Introduction
Selecting embedded software for medical devices is a critical endeavor that significantly impacts patient safety and care. As healthcare technology rapidly advances, it becomes essential for stakeholders to understand the key characteristics and benefits of embedded systems to optimize device performance. However, this landscape presents numerous challenges, including regulatory compliance and integration issues. Consequently, organizations must navigate these complexities to ensure they select the most suitable software solutions for their medical devices.
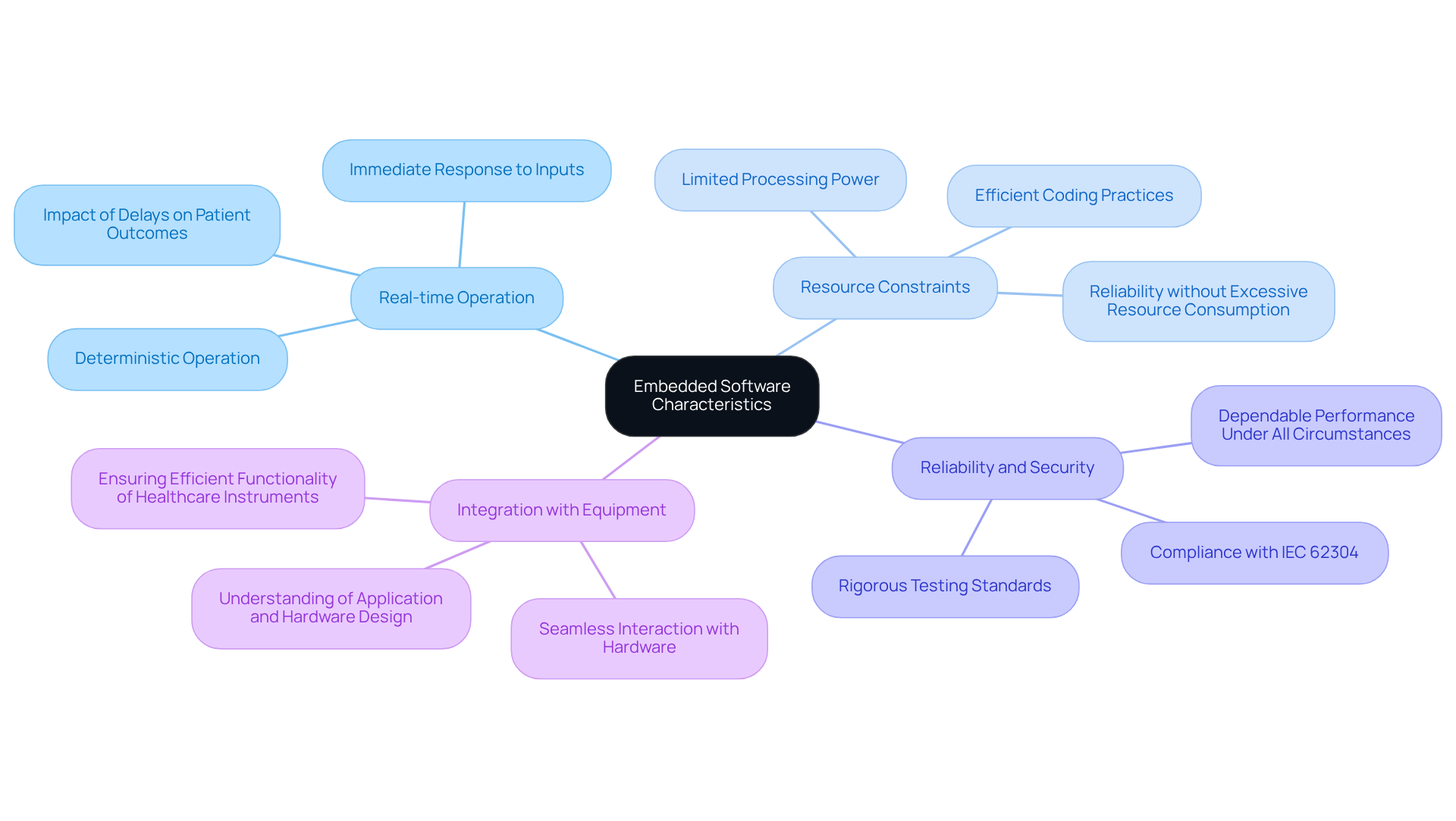
Understand Key Characteristics of Embedded Software
are meticulously crafted to manage hardware within , often operating under stringent constraints. The key characteristics include:
- : Immediate responses to inputs are critical in many medical devices, making real-time processing an essential feature. The capacity of to deliver is vital, as even millisecond delays can significantly affect patient outcomes.
- : Embedded systems typically operate with limited processing power and memory, necessitating efficient coding practices. This efficiency is particularly crucial in healthcare applications that utilize , where devices must function reliably without excessive resource consumption.
- : Given the essential nature of medical applications, must undergo rigorous testing to ensure dependable performance under all circumstances. Adherence to standards such as is crucial for maintaining the safety and effectiveness of .
- : The application must interact seamlessly with the hardware components of the apparatus, requiring a comprehensive understanding of both application and hardware design. This integration is essential for ensuring that can perform their intended functions efficiently.
Recognizing these traits enables stakeholders to with the specific requirements of their healthcare instruments, ultimately ensuring safety and effectiveness.
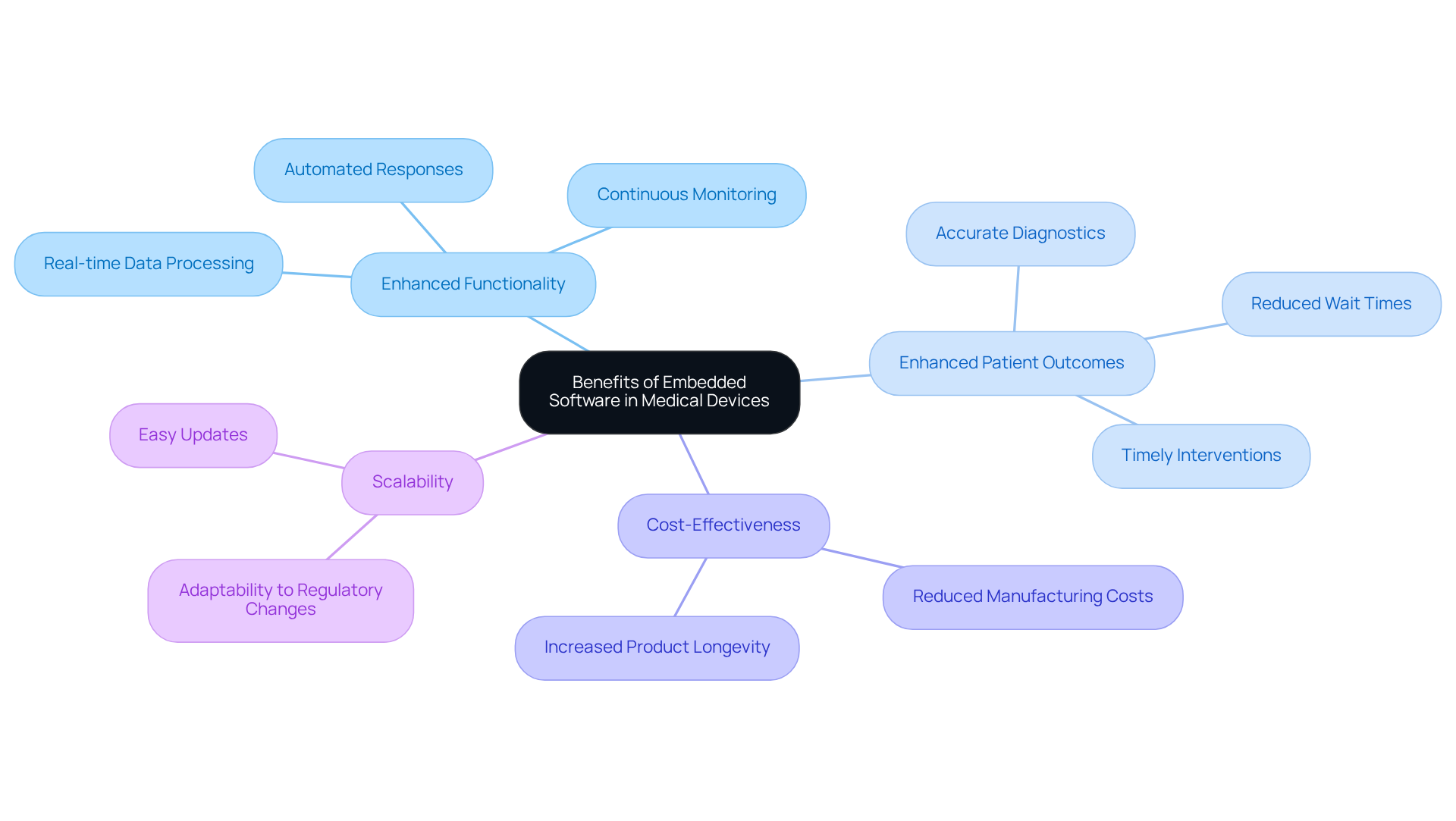
Identify Benefits of Embedded Software in Medical Devices
play a crucial role in advancing , particularly in the realm of innovative wearable health products that continuously monitor vital signs. This technology provides numerous benefits that significantly impact patient care and operational efficiency:
- Enhanced Functionality: equips medical devices with sophisticated capabilities, including , continuous monitoring, and automated responses. These features are vital for devices such as wearable ECG monitors, which can detect abnormal heart rhythms and promptly alert healthcare professionals, thereby advancing cardiac monitoring and transitioning from tethered systems to truly wireless solutions, as demonstrated in our case studies.
- Enhanced Patient Outcomes: By facilitating accurate diagnostics and enabling timely interventions, integrated applications markedly improve and the quality of care. For example, can analyze patient data more swiftly than manual methods, thereby reducing wait times and enhancing treatment accuracy. This is particularly significant given that over 200 public cases of medical equipment vulnerabilities were reported between 2021 and 2023, underscoring the necessity for robust to ensure patient safety and compliance with .
- Cost-Effectiveness: Efficient lead to and increased product longevity. This not only lowers overall expenses for healthcare providers but also ensures that equipment remains reliable over time, which is essential in such as hospitals.
- Scalability: As healthcare technology evolves, embedded programs can be easily updated or modified to incorporate new functionalities without necessitating a complete hardware overhaul. This flexibility is crucial for maintaining compliance with changing regulatory standards and addressing the dynamic needs of healthcare.
These advantages highlight the importance of selecting high-quality to ensure optimal performance of healthcare tools while adhering to stringent regulatory standards. The global embedded application market is projected to reach USD 30.2 billion by 2030, reflecting the growing significance and investment in embedded applications within the healthcare equipment sector. As noted by Exaud, "Embedded programs have become the unseen foundation of healthcare technology," emphasizing their essential role in modern health tools. Testimonials from industry leaders further validate the impact of Voler Systems in fostering innovation and compliance in this rapidly evolving field.
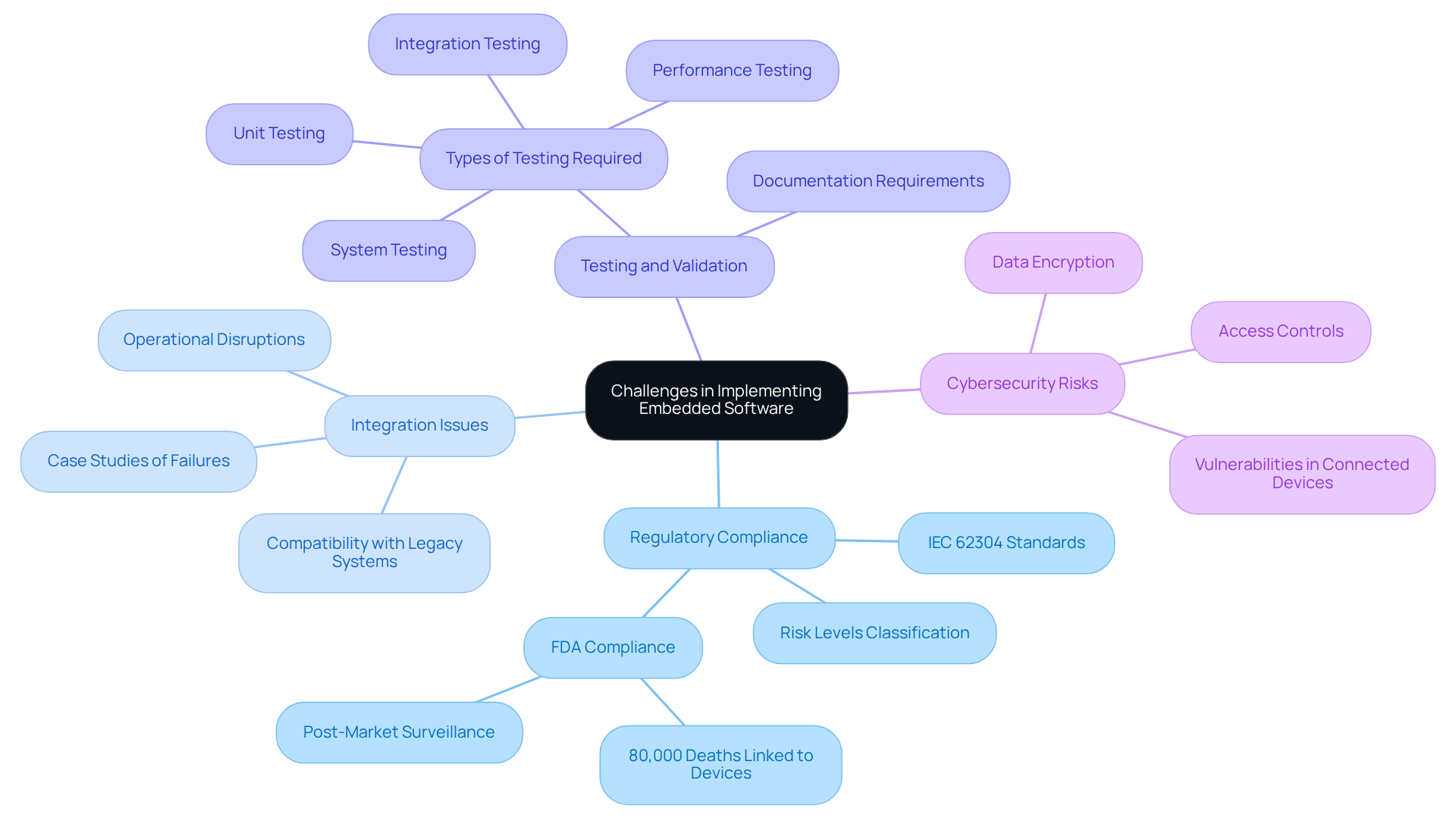
Address Challenges in Implementing Embedded Software
Implementing presents several significant challenges:
- : Navigating the complex landscape of medical device regulations is crucial for market approval. Adherence to standards such as is vital, as it categorizes applications into risk levels, which in turn affects development intensity and regulatory obligations. The U.S. FDA has linked over 80,000 deaths and 1.7 million injuries to medical devices in the past decade, highlighting the necessity of stringent .
- : The seamless incorporation of embedded programs with existing hardware can be particularly challenging, especially when interfacing with legacy systems. Case studies indicate that failures in integration can lead to significant operational disruptions and risks to , underscoring the importance of thorough during the development process.
- : Rigorous testing is essential to ensure that the system performs reliably under all conditions. This process is resource-intensive and must include unit testing, integration testing, performance testing, and system testing to meet . The standard mandates comprehensive documentation throughout the software development lifecycle, which is critical for maintaining compliance.
- : As medical equipment becomes increasingly interconnected, they face heightened vulnerabilities to cyber threats. Manufacturers must implement robust security measures, including data encryption and access controls, to protect patient information and ensure compliance with regulatory standards. The stakes are particularly high for connected technologies, where cybersecurity vulnerabilities can emerge rapidly, necessitating prompt actions while ensuring .
By addressing these challenges proactively, stakeholders can navigate the complexities of implementing embedded software products more effectively, ultimately leading to successful project outcomes.
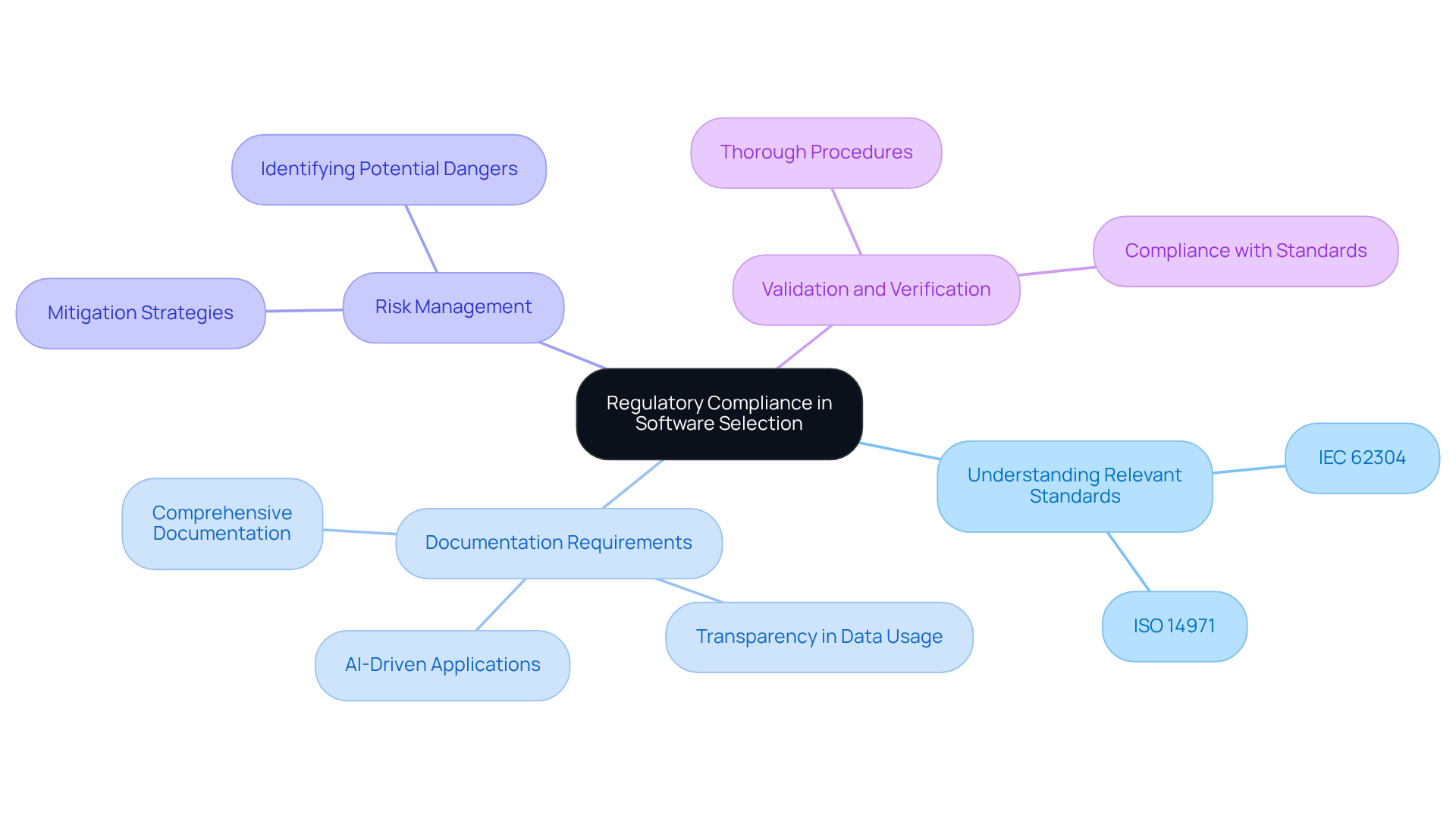
Ensure Regulatory Compliance in Software Selection
When selecting embedded software products for medical devices, is essential. Key considerations include:
- : It is crucial to familiarize yourself with essential standards such as IEC 62304, which outlines software lifecycle processes, and ISO 14971, which focuses on . These standards provide a framework that ensures development aligns with industry expectations.
- : Comprehensive documentation must be maintained throughout the . This practice not only demonstrates compliance but also facilitates audits and regulatory reviews, ensuring that all processes are transparent and traceable. Transparency in documentation is vital, as it enables clear communication of data usage and underlying logic, particularly in AI-driven applications. , assisting startups in navigating these regulatory challenges effectively.
- : A comprehensive strategy should be created to identify potential dangers linked to the application. This plan must outline mitigation strategies to address these risks, ensuring that security is prioritized throughout the development process.
- : Thorough procedures must be implemented to ensure that the application meets all security and performance standards. This step is crucial for confirming that the system functions as intended and complies with regulatory standards.
By prioritizing these elements of , organizations can significantly minimize risks and enhance the safety and effectiveness of their embedded software products, ensuring a successful transition to manufacturing while aligning with best practices in engineering design.
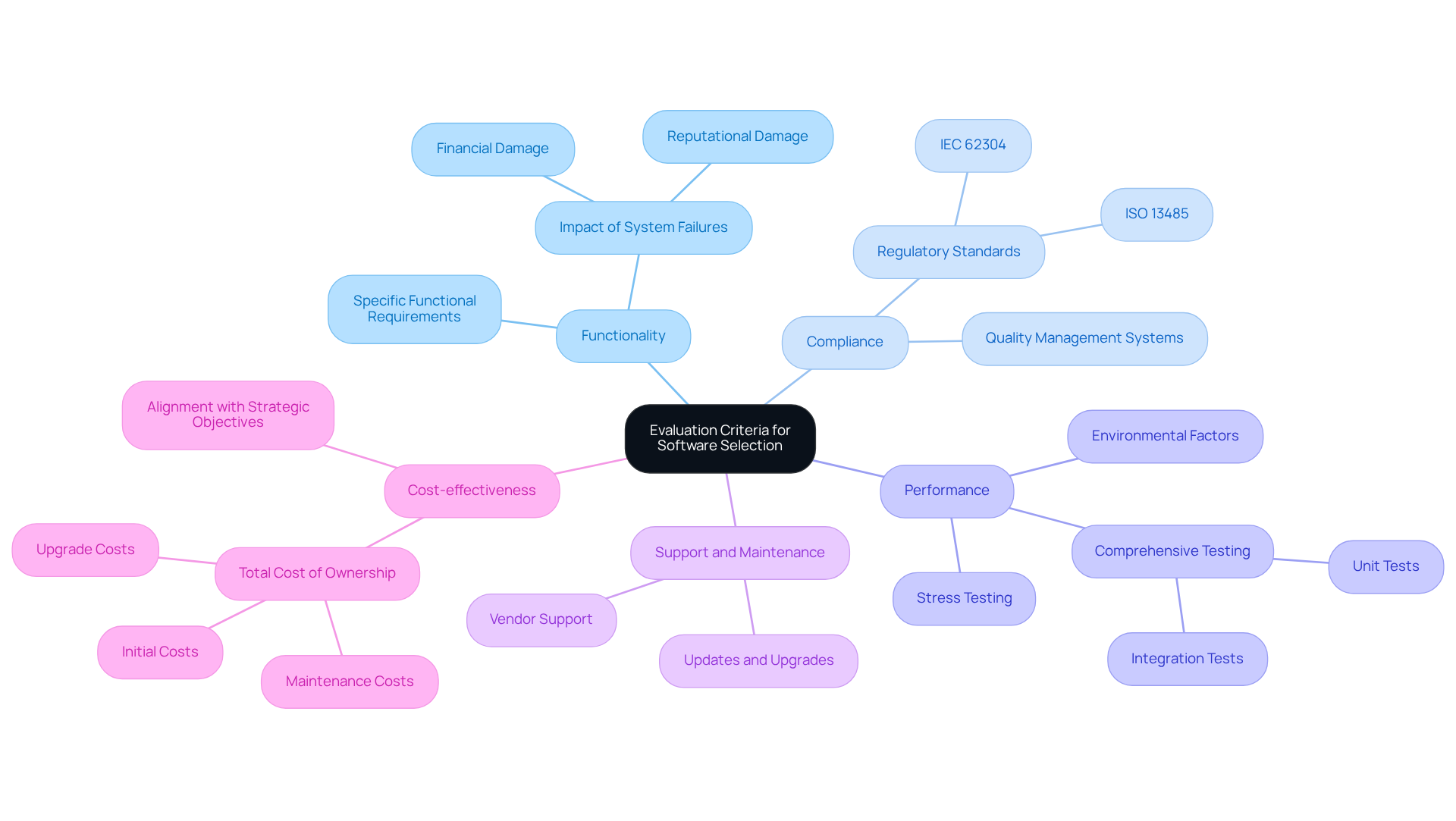
Establish Evaluation Criteria for Software Selection
When selecting for health equipment, it is essential to define clear to ensure adherence and performance. The following key factors should be considered:
- Functionality: Assess whether the software fulfills the specific functional requirements of the . This evaluation is critical, as system failures can lead to , with recall costs often exceeding millions.
- Compliance: Verify that the system complies with relevant , such as . Adhering to these standards is vital for maintaining quality management systems and ensuring patient safety.
- Performance: Analyze the system's performance under various conditions, including stress testing and environmental factors, to guarantee reliability and responsiveness. Comprehensive testing, including unit and integration tests, is necessary to confirm that the software meets security and functionality standards.
- Support and Maintenance: Evaluate the vendor's ability to provide . Effective maintenance is crucial for the long-term performance of equipment, particularly in light of evolving regulatory requirements and technological advancements.
- Cost-effectiveness: Examine the , which encompasses initial costs, maintenance, and potential upgrades. A thorough understanding of costs enables organizations to align their investments with strategic objectives.
By applying these criteria, organizations can make informed decisions that not only fulfill regulatory obligations but also of used in s. Case studies indicate that lead to improved software reliability and a reduced risk of recalls, highlighting the significance of a structured evaluation process.
Conclusion
Selecting the appropriate embedded software products for medical devices is essential for ensuring safety, efficiency, and compliance. A thorough understanding of the unique characteristics of embedded software-such as real-time operation, resource constraints, and the necessity for reliability-forms the foundation for making informed decisions. By concentrating on these aspects, stakeholders can align software choices with the specific requirements of healthcare instruments, ultimately enhancing patient care.
This article presents several key arguments, including the advantages of embedded software, such as improved functionality and better patient outcomes, alongside the challenges related to implementation, including regulatory compliance and integration issues. By establishing clear evaluation criteria that encompass functionality, compliance, performance, support, and cost-effectiveness, organizations can effectively navigate the complexities of software selection. This structured approach not only mitigates risks but also promotes the longevity and reliability of medical devices.
In conclusion, the selection of embedded software in medical devices is a critical process that requires careful consideration of various factors. As the healthcare landscape continues to evolve, prioritizing quality and compliance in embedded software will be vital for advancing patient care and meeting regulatory standards. Stakeholders are encouraged to adopt these best practices, ensuring that the embedded solutions chosen will not only address current needs but also adapt to future challenges in the healthcare sector.
Frequently Asked Questions
What are the key characteristics of embedded software in medical devices?
The key characteristics include real-time operation, resource constraints, reliability and security, and integration with equipment. Real-time operation ensures immediate responses to inputs, resource constraints require efficient coding, reliability and security involve rigorous testing, and integration with equipment ensures seamless interaction with hardware components.
Why is real-time operation important in embedded software for healthcare?
Real-time operation is critical because immediate responses to inputs can significantly affect patient outcomes. Delays, even in milliseconds, can impact the effectiveness of medical devices.
What challenges do embedded systems face due to resource constraints?
Embedded systems typically operate with limited processing power and memory, necessitating efficient coding practices to ensure reliable performance without excessive resource consumption.
How does reliability and security affect embedded software in medical applications?
Given the essential nature of medical applications, embedded software must undergo rigorous testing to ensure dependable performance. Adherence to standards like IEC 62304 is crucial for maintaining safety and effectiveness.
What benefits do embedded systems provide in medical devices?
Benefits include enhanced functionality, improved patient outcomes, cost-effectiveness, and scalability. These systems enable real-time data processing, accurate diagnostics, reduced manufacturing costs, and the ability to update functionalities without hardware changes.
How do embedded systems improve patient outcomes in healthcare?
By facilitating accurate diagnostics and enabling timely interventions, embedded systems improve patient well-being and the quality of care, reducing wait times and enhancing treatment accuracy.
What is the significance of cost-effectiveness in embedded systems for healthcare?
Cost-effectiveness leads to reduced manufacturing costs and increased product longevity, lowering overall expenses for healthcare providers and ensuring reliable equipment over time.
How does scalability benefit healthcare technology?
Scalability allows embedded programs to be easily updated or modified to incorporate new functionalities, ensuring compliance with changing regulatory standards and addressing dynamic healthcare needs.
What is the projected growth of the embedded application market in healthcare?
The global embedded application market is projected to reach USD 30.2 billion by 2030, reflecting the growing significance and investment in embedded applications within the healthcare equipment sector.
List of Sources
- Understand Key Characteristics of Embedded Software
- deliberatedirections.com (https://deliberatedirections.com/quotes-future-of-healthcare)
- How Embedded Software is transforming Healthcare devices (https://exaud.com/blog/embedded-software-solutions-in-healthcare-devices)
- Healthcare is Moving to an Embedded – First Architecture – Here’s What’s Driving It (https://multicorewareinc.com/healthcare-is-moving-to-an-embedded-first-architecture-heres-whats-driving-it)
- Embedded Systems Statistics By Market Size And Technologies (https://electroiq.com/stats/embedded-systems-statistics)
- Recent trends in embedded systems 2023 (https://srmtech.com/knowledge-base/blogs/recent-trends-in-embedded-system)
- Identify Benefits of Embedded Software in Medical Devices
- How Embedded Software is transforming Healthcare devices (https://exaud.com/blog/embedded-software-solutions-in-healthcare-devices)
- Embedded Systems Statistics By Market Size And Technologies (https://electroiq.com/stats/embedded-systems-statistics)
- testresults.io (https://testresults.io/articles/medtech-in-2026-the-trends-redefining-medical-device-innovation)
- Address Challenges in Implementing Embedded Software
- 7 quotes from 2023 to guide you on the medtech market (https://tiinatyni.com/blogi/7-quotes-from-2023-to-guide-you-on-the-medtech-market)
- Smart Devices, Tougher Rules: The Future of Medical Device Compliance | IoT For All (https://iotforall.com/security-safety-medical-iot)
- FDA Aligns U.S. Medical Device Rules with Global Standards (https://mddionline.com/regulatory-quality/medical-device-companies-must-navigate-historic-fda-rule-change)
- Medical Device Software Development (2026 Guide) - CLEIO (https://cleio.com/insights/blog/medical-device-software-development)
- medicaleconomics.com (https://medicaleconomics.com/view/the-2026-challenges-facing-the-medtech-industry)
- Ensure Regulatory Compliance in Software Selection
- lumafield.com (https://lumafield.com/case-studies/case-study-maintaining-fda-compliance-with-validated-ct-technology)
- FDA Device Guidance Agenda: What to Watch in 2026 (https://hoganlovells.com/en/publications/fda-device-guidance-agenda-what-to-watch-in-2026)
- variation.com (https://variation.com/product/statistical-procedures-for-the-medical-device-industry)
- FDA Aligns U.S. Medical Device Rules with Global Standards (https://mddionline.com/regulatory-quality/medical-device-companies-must-navigate-historic-fda-rule-change)
- faegredrinker.com (https://faegredrinker.com/en/insights/publications/2026/1/key-updates-in-fdas-2026-general-wellness-and-clinical-decision-support-software-guidance)
- Establish Evaluation Criteria for Software Selection
- Medical Device Software Development (2026 Guide) - CLEIO (https://cleio.com/insights/blog/medical-device-software-development)
- Medical Device Software Validation: Meeting FDA Expectations for Embedded and Cloud Systems | Arbour Group (https://arbourgroup.com/blog/2026/january/medical-device-software-validation-meeting-fda-expectations-for-embedded-and-cloud-systems)
- FDA Updates Broaden Wellness and Clinical Decision Support Software Devices Under Enforcement (https://emergobyul.com/news/fda-updates-broaden-wellness-and-clinical-decision-support-software-devices-under-enforcement)
- Embedded Software Testing for Medical Devices - Parasoft (https://parasoft.com/blog/medical-devices-embedded-software-testing)
- 7 quotes from 2023 to guide you on the medtech market (https://tiinatyni.com/blogi/7-quotes-from-2023-to-guide-you-on-the-medtech-market)






