Master FMEA Failure Mode: A Step-by-Step Guide for Medical Devices
Introduction Identifying potential failure modes in medical devices is crucial not only...

In the complex realm of medical devices, software malfunctions can have severe repercussions. Thus, the implementation of Software Failure Mode and Effects Analysis (FMEA) serves as a vital safeguard. This systematic approach not only identifies potential failure modes but also prioritizes them according to their severity, thereby enhancing product reliability and ensuring adherence to stringent regulations.
However, a significant challenge remains: how can we effectively navigate the complexities of this process? Identifying best practices is essential to transform FMEA from a mere compliance exercise into a robust framework that improves patient safety and device performance.
is a systematic approach that is essential for identifying within the software components of . This process and prioritizes potential issues based on their severity and likelihood of occurrence. In the medical equipment sector, where software malfunctions can lead to and regulatory non-compliance, the use of software FMEA is critical. A recent study highlighted that fewer than 2% of FDA-approved AI/ML devices were supported by randomized clinical trials, underscoring the need for robust evaluation methodologies, such as , to enhance product reliability.
The importance of software FMEA extends beyond mere compliance; it is vital for and maintaining manufacturer credibility. By proactively addressing potential issues, engineers can substantially mitigate the risk of expensive recalls or redesigns. The FDA's updated guidance emphasizes the necessity for of medical devices, further reinforcing the significance of Failure Mode and Effects Analysis in ensuring that products meet stringent safety standards like . As the market for AI products is projected to expand significantly, with estimates reaching $255 billion by 2033, the integration of software FMEA will be crucial for navigating the complexities of and enhancing the overall safety of medical technologies.
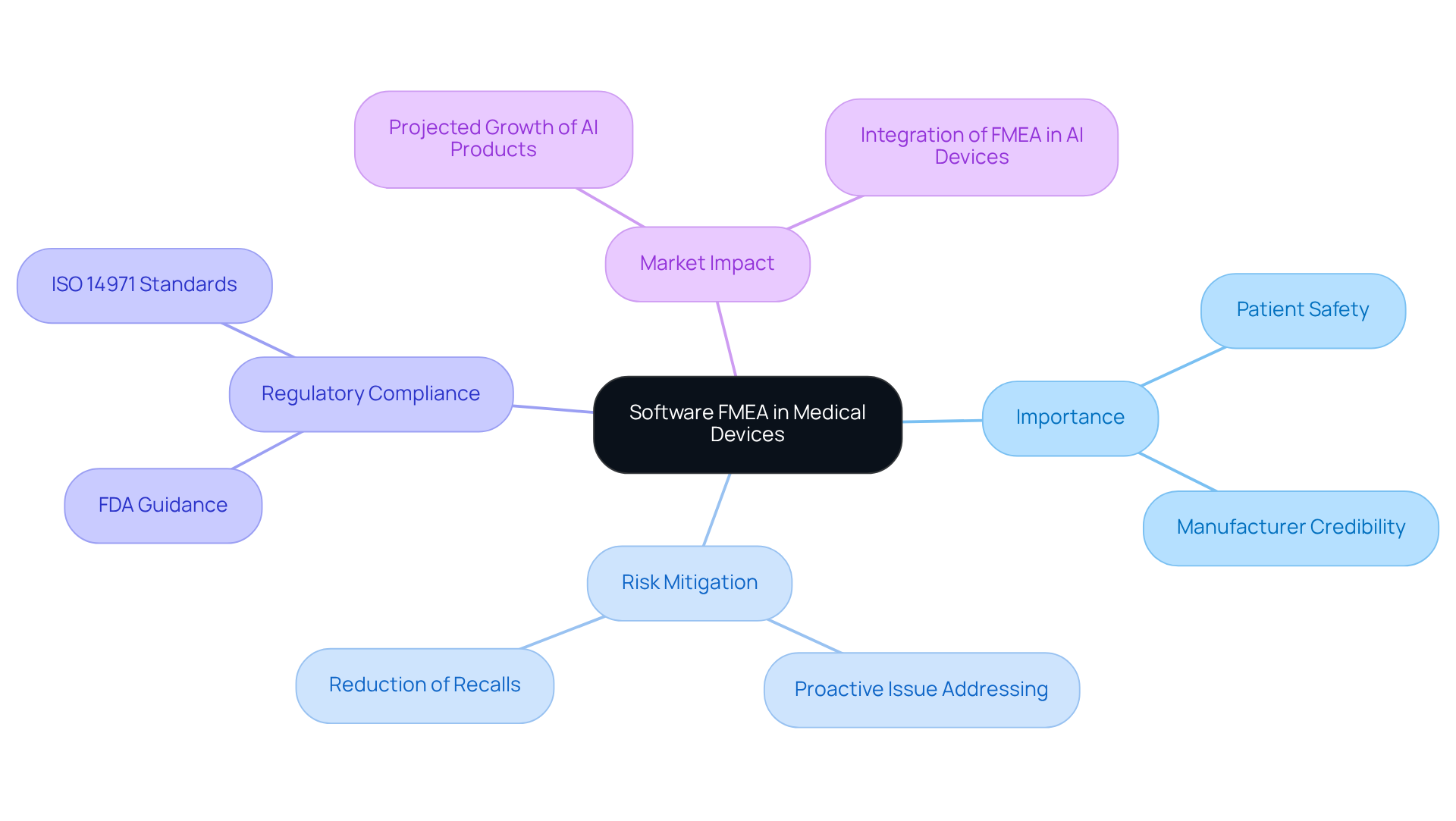
To conduct an , it is essential to establish clear objectives to guide the analysis. These objectives may encompass:
By setting specific goals, such as minimizing the likelihood of software-related failures by a defined percentage or , teams can focus their efforts on the most significant risks.
Failure Mode and Effects Analysis can be applied at any stage of the design process or throughout the lifecycle of a product or service, facilitating continuous improvement. Furthermore, , enabling teams to monitor progress and make necessary adjustments during development. This structured approach not only enhances the quality of the FMEA but also promotes a culture of ongoing improvement within the organization. As Ryan Detwiller states, " are the goals," underscoring the importance of clear objectives in achieving these outcomes.
Moreover, implementing robust testing systems is crucial for ensuring the quality and reliability of electronic equipment design, particularly in the . Voler Systems offers vital documentation compliance support for startups, assisting them in navigating effectively. However, it is important to avoid viewing merely as a checklist, as this can lead to ineffective hazard management. By concentrating on significant objectives, organizations can , ultimately resulting in safer medical equipment.
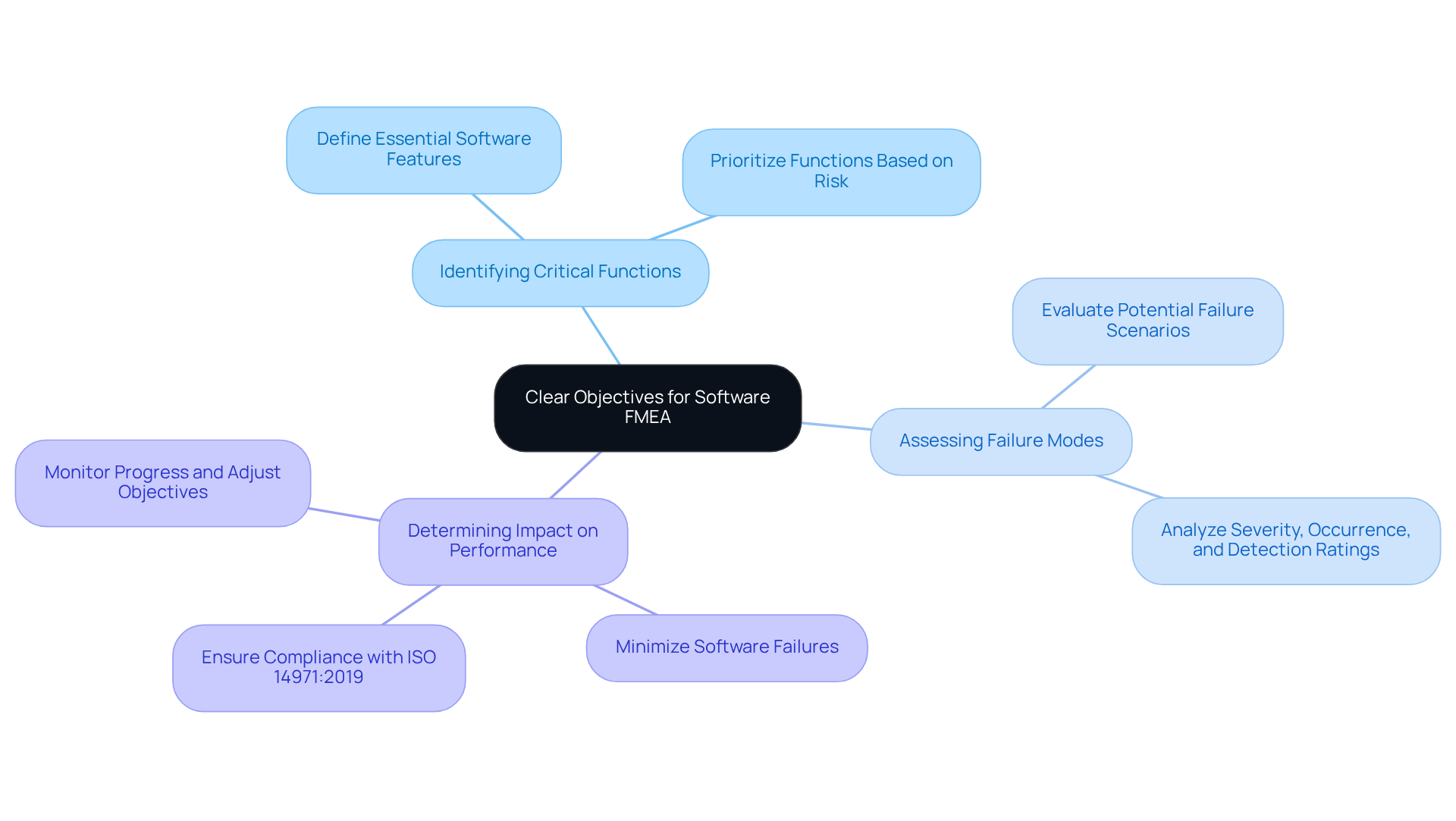
A is essential for ensuring a comprehensive . Begin by assembling a cross-functional group that includes software engineers, quality assurance professionals, and regulatory experts. This diverse composition minimizes blind spots and fosters collaboration, which is critical for . A typical core group for software FMEA might include representatives from software development, quality assurance, and regulatory compliance, ensuring that all critical perspectives are considered.
Next, define the scope of the analysis by identifying the software components and their interactions within the medical device. This step is vital for understanding how different elements may affect one another and contribute to potential failure modes. Systematically identify these failure modes for each software component, assessing their causes and effects. Utilizing tools such as risk matrices can help prioritize identified risks based on their severity and likelihood, enabling the group to focus on the most critical issues first.
Documentation is another key aspect of the process. Results should be documented in a clear and structured manner, ensuring that all members can access and comprehend the analysis. This organized approach not only improves the quality of the analysis but also aids communication and cooperation among team members. Voler Systems provides essential for medical equipment startups, including the creation of user manuals that conform to regulatory standards. For instance, MediHeart Devices Inc. effectively employed SoftComply Risk Manager Plus to record their FMEA process, linking uncertainties to specific product requirements and verification tests.
Statistics indicate that efficient cooperation among departments is crucial for the successful management of uncertainties in medical devices, with organizations utilizing . Moreover, the severity rating of data integrity issues is approximately 9, underscoring the critical nature of challenges that cross-functional teams must address. The 2017 WannaCry ransomware attack, which impacted over 40 hospitals, exemplifies the real-world consequences of inadequate management, highlighting the necessity of a systematic method in software FMEA. Marion Lepmets, CEO, emphasizes that , reinforcing the significance of this practice. By implementing this structured approach, groups can enhance the safety and effectiveness of medical devices, ultimately leading to improved patient outcomes.
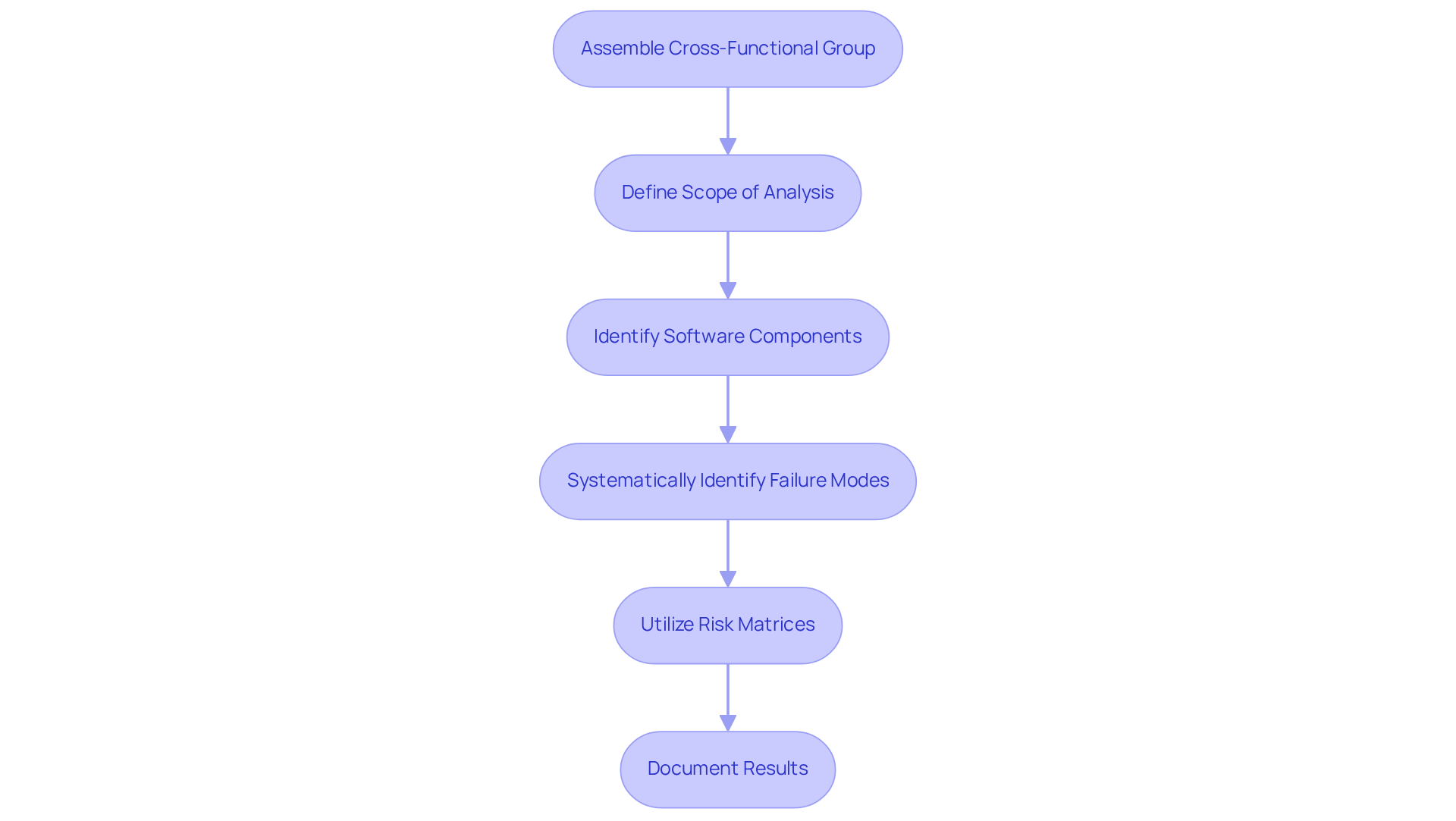
Common pitfalls in Software Failure Mode and Effects Analysis (FMEA) include:
To address these issues, it is essential to in the FMEA process from the outset. This involvement should extend beyond software engineers to include , who can offer valuable insights.
Statistics indicate that significantly enhances the quality of outcomes, as diverse perspectives lead to more comprehensive assessments of potential issues. For example, for successful FMEAs, as it provides the necessary incentives and resources. Additionally, teams should establish clear objectives and maintain throughout the analysis process.
Regular evaluations and revisions of the FMEA can help identify new risks or changes in software functionality that may impact the assessment. By addressing these common mistakes, teams can of their , which ultimately enhances the safety and effectiveness of their medical products. At Voler Systems, the integration of FMEA into our operational systems has demonstrated improvements in collaboration and decision-making, thereby enhancing the and ensuring the safety and effectiveness of .
Implementing effective Software Failure Mode and Effects Analysis (FMEA) is crucial in the medical device industry, where software reliability directly influences patient safety and regulatory compliance. This structured approach not only identifies potential failure modes but also prioritizes risks, ensuring that critical issues are addressed proactively. By integrating Software FMEA into the design and development processes, manufacturers can enhance the safety and effectiveness of their products, ultimately maintaining trust and credibility within the healthcare sector.
Throughout this article, key practices for successful Software FMEA have been highlighted, including:
Emphasizing the significance of continuous evaluation and documentation, it becomes evident that avoiding common pitfalls - such as inadequate stakeholder involvement and unclear goals - can lead to more reliable outcomes. By focusing on these best practices, organizations can significantly improve their FMEA processes and ensure that their medical devices meet stringent safety standards.
In conclusion, embracing the principles of Software FMEA transcends mere compliance; it represents a commitment to safeguarding patient health and advancing the quality of medical technology. As the healthcare landscape continues to evolve, the integration of robust evaluation methodologies will be essential for navigating regulatory complexities and fostering innovation. Organizations are encouraged to adopt these practices diligently, ensuring that their medical devices are not only effective but also safe for patient use.
What is Software FMEA?
Software FMEA (Failure Mode and Effects Analysis) is a systematic approach used to identify potential failure modes within the software components of medical equipment, assess the impacts of these failures, and prioritize issues based on their severity and likelihood of occurrence.
Why is Software FMEA important in medical devices?
Software FMEA is crucial in medical devices because software malfunctions can lead to significant safety risks and regulatory non-compliance. It helps safeguard patient health, maintains manufacturer credibility, and mitigates the risk of expensive recalls or redesigns.
What recent findings highlight the need for Software FMEA in medical devices?
A recent study found that fewer than 2% of FDA-approved AI/ML devices were supported by randomized clinical trials, indicating a need for robust evaluation methodologies like Software FMEA to enhance product reliability.
How does the FDA view Software FMEA?
The FDA emphasizes the necessity for continuous monitoring and performance evaluation of medical devices, reinforcing the significance of Software FMEA in ensuring that products meet stringent safety standards, such as ISO 14971.
What is the projected market growth for AI products, and how does it relate to Software FMEA?
The market for AI products is projected to expand significantly, with estimates reaching $255 billion by 2033. The integration of Software FMEA will be crucial for navigating regulatory compliance complexities and enhancing the overall safety of medical technologies.
