5 Best Practices for Using 3D Prototypes in Medical Devices
Introduction The rapid evolution of technology has significantly transformed the...

The rapid evolution of 3D printing technology has fundamentally transformed the medical device industry, facilitating the development of complex prototypes that traditional manufacturing methods cannot replicate. This article explores best practices for employing 3D printing in medical device prototyping, providing insights into:
As the landscape grows increasingly intricate, manufacturers must consider how to ensure their prototypes not only comply with industry standards but also effectively address the unique challenges inherent in healthcare applications.
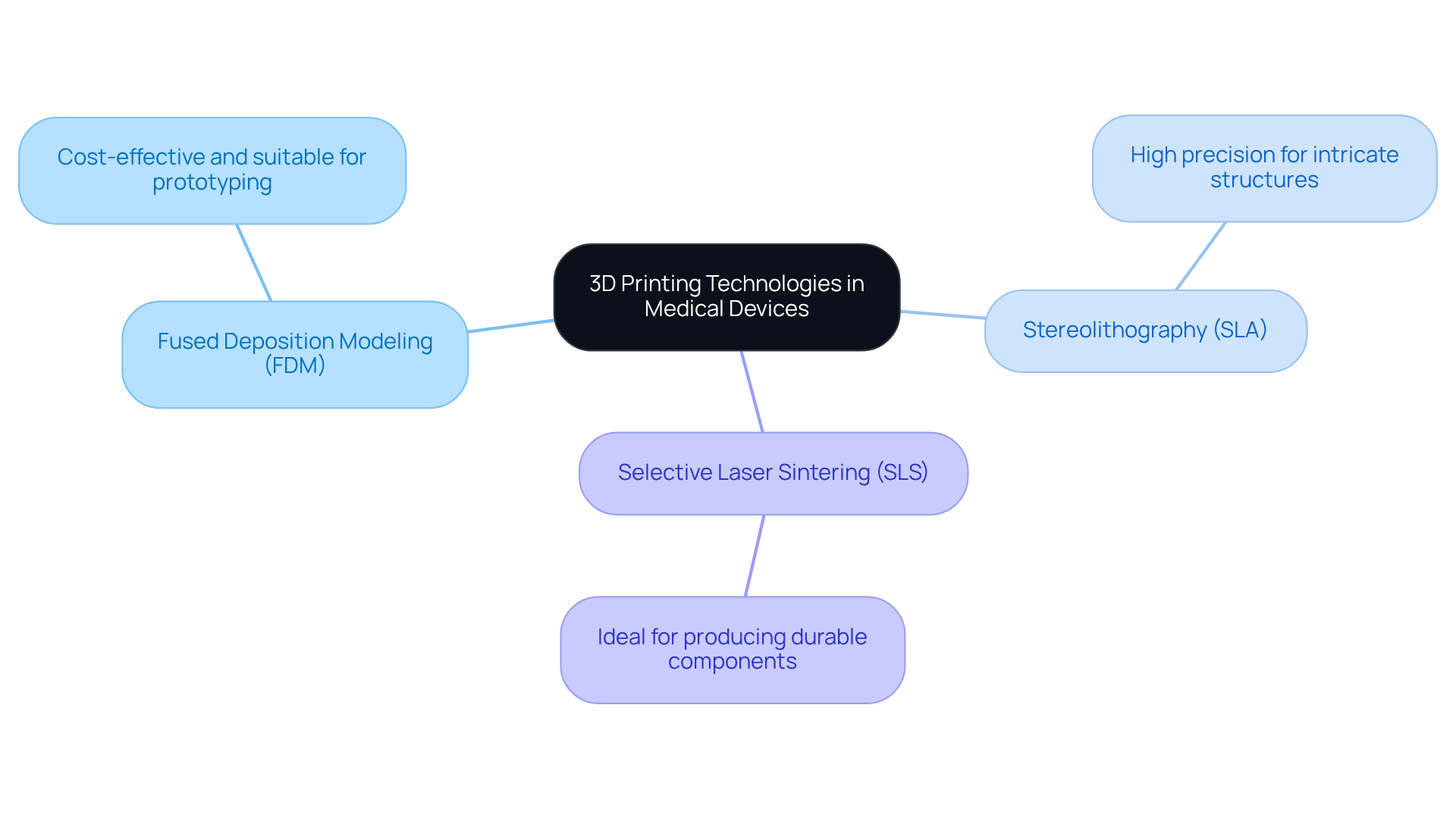
3D printing, also known as additive manufacturing, has significantly transformed the healthcare equipment industry by enabling the rapid creation of complex shapes that traditional manufacturing methods cannot achieve. The primary technologies involved include:
Each of these methods offers distinct advantages:
Understanding these technologies is crucial for , as they support manufacturers in developing that ensure designs are both innovative and compliant with . This expertise aids in selecting the most appropriate method for prototypes, streamlining hardware development, and facilitating a successful transition to manufacturing. For instance, a recent project involved the application of SLA technology to create a highly detailed prototype for a wearable health product, significantly reducing the time to market while ensuring compliance with .
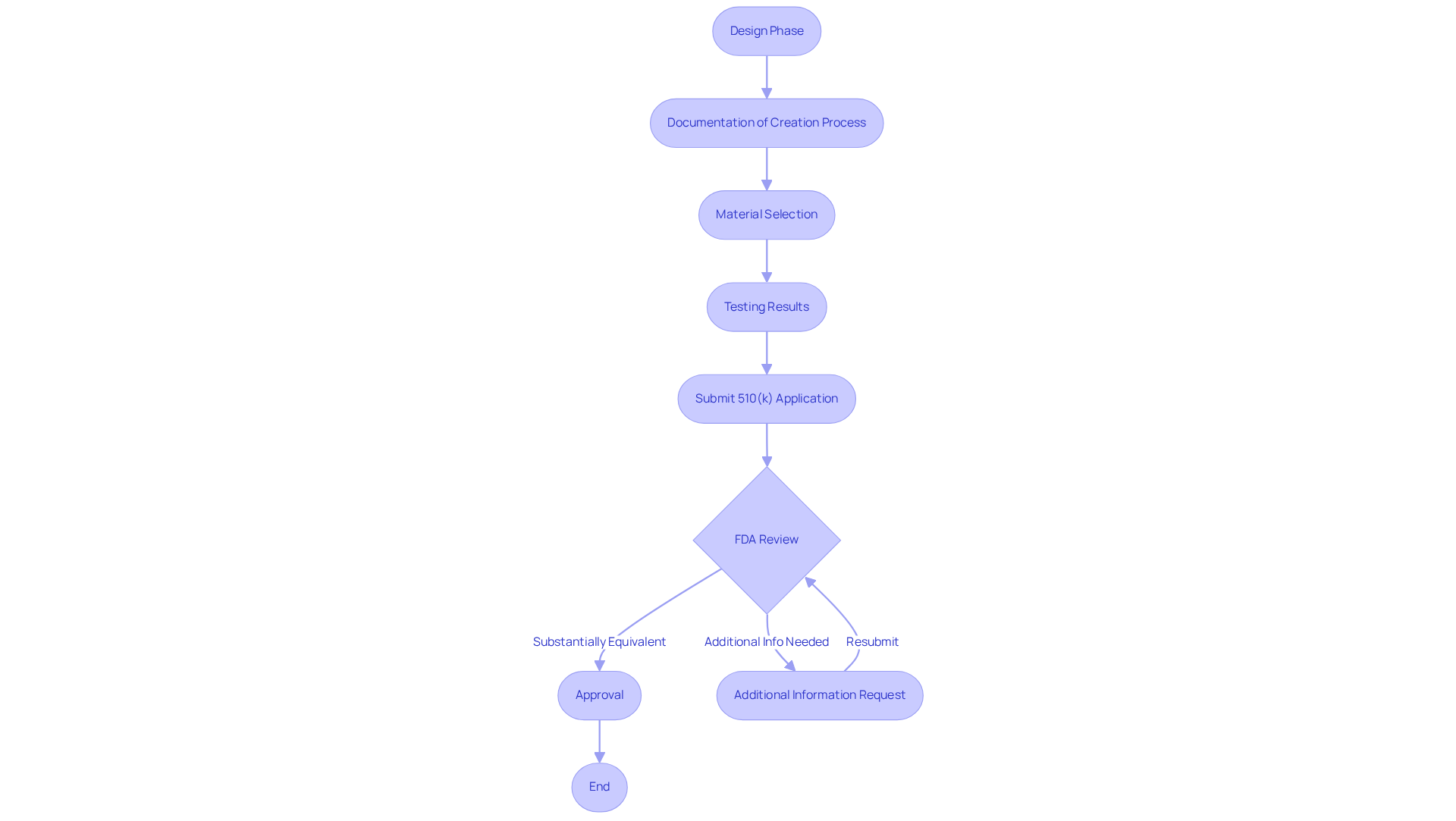
Adherence to established guidelines is essential in the development of . The , which are critical for ensuring product safety and efficacy. Documentation of the creation process, material selection, and testing results is essential to demonstrate compliance.
In 2021, for instance, 85% of 510(k) applications obtained a Substantially Equivalent decision, emphasizing the significance of . Interacting with oversight organizations early in the design process can help identify potential obstacles, streamline the approval process, and significantly reduce time to market. This proactive approach is crucial, especially considering that the is approximately five months, with many submissions facing additional information requests that can delay approval.
By adhering to and maintaining rigorous documentation, manufacturers can enhance their chances of successful .
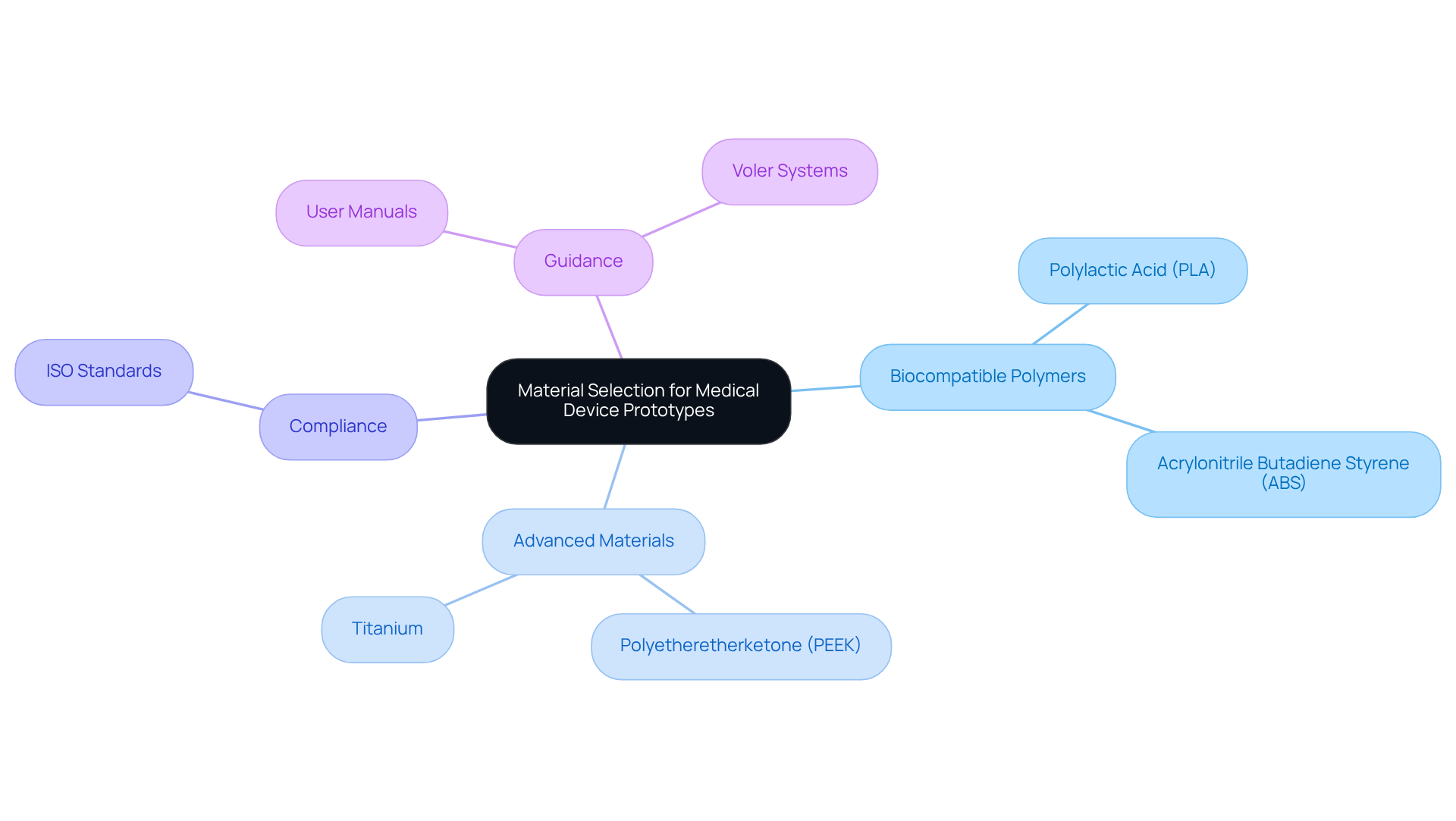
Selecting appropriate materials for 3D printed healthcare products is critical, as it significantly impacts biocompatibility, mechanical performance, and sterilization capabilities. , such as polylactic acid (PLA) and acrylonitrile butadiene styrene (ABS), are frequently utilized due to their advantageous properties and ease of printing.
In scenarios that demand greater strength and durability, advanced materials like polyetheretherketone (PEEK) and titanium are favored, particularly in load-bearing applications. It is essential to align the with the specific application of the equipment, ensuring it can endure the necessary operational conditions while remaining safe for patient interaction.
Compliance with is vital, as it guarantees that the materials employed meet the stringent safety requirements of the healthcare sector. User manuals are instrumental in guiding manufacturers through the , outlining the essential steps for .
Leveraging ' expertise in documentation compliance can aid manufacturers of in navigating these regulatory challenges effectively, ensuring they stay informed about current trends and advancements in prototype 3D printing.
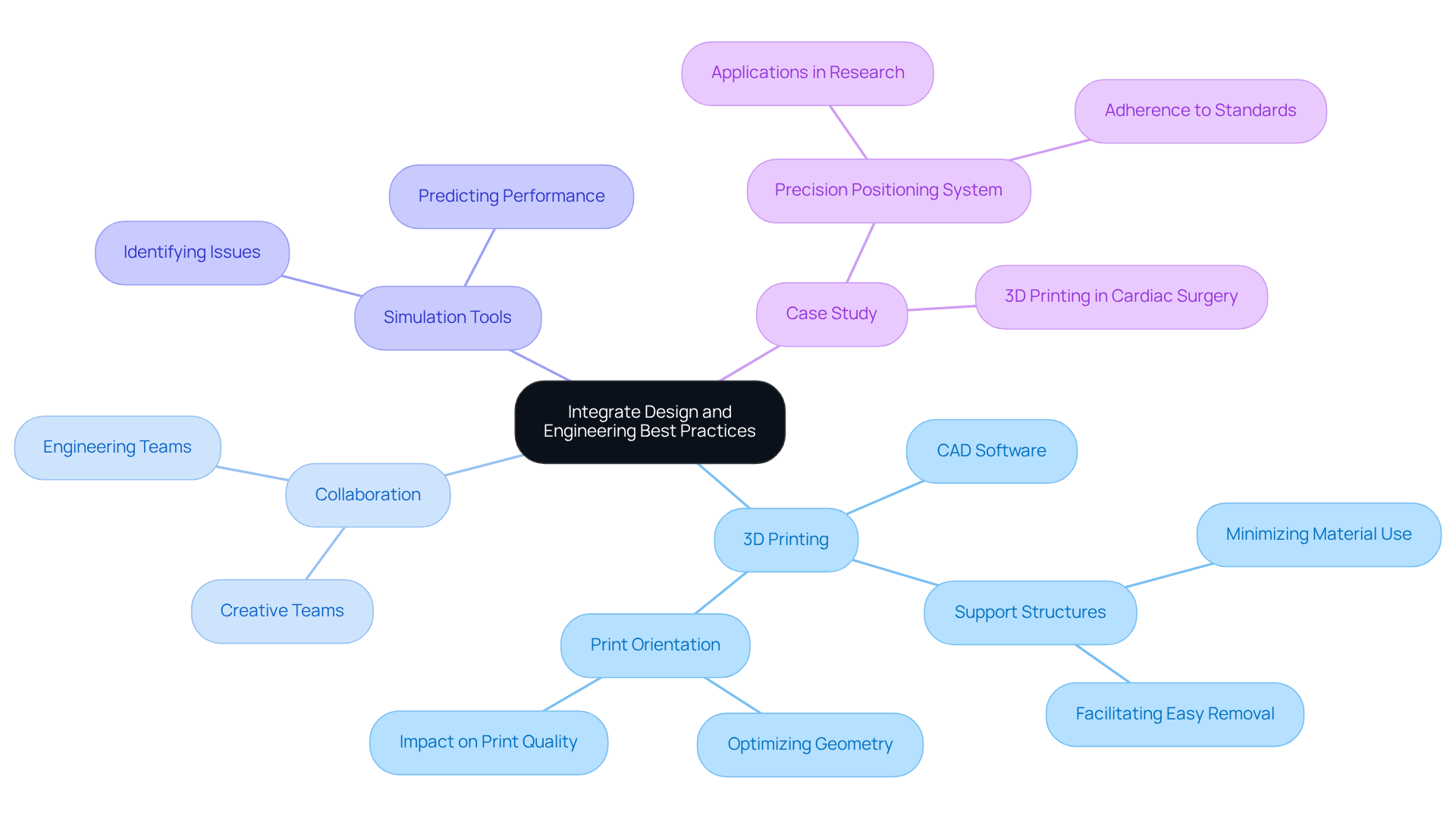
Creating effective medical device prototypes requires the incorporation of from the outset in prototype . Utilizing CAD software proficiently can significantly enhance models for by addressing critical factors such as and . Engineers often highlight the importance of geometry in models to optimize , thereby minimizing the need for and improving overall print quality.
is essential for fostering innovative solutions that enhance both functionality and manufacturability. Additionally, employing simulation tools enables teams to predict performance and identify potential issues before physical prototyping, ultimately conserving both time and resources. This cooperative approach is vital for increasing the success rates of , ensuring that prototypes are not only innovative but also functional and ready for regulatory examination.
exemplifies this through the successful creation of a Precision Positioning System for advanced research applications, adhering to emissions and ESD standards.
Iterative play a crucial role in the lifecycle. By generating and actively seeking feedback from stakeholders, designers can identify areas for improvement and implement necessary modifications. This approach not only enhances the but also assists in meeting by demonstrating a thorough testing process.
, such as , enable swift iterations, allowing teams to refine designs based on actual performance and user insights. Ultimately, this leads to the development of safer, more effective medical devices that closely align with and market demands.

The integration of 3D printing technology into the medical device industry has fundamentally transformed prototype development, underscoring the necessity of comprehending various printing methods and their applications. By utilizing techniques such as:
manufacturers can devise innovative designs that adhere to stringent regulatory standards while improving patient safety and product efficacy.
This article has highlighted key practices, including:
Each of these components is vital in ensuring that prototypes not only fulfill compliance requirements but also excel in functionality and user experience. The iterative prototyping approach further refines the development process, facilitating continuous feedback and enhancements that lead to safer and more effective medical devices.
Ultimately, adopting these best practices in 3D printing for medical devices is crucial for manufacturers striving to remain competitive in a swiftly evolving landscape. By prioritizing innovation, compliance, and collaboration, stakeholders can significantly elevate the quality of healthcare products, ensuring they are equipped to meet the needs of both practitioners and patients. The future of medical device development rests with those willing to adapt and innovate, making it essential to implement these strategies effectively.
What is 3D printing technology in medical devices?
3D printing, or additive manufacturing, is a technology that allows for the rapid creation of complex shapes in the healthcare equipment industry, which traditional manufacturing methods cannot achieve.
What are the primary technologies involved in 3D printing?
The primary technologies involved in 3D printing include Fused Deposition Modeling (FDM), Stereolithography (SLA), and Selective Laser Sintering (SLS).
What are the advantages of Fused Deposition Modeling (FDM)?
FDM is cost-effective and suitable for prototyping, making it accessible for developing initial designs.
What are the benefits of Stereolithography (SLA)?
SLA offers high precision for creating intricate structures, which is important for detailed prototypes.
Why is Selective Laser Sintering (SLS) advantageous?
SLS is ideal for producing durable components, which are essential for medical devices that require strength and reliability.
How does understanding these technologies benefit engineers at Voler Systems?
It helps engineers support manufacturers in developing prototype strategies that ensure innovative designs comply with industry standards, streamlining hardware development and facilitating a successful transition to manufacturing.
Can you provide an example of 3D printing application in medical devices?
A recent project involved using SLA technology to create a highly detailed prototype for a wearable health product, which significantly reduced the time to market while ensuring compliance with industry standards.
What regulatory guidelines must be followed for 3D printed medical devices?
Manufacturers must adhere to FDA guidelines, including pathways such as 510(k) clearance and pre-market approval, to ensure product safety and efficacy.
Why is documentation important in the 3D printing process?
Documentation of the creation process, material selection, and testing results is essential to demonstrate compliance with regulatory requirements.
What was the success rate for 510(k) applications in 2021?
In 2021, 85% of 510(k) applications received a Substantially Equivalent decision, highlighting the importance of careful preparation for compliance.
How can manufacturers streamline the FDA approval process?
Interacting with oversight organizations early in the design process can help identify potential obstacles and reduce time to market, as the average time for FDA 510(k) decisions is approximately five months.
What is the significance of maintaining rigorous documentation?
Maintaining rigorous documentation enhances manufacturers' chances of successful regulatory approval for prototype 3D printing.
