4 Best Practices for Embedded Application Development in Medical Devices
Introduction The landscape of medical device development is evolving rapidly, driven by...

Understanding the software product development process for medical devices is essential in a landscape where innovation intersects with stringent regulatory demands. This article explores the critical stages of development, from initial planning to ongoing maintenance, emphasizing how each phase is structured to ensure compliance and enhance patient safety. As technology and methodologies evolve rapidly, developers must navigate these complexities effectively while addressing both user needs and regulatory standards.
The software product development process for healthcare tools encompasses a series of organized stages that guide the creation of applications designed for health-related purposes. This process typically includes stages such as:
Each phase is crucial to ensure that the software adheres to regulatory standards, such as IEC 62304, and fulfills its intended functions.
[Voler Systems](https://volersystems.com), leveraging its extensive experience in developing numerous [health-related technologies](https://www.volersystems.com/blog/10-machine-to-machine-examples-transforming-industries-today) - including wearable devices, heart pumps, and liquid biopsy platforms - underscores the significance of AI-assisted engineering within this framework. The updated IEC 62304 introduces new Software Process Rigor Levels, replacing the previous safety classification system. This change simplifies risk management and enhances compliance with evolving regulatory expectations.
Understanding the software product development process is vital for developers to navigate the complexities of compliance and quality assurance in the healthcare sector. Successful case studies from Voler Systems illustrate the necessity of rigorous testing and validation, demonstrating that adherence to IEC 62304 not only ensures regulatory compliance but also bolsters patient safety and product reliability. Notably, the software as a medical device (SaMD) market is projected to reach $96.2 billion by 2033, highlighting the growing demand for innovative healthcare applications.
The evolution of product creation in the medical device industry has been shaped by significant milestones that highlight technological innovations and the evolving demands of the market. Initially, computer programming relied heavily on manual techniques, utilizing punch cards and basic coding languages. As technology advanced, structured methodologies such as the Waterfall model became prevalent, offering a more systematic approach to the software product development process.
The introduction of Agile methodologies in the early 2000s marked a pivotal shift in the software product development process, fostering iterative progress and adaptability, which are essential qualities in the rapidly evolving healthcare device landscape. Currently, the integration of artificial intelligence and machine learning into programming processes is driving further enhancements in efficiency and innovation, enabling the development of more intelligent and responsive healthcare solutions.
This transformation not only streamlines progress but also aligns with the industry's growing emphasis on real-time data utilization and patient-centered care.
The software product development process for healthcare tools includes several essential stages: planning, requirements analysis, design, implementation, testing, and maintenance. Each phase is critical to the overall success of the project.
Planning involves defining the project scope, objectives, and timelines, establishing the foundation for the entire development process. Voler Systems excels in this phase by ensuring that all aspects of the project align with regulatory requirements and user needs.
Requirements analysis is vital, focusing on gathering and documenting user needs alongside regulatory requirements. This phase is particularly important, as studies indicate that approximately 70% of medical device application projects fail during the testing stage due to inadequate requirements analysis. Voler Systems emphasizes comprehensive documentation compliance support, assisting startups in navigating the complexities of regulatory challenges.
Design translates these requirements into technical specifications, including system architecture and user interface design, ensuring that the application meets user expectations and adheres to regulatory standards such as IEC 62366 and ISO 14971. Voler Systems integrates advanced engineering solutions and AI-driven design to enhance the effectiveness of this phase.
Implementation is where the actual coding takes place, followed by rigorous testing to validate functionality, usability, and compliance with established standards. This phase is essential for confirming that the application fulfills its intended purpose and complies with regulatory guidelines. Voler Systems applies best practices in engineering design to ensure successful outcomes during this phase.
Finally, maintenance involves continuous support and updates to address any issues that arise post-launch, ensuring the system remains effective and compliant over time. Voler Systems' commitment to cutting-edge embedded systems design, particularly in enhancing battery longevity for wireless health-related equipment, is crucial for ensuring product dependability.
Understanding these stages of the software product development process is essential for manufacturers in the healthcare sector, as it helps guarantee that the application not only meets user requirements but also adheres to stringent regulatory criteria, ultimately enhancing patient safety and product efficacy. With the Software as a Medical Device (SaMD) market projected to reach a valuation of USD 167.59 billion by 2032, reflecting a compound annual growth rate (CAGR) of 23.6%, the importance of a structured creation process becomes increasingly relevant. According to Weronika Michaluk, MedTech Practice Lead at HTD Health, "It is necessary to implement thorough risk management activities throughout the SaMD lifecycle," underscoring the complexities involved in health technology creation.
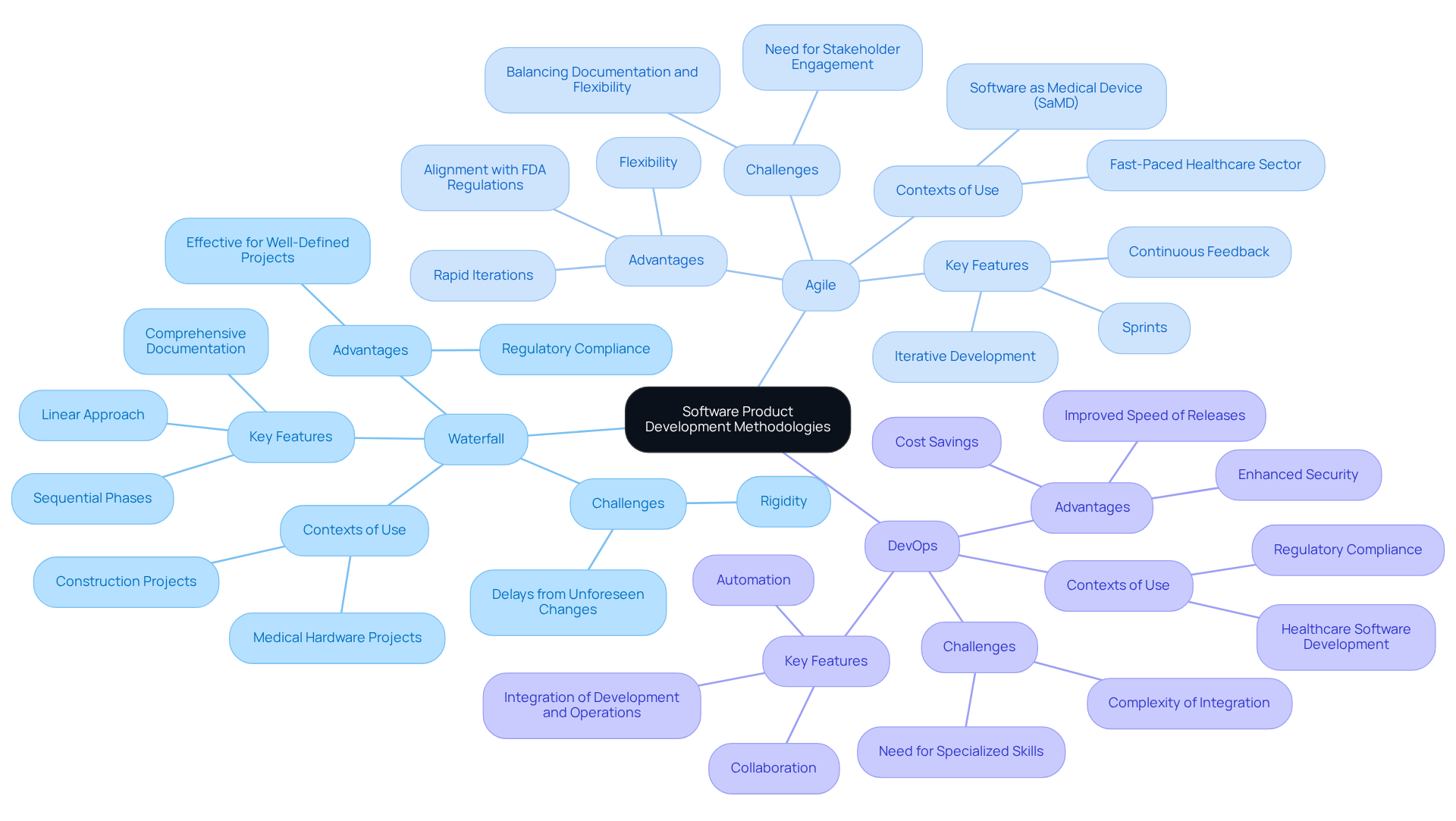
In the realm of the software product development process for healthcare devices, three primary methodologies stand out: Waterfall, Agile, and DevOps.
Waterfall represents a traditional linear approach, where each phase - requirements gathering, design, implementation, testing, and maintenance - must be completed in a sequential manner. This methodology proves particularly effective for projects with well-defined requirements, ensuring comprehensive documentation and strict adherence to regulatory standards. However, its inherent rigidity can result in delays when unforeseen changes occur during validation studies.
Agile, on the other hand, promotes an iterative development process that enables teams to adapt to changing user needs and regulatory requirements. By segmenting projects into short cycles known as Sprints, Agile fosters continuous feedback and rapid iterations, which are crucial in the fast-paced healthcare sector. This adaptability not only enhances product quality but also aligns well with FDA regulations, making it a preferred choice for numerous healthcare equipment developers.
DevOps merges development and operations, placing a strong emphasis on collaboration and automation to optimize the software product development process. This methodology significantly improves the speed and quality of releases, which is essential in an industry where timely compliance and patient safety are of utmost importance. Organizations that adopt mature DevOps practices can realize substantial cost savings; studies indicate that those with effective security automation save an average of $1.76 million per breach compared to their counterparts lacking such measures.
Each methodology possesses distinct strengths and is chosen based on specific project requirements, team dynamics, and regulatory considerations. Ultimately, the selection of methodology ensures that the final product is not only effective but also compliant with the stringent standards governing the medical device industry.
The software product development process for medical devices represents a structured and critical journey, ensuring the creation of safe, effective, and compliant healthcare applications. By navigating through stages such as:
developers can effectively address the unique challenges posed by the healthcare industry. This process not only adheres to regulatory standards but also prioritizes patient safety and product reliability.
Key insights throughout the article underscore the significance of each phase in the development cycle. The evolution from traditional methodologies like Waterfall to more adaptive approaches such as Agile and DevOps illustrates the industry's response to the fast-paced demands of healthcare technology. Furthermore, the integration of artificial intelligence and machine learning is reshaping how developers approach software creation, enhancing both efficiency and innovation in medical devices. Notably, the projected growth of the Software as a Medical Device market highlights the increasing relevance of a well-structured development process.
In conclusion, as the landscape of medical device software continues to evolve, embracing a comprehensive understanding of the software product development process is essential for success. Stakeholders in the healthcare sector must prioritize adherence to regulatory requirements and implement best practices to effectively navigate the complexities of this field. By doing so, they can not only meet the growing demand for innovative healthcare solutions but also ensure the safety and satisfaction of patients relying on these technologies.
What is the software product development process for healthcare tools?
The software product development process for healthcare tools includes organized stages such as planning, requirements gathering, design, implementation, testing, and maintenance to ensure the applications meet regulatory standards and fulfill their intended functions.
Why is the software product development process important in healthcare?
It is crucial for ensuring that software adheres to regulatory standards, such as IEC 62304, and for navigating the complexities of compliance and quality assurance in the healthcare sector.
What is IEC 62304 and how does it relate to software development in healthcare?
IEC 62304 is a regulatory standard that outlines the requirements for the life cycle of medical software. The updated version introduces new Software Process Rigor Levels, simplifying risk management and enhancing compliance with evolving regulatory expectations.
How does Voler Systems contribute to the software product development process?
Voler Systems leverages its extensive experience in developing health-related technologies, emphasizing the significance of AI-assisted engineering and showcasing successful case studies that highlight the importance of rigorous testing and validation.
What are the implications of adhering to IEC 62304 for software developers?
Adhering to IEC 62304 ensures regulatory compliance, bolsters patient safety, and enhances product reliability, which is essential for developers in the healthcare sector.
What is the projected market size for software as a medical device (SaMD)?
The software as a medical device market is projected to reach $96.2 billion by 2033, indicating a growing demand for innovative healthcare applications.
