5 Best Practices for Using 3D Prototypes in Medical Devices
Discover best practices for optimizing 3D prototypes in medical device development.

Prototyping serves as a cornerstone in medical device development, where precision and safety are paramount. This article explores the detailed process of prototype development, highlighting its critical role in converting innovative concepts into dependable medical instruments. As the industry progresses with rapid technological advancements, manufacturers must consider how to ensure their prototyping practices not only comply with regulatory standards but also promote innovation and efficiency.
The prototype development definition refers to the process of creating an initial model or sample of a product, which is particularly crucial in the medical equipment sector. This early version is essential for testing and validating concepts, functionality, and design prior to full-scale production. Prototyping enables engineers and designers to explore various facets of a product, including usability, safety, and compliance with regulatory standards.
In the medical field, where accuracy and dependability are paramount, modeling plays a critical role in confirming that instruments meet the necessary standards for patient care and safety. By iterating on prototypes, teams can refine their designs based on feedback and testing results, ultimately leading to more effective and safer medical devices.
Rapid modeling significantly accelerates the development cycle, allowing for quicker iterations and faster market entry. This speed is crucial in a competitive environment where time-to-market can dictate success. Furthermore, studies indicate that effective modeling can mitigate the financial risks associated with product development, making it a strategic necessity for manufacturers aiming to innovate while adhering to stringent industry standards.
The advancement of model development in engineering and design has significantly transformed, particularly within the medical device industry. The introduction of rapid prototyping technologies in the 1980s, notably 3D printing, marked a pivotal shift. This innovation enabled faster and more cost-effective production in line with the prototype development definition, allowing designers to iterate quickly and efficiently. For instance, Clubfoot Solutions created 18 variations for a pediatric brace in just a few weeks, showcasing the rapid capabilities of 3D printing.
Moreover, the incorporation of digital tools and software has simplified the model development process, enabling the creation of intricate designs and simulations. Today, the prototype development definition serves not only as a means of testing functionality but also as a critical component in adhering to stringent regulatory standards set by agencies like the FDA. The medical equipment development process comprises eight essential steps, with the prototype development definition playing a crucial role in ensuring safety and efficacy.
This historical perspective highlights the importance of the prototype development definition in fostering innovation and meeting the evolving demands of the healthcare sector. Voler Systems illustrates this evolution with their model strategy, which emphasizes optimizing hardware development and making modifications for medical equipment manufacturing. Their creative strategy encompasses AI-driven engineering, ultra-low power solutions, and recent advancements like NFC-to-WiFi tools and IoT sensors, ensuring that models not only meet compliance standards but also extend the limits of what is achievable in medical applications.
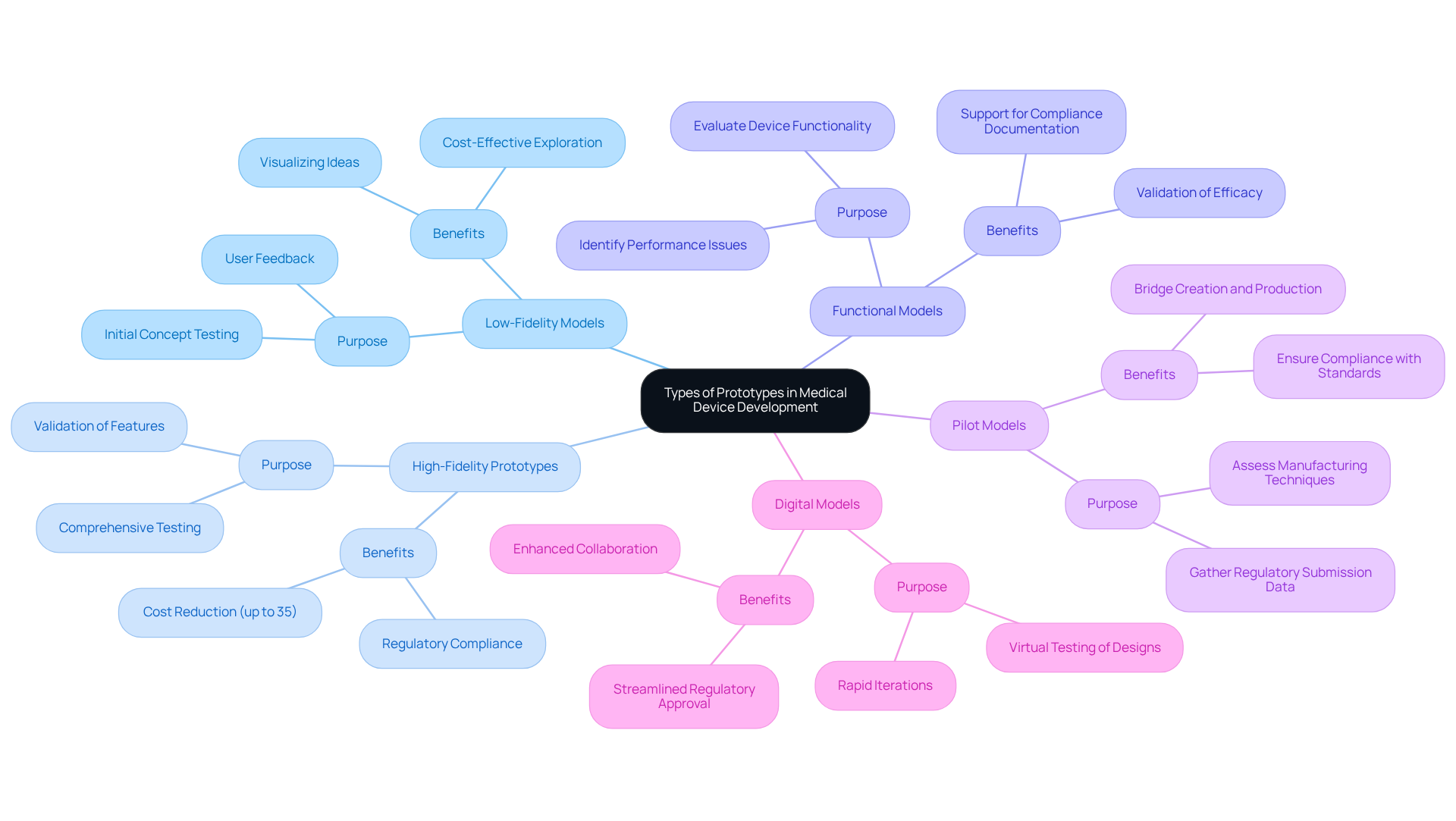
In medical device development, various types of prototypes serve distinct purposes, each contributing to the iterative design process:
Each prototype category plays a critical role in refining products based on real-world testing and feedback, ultimately improving the development process and ensuring that medical equipment is effective and compliant with industry standards. Additionally, the use of specific materials, such as long-term implantable grades of PEEK and polysulfone, can further enhance performance and longevity.
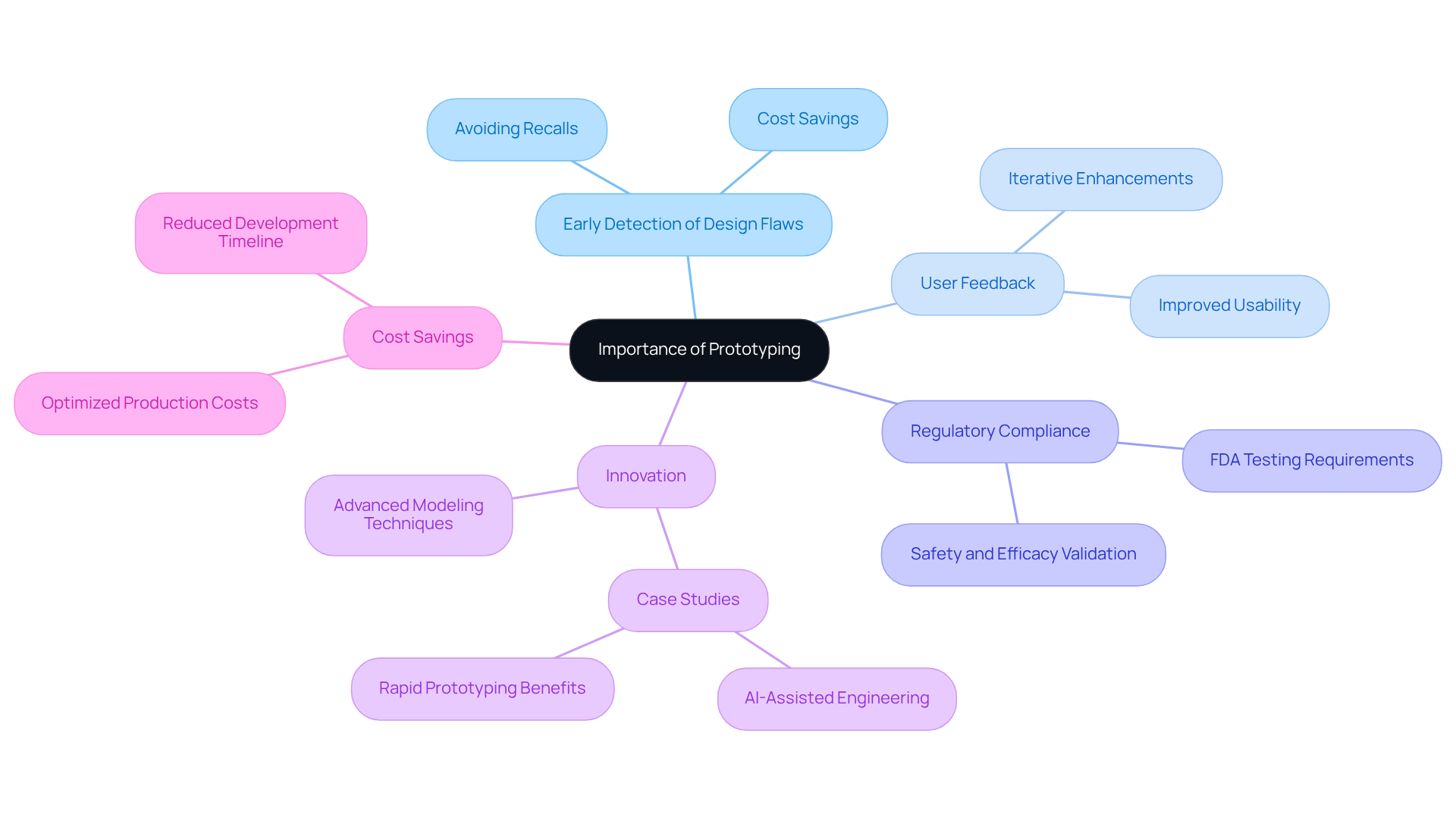
Prototyping is a fundamental aspect of the medical device development process, fulfilling several critical functions. It enables the early detection of design flaws, which can result in significant time and cost savings during later stages. By experimenting with models, teams can gather valuable user feedback, allowing for necessary modifications before large-scale production. This iterative approach not only improves the quality of the final product but also plays a vital role in adhering to regulatory standards. The FDA mandates comprehensive testing to demonstrate safety and efficacy, making model development essential to this validation process. For instance, the lack of prototypes can complicate compliance, as testing and validation are typically required.
Moreover, effective modeling encourages innovation that can enhance patient outcomes, streamline manufacturing processes, and lower overall development costs. Statistics reveal that neglecting the initial development phase can expose manufacturers to considerable risks, including expensive recalls and safety concerns linked to design flaws. Notably, software-related issues have resulted in 627 FDA recalls affecting 1.4 million units over five years, underscoring the critical importance of the prototype development definition.
Additionally, Voler Systems utilizes AI-assisted engineering to refine the model creation process, ensuring that prototypes are both innovative and compliant with regulatory standards. Their compliance review process specifically addresses emissions and ESD standards, further emphasizing the necessity of thorough testing. Case studies from Voler Systems demonstrate how efficient model creation has led to successful product launches while adhering to stringent regulations. In conclusion, the significance of prototyping in medical device development cannot be overstated; it is essential for delivering safe, effective, and market-ready products while ensuring compliance with rigorous regulatory requirements.
Prototyping is a fundamental aspect of medical device development, acting as the essential link between concept and reality. This process not only allows for the testing of ideas but also ensures that products comply with the rigorous safety and efficacy standards mandated in healthcare. By developing initial models, designers and engineers can investigate various functionalities and obtain critical feedback, ultimately fostering innovations that enhance patient care.
The significance of prototype development is highlighted throughout its historical evolution, diverse applications, and its pivotal role in meeting regulatory standards. From low-fidelity models that encourage early-stage exploration to high-fidelity prototypes that closely resemble final products, each type is crucial in refining designs and improving the overall development process. The incorporation of advanced technologies, such as 3D printing and AI-driven engineering, has further transformed this field, facilitating quicker iterations and cost reductions while ensuring compliance with industry regulations.
Given these insights, it is clear that effective prototyping practices are not just advantageous but vital for success in the medical device sector. As manufacturers navigate the complexities of compliance and pursue innovation, prioritizing robust prototype development can result in safer, more effective products that ultimately improve patient outcomes. Adopting these practices will not only mitigate risks but also position companies to excel in a competitive market where quality and compliance are of utmost importance.
What is prototype development?
Prototype development is the process of creating an initial model or sample of a product, particularly important in the medical equipment sector for testing and validating concepts, functionality, and design before full-scale production.
Why is prototyping important in the medical field?
Prototyping is crucial in the medical field because it helps ensure that instruments meet necessary standards for patient care and safety, allowing for the exploration of usability, safety, and compliance with regulatory standards.
How does prototyping improve product design?
By iterating on prototypes, teams can refine their designs based on feedback and testing results, leading to more effective and safer medical devices.
What role does rapid modeling play in prototype development?
Rapid modeling accelerates the development cycle by allowing for quicker iterations and faster market entry, which is essential in a competitive environment.
How does effective modeling impact financial risks in product development?
Effective modeling can mitigate the financial risks associated with product development, making it a strategic necessity for manufacturers who want to innovate while adhering to strict industry standards.
