that fulfill the needs of patients and healthcare providers alike.
What is the role of prototyping in medical device development?
Prototyping serves as a vital phase in the medical device development lifecycle, acting as a crucial link between initial concepts and the final product. It allows designers to visualize and rigorously test their ideas.
How does prototyping help in identifying design flaws?
Early-stage prototypes are instrumental in identifying potential design flaws, enabling teams to implement necessary modifications before progressing to more costly production stages.
What impact does effective prototyping have on clinical trials?
Effective model creation can significantly enhance success rates during clinical trials by identifying issues early, thereby conserving valuable time and resources.
How does the prototyping stage foster innovation in medical device development?
The development stage fosters innovation by permitting teams to explore various materials and technologies, which can lead to the creation of more efficient and user-friendly health products.
What are some examples of medical devices that benefit from prototyping?
Examples of medical devices that benefit from prototyping include heart pumps and liquid biopsy platforms.
How does prioritizing model creation affect regulatory approval and market success?
By prioritizing model creation, developers can markedly improve the likelihood of regulatory approval and market success, ultimately enhancing patient care and healthcare outcomes.
