Best Practices in Embedded System Software Development for Medical Devices
Introduction Embedded software is fundamental to the functionality of modern medical...

A comprehensive understanding of medical device software development is essential for ensuring patient safety and meeting regulatory standards. The IEC 62304 framework serves as a cornerstone in this domain, guiding manufacturers through the complexities of software classification and lifecycle management. Many developers struggle to grasp the complexities of IEC 62304, especially when balancing innovation with the stringent demands of safety and documentation.
Identifying best practices for the effective implementation of IEC 62304 is essential for enhancing the reliability of medical devices while minimizing risks and ensuring compliance. Without a robust approach to IEC 62304, the integrity of medical devices and patient safety may be at risk.
IEC 62304 serves as a critical framework for the life cycle management of medical devices, ensuring safety and compliance. This globally acknowledged standard outlines the life cycle activities for medical device applications, including development, maintenance, and decommissioning. It categorizes applications into three safety groups based on potential patient risk:
Compliance with IEC 62304 fulfills regulatory requirements and simplifies market entry, thereby enhancing patient safety. The standard emphasizes the importance of comprehensive documentation and traceability throughout the application life cycle, a key factor in achieving successful audits and regulatory assessments.
For instance, manufacturers are required to establish comprehensive application development plans that detail activities, responsibilities, and configuration management, ensuring clarity and effectiveness in the development process. Adhering to IEC 62304 guidelines fosters trust within the medical community, directly contributing to long-term business success and improved patient outcomes.
Voler Systems, with its extensive experience in developing hundreds of medical devices, including wearable devices, heart pumps, and liquid biopsy platforms, leverages AI-assisted engineering to optimize battery life and enhance product reliability. However, these oversights can lead to costly delays and regulatory setbacks. Therefore, neglecting this framework can result in compromised patient safety and increased liability. Understanding and implementing the 62304 software classification framework is essential not only for regulatory compliance but also for safeguarding patient health and enhancing market competitiveness.
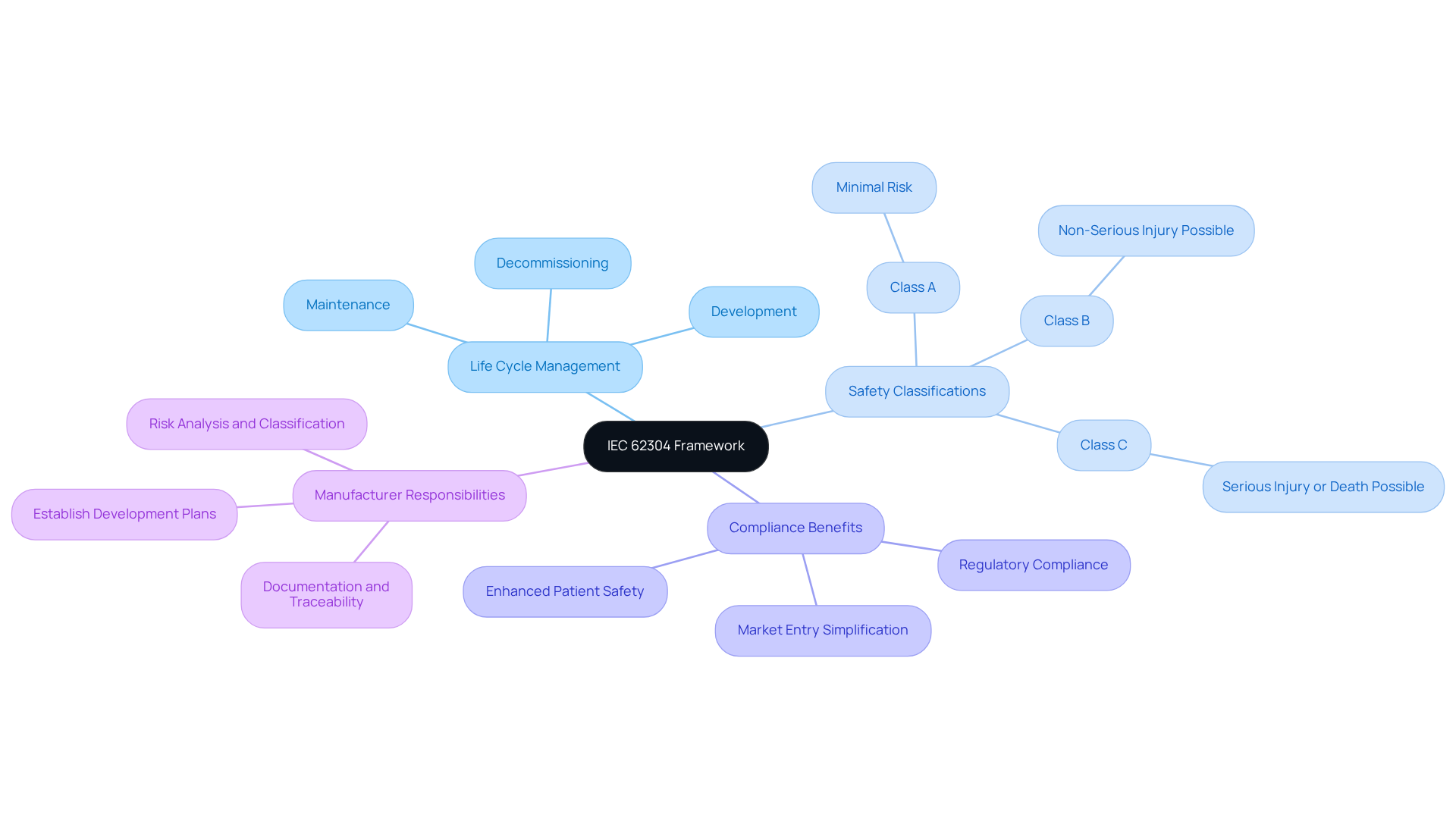
To ensure the safety of medical devices, developers must implement effective safety classification strategies through comprehensive risk assessments. This evaluation must address critical questions, including whether the program can contribute to hazardous situations and the potential consequences of system failures. Based on the findings, programs can be classified into three categories:
Each class entails specific requirements for documentation, testing, and validation. For example, Class C applications necessitate extensive verification and validation activities, including rigorous testing and comprehensive documentation to ensure safety and compliance. Implementing these classification strategies enables developers to meet essential safety and regulatory standards, ultimately resulting in the creation of safer medical devices. Failure to adhere to these strategies could lead to significant risks, undermining the safety of medical devices.
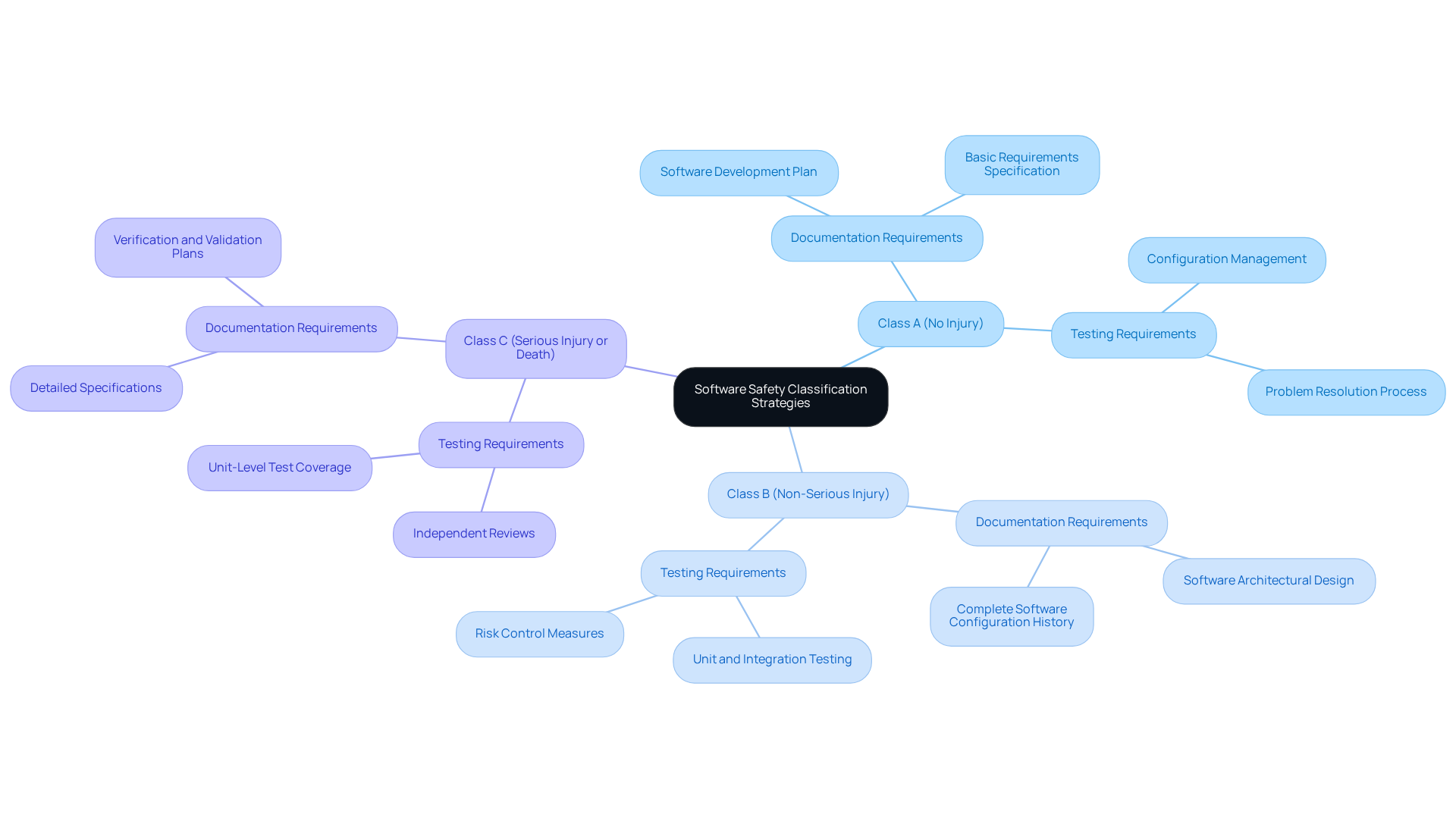
Implementing a structured application creation methodology is crucial for effectively managing the entire lifecycle of a project, including:
To ensure project success, developers must select an appropriate methodology, such as Agile or Waterfall, based on specific project requirements and team dynamics.
Defining clear roles and responsibilities is a fundamental practice that enhances project clarity and accountability, while maintaining comprehensive documentation and conducting regular reviews and audits further supports this clarity.
For instance, applying a phase-gate framework guarantees that each stage of advancement satisfies established criteria before moving forward.
This structured approach not only improves project outcomes but also ensures compliance with industry regulations, such as the 62304 software classification, by promoting traceability and accountability throughout the creation phase.
Ultimately, this structured approach fosters a culture of accountability and quality assurance in the development process.
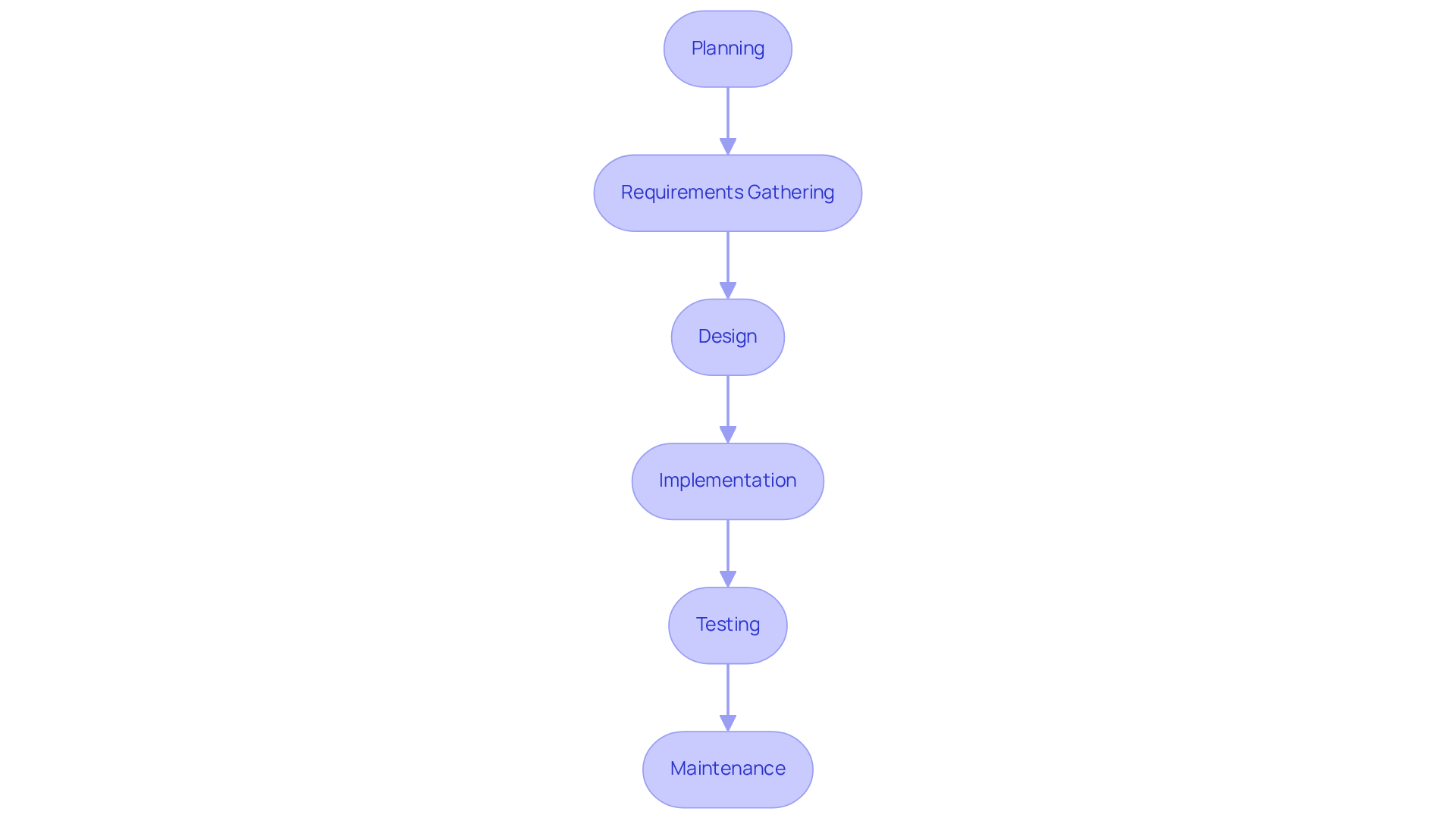
Inadequate hazard management in software development can lead to severe risks in medical device safety and compliance. Incorporating thorough hazard management strategies into the software development workflow is essential for recognizing and reducing potential dangers linked to medical devices. This integration relies on compliance with ISO 14971:2019, which provides a robust framework for managing uncertainties alongside the 62304 software classification. Key steps in this process include:
Developers must record all recognized threats along with their associated mitigation strategies, ensuring traceability to specific application requirements. Regular evaluations should be performed throughout the application lifecycle to adjust to any modifications in the program or its intended purpose. Incorporating hazard management into the development process enhances the safety and effectiveness of medical device software, ultimately safeguarding patients and ensuring adherence to the 62304 software classification regulatory standards. This proactive approach minimizes the risk of product recalls and ensures alignment with evolving regulatory standards, where compliance with ISO 14971 is increasingly acknowledged as crucial for effective management in medical device manufacturing. Furthermore, with the upcoming updates to IEC 62304 in 2026, including the introduction of new Software Process Rigor Levels, manufacturers must stay informed to ensure compliance and address the challenges posed by AI and machine learning in risk management. Voler Systems is at the forefront of these innovations, providing essential documentation compliance support for startups and established manufacturers alike, ensuring that they navigate regulatory challenges effectively while leveraging the latest advancements in artificial intelligence. Manufacturers who neglect these updates risk falling behind in compliance and compromising patient safety.
The IEC 62304 framework is critical for ensuring the safety and compliance of medical device software development. This standard categorizes software according to patient risk and provides a roadmap for manufacturers to meet regulatory requirements and ensure compliance for market entry. Adhering to these guidelines enhances patient safety and builds trust within the medical community, directly contributing to long-term success.
The article outlined four best practices for effective software classification in medical devices:
Each of these elements plays a pivotal role in ensuring that medical devices are developed with the utmost care and attention to safety. From conducting thorough risk assessments to maintaining clear documentation and structured methodologies, these practices are crucial for minimizing risks and enhancing the overall quality of medical software.
Manufacturers must remain informed and proactive as the medical device development landscape evolves, especially with the upcoming updates to IEC 62304. Embracing these best practices not only safeguards patient health but also positions companies to navigate regulatory challenges effectively. Neglecting these best practices jeopardizes patient safety and undermines competitive positioning in the evolving medical device landscape.
What is IEC 62304?
IEC 62304 is a critical framework for the life cycle management of medical devices, focusing on ensuring safety and compliance throughout the development, maintenance, and decommissioning of medical device applications.
Why is IEC 62304 important?
IEC 62304 is important because it helps fulfill regulatory requirements, simplifies market entry, enhances patient safety, and emphasizes comprehensive documentation and traceability, which are essential for successful audits and regulatory assessments.
How does IEC 62304 categorize medical device applications?
IEC 62304 categorizes medical device applications into three safety groups based on potential patient risk: Class A (minimal risk), Class B (non-serious injury possible), and Class C (serious injury or death possible).
What are the requirements for manufacturers under IEC 62304?
Manufacturers are required to establish comprehensive application development plans that detail activities, responsibilities, and configuration management to ensure clarity and effectiveness in the development process.
How does adherence to IEC 62304 guidelines benefit the medical community?
Adhering to IEC 62304 guidelines fosters trust within the medical community, contributing to long-term business success and improved patient outcomes.
What risks are associated with neglecting the IEC 62304 framework?
Neglecting the IEC 62304 framework can lead to costly delays, regulatory setbacks, compromised patient safety, and increased liability.
How can companies enhance their product reliability in line with IEC 62304?
Companies can enhance product reliability by leveraging AI-assisted engineering to optimize aspects such as battery life, as demonstrated by organizations like Voler Systems, which has extensive experience in developing medical devices.
