Introduction
In the intricate realm of medical device development, the integration of embedded system software is vital for ensuring compliance and safety. As healthcare technology advances, it becomes essential for manufacturers to understand and implement best practices to deliver reliable products. However, with the evolution of regulations and the increasing demand for user-centered designs, organizations face significant challenges. How can they navigate these complexities effectively while upholding high standards of quality and safety? This article examines four best practices that not only enhance the development process but also protect patient health and promote long-term success in the medical device industry.
Understand Regulatory Compliance and Safety Standards
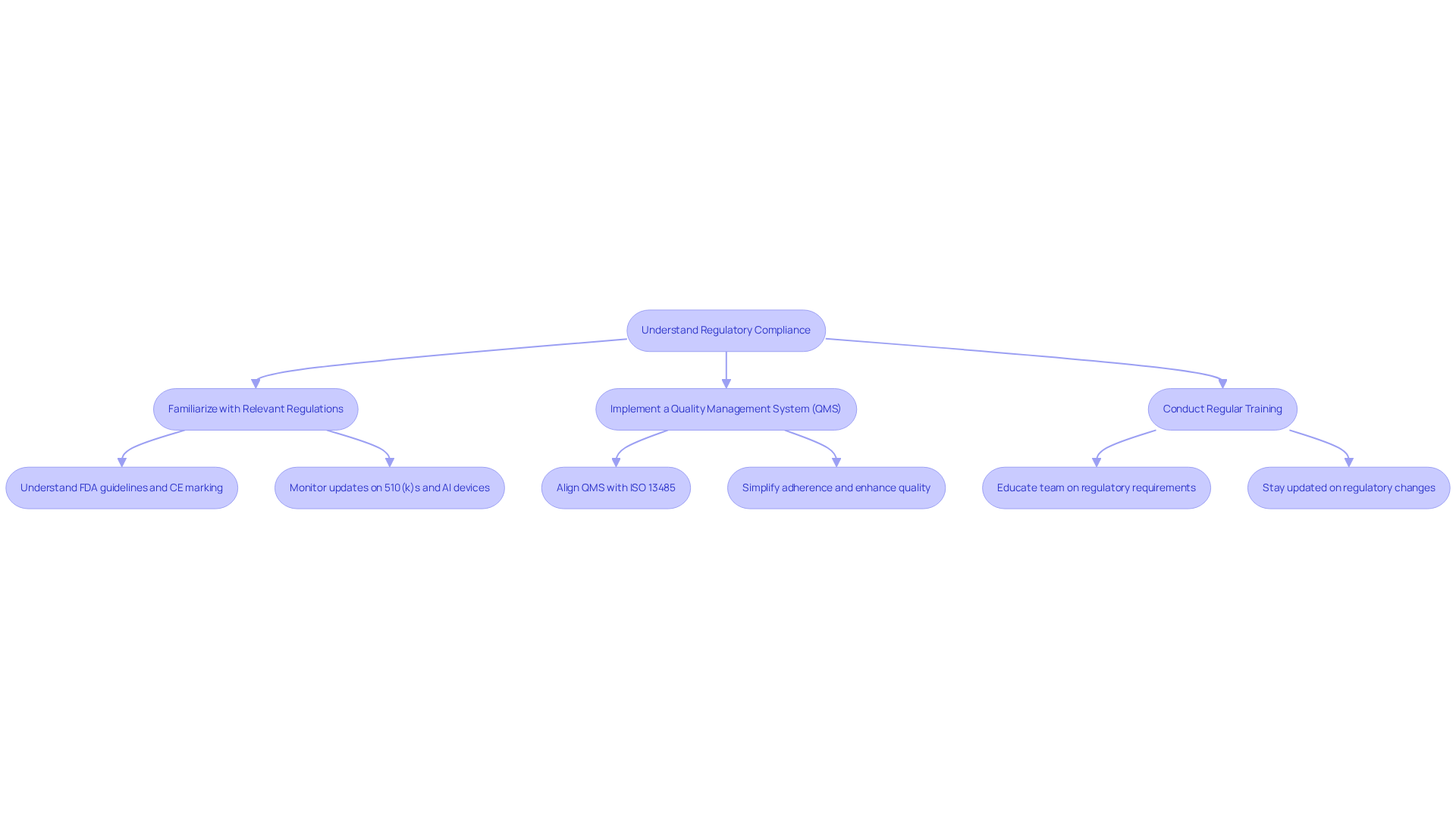
In the medical equipment sector, adherence to [[regulatory standards](https://www.volersystems.com/blog/10-essential-items-for-your-62304-checklist-in-medical-device-design)](https://www.volersystems.com/blog/10-essential-items-for-your-62304-checklist-in-medical-device-design) such as IEC 62304 and ISO 13485 is essential. These standards govern the software development lifecycle, encompassing risk management, design controls, and validation processes. To ensure compliance, teams should take the following steps:
- Familiarize with Relevant Regulations: Understand the specific regulations applicable to your device type and market, including FDA guidelines in the U.S. and CE marking in Europe. In 2025, the FDA approved a substantial number of 510(k)s and AI-enabled devices, reflecting the increasing significance of adherence in innovation.
- Implement a [[Quality Management System (QMS)](https://aabb.org/news-resources/news/article/2026/02/04/fda-implements-quality-management-system-regulation-for-medical-devices)](https://aabb.org/news-resources/news/article/2026/02/04/fda-implements-quality-management-system-regulation-for-medical-devices): Establish a QMS that aligns with ISO 13485 to simplify adherence and enhance product quality. The FDA's recent shift to the Quality Management System Regulation (QMSR) incorporates ISO 13485:2016, establishing a unified framework that eases adherence for manufacturers.
- Conduct Regular Training: Ensure that all team members are educated on regulatory requirements and best practices to foster a culture of quality and safety. Regular training helps teams stay updated on the latest regulatory changes, such as the updated IEC 62304 standard, which will introduce new Software Process Rigor Levels in 2026.
By integrating compliance into the creation process from the beginning, organizations can reduce risks and improve the dependability of their healthcare products. This proactive strategy not only fulfills regulatory requirements but also fosters the long-term success of health technology innovations.
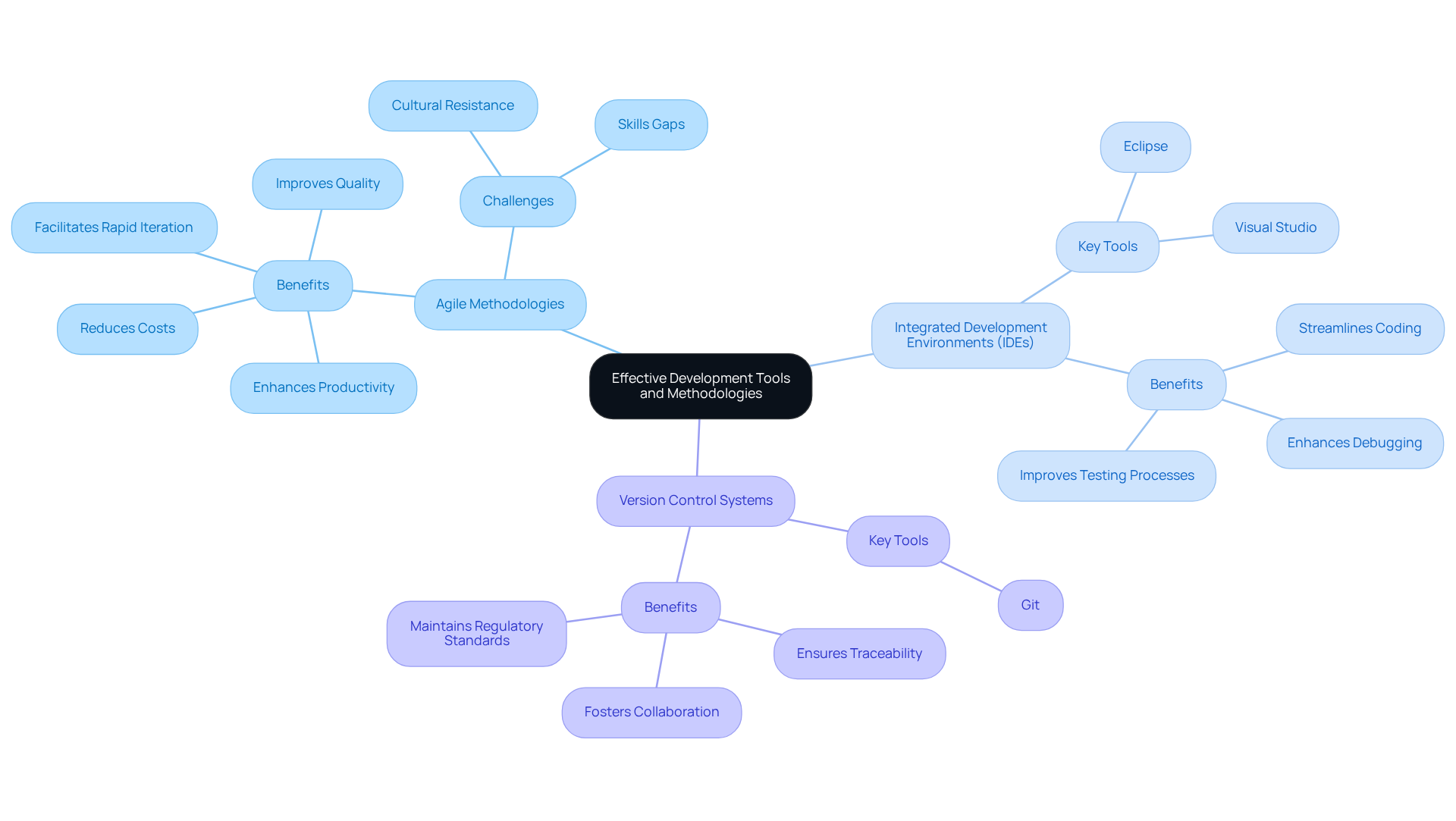
Selecting the appropriate tools and approaches is essential for enhancing the effectiveness and quality of embedded system software used in healthcare. Here are several best practices to consider:
- Adopt Agile Methodologies: Agile practices facilitate iterative development and adaptability, enabling teams to respond swiftly to changes and feedback. This flexibility is particularly crucial in the highly regulated healthcare equipment sector, where adherence and rapid iteration are necessary. By following best practices for successful engineering design projects, teams can ensure that their projects remain on time and within budget. Agile methodologies enhance productivity, reduce costs, and improve quality in the development of embedded system software, making them a valuable approach.
- Utilize Integrated Development Environments (IDEs): Integrated Development Environments such as Eclipse and Visual Studio streamline coding, debugging, and testing processes. These tools not only enhance productivity but also improve software quality by providing features that assist in error detection and resolution, which is vital for ensuring compliance in device development.
- Incorporate Version Control Systems: Implementing version control systems like Git is crucial for managing code changes effectively. These systems ensure traceability and foster collaboration among team members, which is essential for maintaining regulatory standards and quality throughout the project lifecycle. [[Voler Systems offers extensive documentation compliance assistance](https://volersystems.com)](https://volersystems.com) for startups in health technology, aiding in navigating regulatory challenges effectively.
By employing these approaches and resources, teams can improve their workflows, minimize errors, and uphold regulatory standards, ultimately leading to the successful development of high-quality healthcare products. Furthermore, it is important to recognize that 72% of research and development leaders in the healthcare sector identified organizational cultural resistance as a significant barrier to agile adoption. Addressing this challenge is vital for successful implementation.
Implement Comprehensive Testing and Validation Protocols
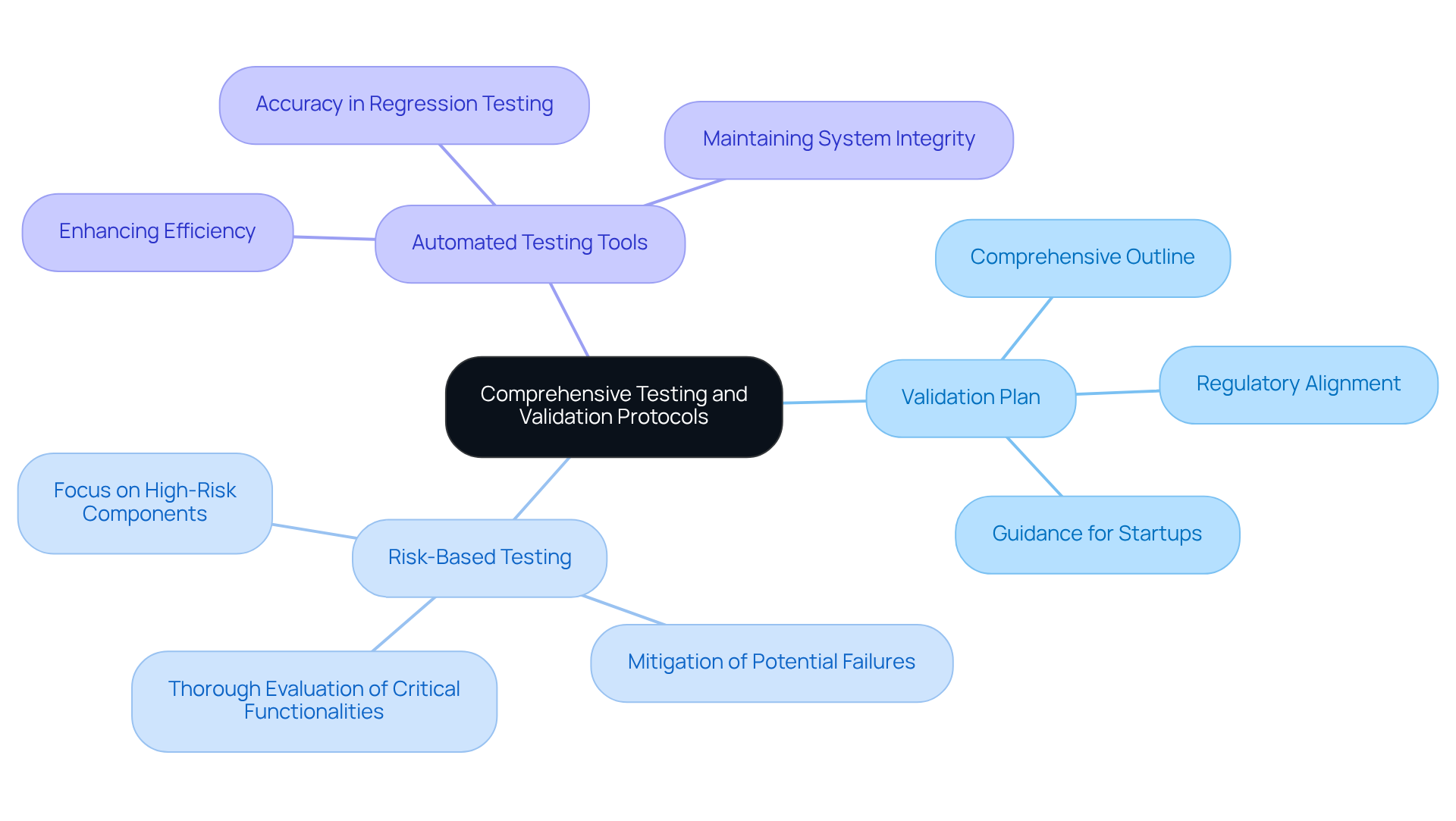
Testing and validation are crucial for ensuring the safety and compliance of healthcare software. Implementing best practices in this domain can significantly enhance product reliability and adherence to regulatory standards. Key strategies include:
- Develop a Validation Plan: Formulate a comprehensive validation plan that outlines testing objectives, methodologies, and acceptance criteria. This vital document guides the validation process and aligns with regulatory requirements, particularly for startups navigating the complexities of health product regulations with [[[support from Voler Systems](https://volersystems.com)](https://volersystems.com)](https://volersystems.com).
- Conduct Risk-Based Testing: Direct testing efforts toward high-risk software components to mitigate potential failures. This strategy prioritizes areas that could substantially affect patient safety, ensuring that critical functionalities undergo thorough evaluation.
- Utilize Automated Testing Tools: Adopt automated testing solutions to enhance efficiency and accuracy in regression testing. Automation helps confirm that new software modifications do not introduce defects, thereby maintaining the integrity of the system's performance.
Establishing a robust testing and validation framework not only meets regulatory standards but also enhances the overall reliability of healthcare products in practical applications. With software issues accounting for 23% of recalls in recent quarters, a proactive approach to validation is increasingly vital. As Brian Callahan, president of EEC & Associates, states, "Establishing a validation process is part of the price of staying in business." By steering clear of common pitfalls in testing and validation, manufacturers can ensure their software complies with regulatory standards and operates reliably.
Adopt a User-Centered Design Approach
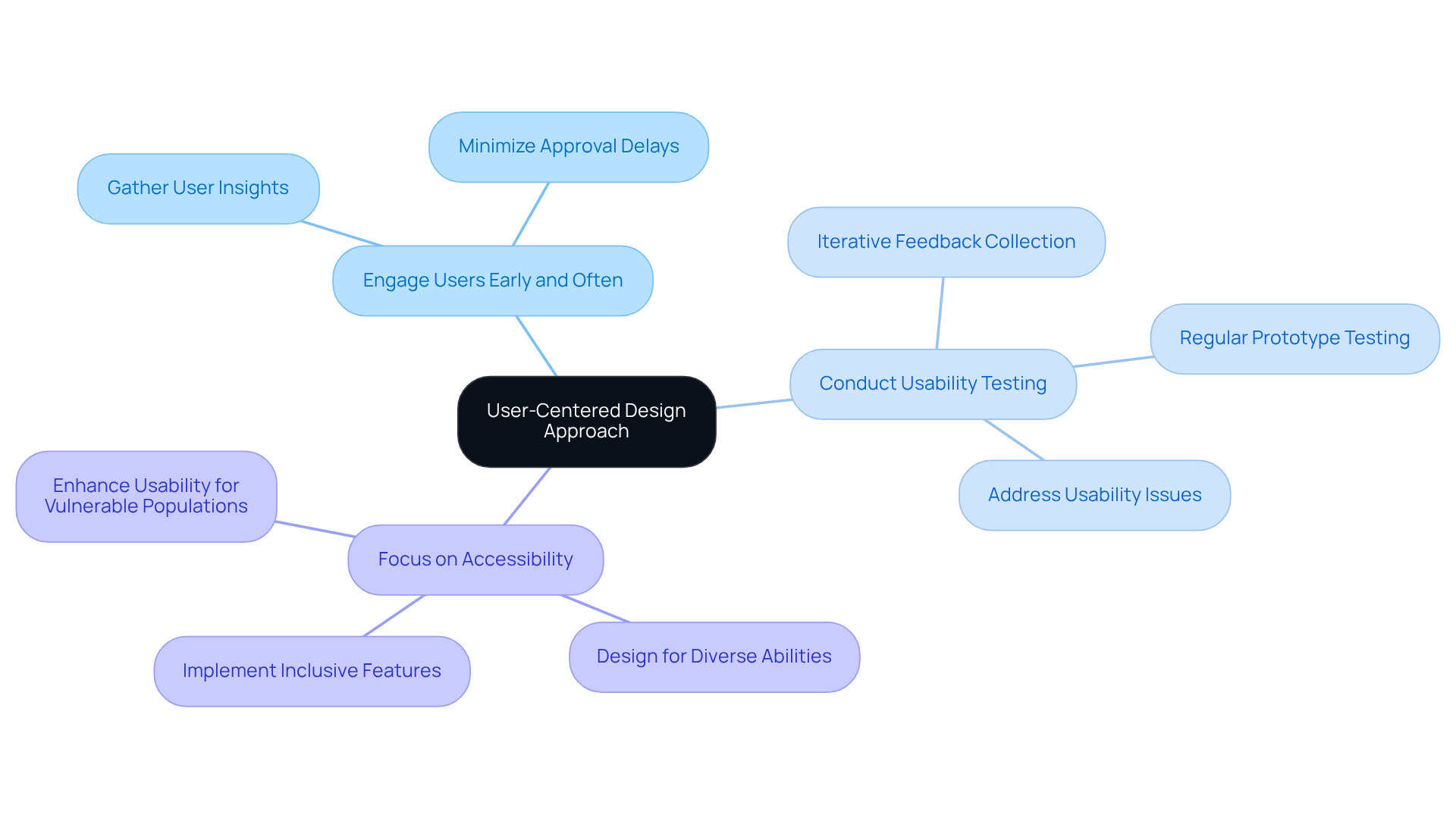
[[User-centered design](https://www.volersystems.com/blog/4-best-practices-for-effective-product-development-prototyping)](https://www.volersystems.com/blog/4-best-practices-for-effective-product-development-prototyping) (UCD) is a vital component in the development of effective healthcare tools, particularly in relation to [[Voler Systems](https://volersystems.com)](https://volersystems.com)' innovative approach to hardware creation and adherence. To implement UCD effectively, consider the following best practices:
- Engage Users Early and Often: Involve end-users in the design process from the beginning to gather insights into their needs, preferences, and limitations. Early engagement minimizes the risk of delays during approval and distribution, ensuring that the final product meets user expectations. This is especially critical for medical equipment, where compliance with regulatory standards is paramount.
- Conduct Usability Testing: Regularly test prototypes with actual users to identify usability issues and collect feedback for iterative improvements. Usability testing should occur early and consistently throughout the product development lifecycle, as this approach enhances device safety and effectiveness. For example, over one-third of 9,000 medication-related safety reports were associated with EHR usability issues, highlighting the necessity of thorough testing. Voler Systems underscores this in their prototype strategy, ensuring that software development aligns with user needs.
- Focus on Accessibility: Ensure that the design accommodates users with diverse abilities, thereby enhancing overall usability and adherence to accessibility standards. Features such as large displays, adjustable settings, and tactile feedback are essential for inclusivity, particularly for vulnerable populations like pediatric patients. Dr. Raj Ratwani emphasizes that inadequate interface design can lead to errors that may result in patient harm, underscoring the importance of accessible designs.
By prioritizing user needs throughout the design process, organizations can develop medical devices that are not only effective but also safe and user-friendly. The redesign of Vave’s Health wireless ultrasound platform, which successfully conducted over one million scans, exemplifies the practical benefits of applying UCD principles, serving as a testament to the successful collaboration and compliance strategies employed by Voler Systems.
Conclusion
Ensuring the effectiveness and safety of embedded system software in medical devices is essential for advancing healthcare technology. By integrating best practices such as:
- Regulatory compliance
- Effective development methodologies
- Comprehensive testing
- A user-centered design approach
organizations can significantly enhance the reliability and usability of their products. These strategies not only facilitate adherence to essential safety standards but also foster innovation within a highly regulated industry.
The article emphasizes several key practices, including:
- The necessity of understanding regulatory requirements like IEC 62304 and ISO 13485
- Adopting agile methodologies for flexibility
- Implementing robust testing protocols
- Prioritizing user engagement in the design process
Each of these elements plays a critical role in developing medical devices that satisfy both regulatory demands and user needs, ultimately leading to improved patient outcomes and a reduced risk of recalls.
In light of these insights, it is imperative for stakeholders in the medical device sector to prioritize these best practices in their development processes. By doing so, they not only ensure compliance with current regulations but also contribute to the creation of safer, more effective healthcare solutions. Embracing a proactive approach to software development in medical devices can pave the way for innovation and enhanced healthcare delivery, making it crucial for teams to remain informed and adaptable in this dynamic landscape.
Frequently Asked Questions
What are the key regulatory standards in the medical equipment sector?
The key regulatory standards include IEC 62304 and ISO 13485, which govern the software development lifecycle, risk management, design controls, and validation processes.
Why is it important to understand relevant regulations for medical devices?
Understanding relevant regulations, such as FDA guidelines in the U.S. and CE marking in Europe, is crucial for ensuring compliance and facilitating innovation in the medical equipment sector.
What is the role of a Quality Management System (QMS) in regulatory compliance?
A Quality Management System (QMS) aligned with ISO 13485 simplifies adherence to regulatory standards and enhances product quality, especially with the FDA's shift to the Quality Management System Regulation (QMSR).
How can teams stay updated on regulatory requirements?
Teams can stay updated by conducting regular training sessions to educate members on regulatory requirements and best practices, including changes to standards like IEC 62304.
What is the significance of integrating compliance into the creation process?
Integrating compliance from the beginning helps organizations reduce risks and improve the reliability of healthcare products, fulfilling regulatory requirements and supporting the long-term success of health technology innovations.
List of Sources
- Understand Regulatory Compliance and Safety Standards
- 2026 Commences with a Look Back, a Look Forward and New Legislation and Guidance (https://emergobyul.com/news/2026-commences-look-back-look-forward-and-new-legislation-and-guidance)
- IEC 62304 Update 2026: Key Changes & Compliance Tips (https://lfhregulatory.co.uk/iec-62304-update-2026)
- ISO 13485 and FDA Compliance Guide | NQA (https://nqa.com/en-us/resources/blog/february-2026/iso-13485-fda-compliance)
- FDA Implements Quality Management System Regulation for Medical Devices (https://aabb.org/news-resources/news/article/2026/02/04/fda-implements-quality-management-system-regulation-for-medical-devices)
- A QMSR State of Mind: FDA Adopts New Inspection Approach for Medical Devices as Quality Management System Regulation Takes Effect | Insights | Ropes & Gray LLP (https://ropesgray.com/en/insights/alerts/2026/02/a-qmsr-state-of-mind-fda-adopts-new-inspection-approach-for-medical-devices)
- Choose Effective Development Tools and Methodologies
- Built to comply: How AI is reshaping the medical device landscape (https://todaysmedicaldevelopments.com/news/built-comply-how-ai-reshaping-medical-device-landscape)
- Sprint to success: enhancing productivity with Agile methodologies in Med Tech (https://htdhealth.com/insights/agile-methodologies-in-med-tech)
- Agile-Based Medical Devices Design Market Research Report 2034 (https://dataintelo.com/report/global-agile-based-medical-devices-design-market)
- Applying FDA TIR45: Improving Medical Device Software Through Agile Risk Management (https://bermondseyelectronics.com/news/applying-fda-tir45-improving-medical-device-software-through-agile-risk-management)
- Implement Comprehensive Testing and Validation Protocols
- Oracle Health Launches Device Validation Program to Streamline Medical Device Integration and Accelerate Innovation in Healthcare (https://oracle.com/news/announcement/oracle-health-launches-device-validation-program-2026-02-25)
- 5 Staggering Medical Device Recall Statistics That Should Concern Everyone (https://qualio.com/blog/medical-device-recall-statistics)
- Oracle Health launches medical device validation program (https://healthcareitnews.com/news/oracle-health-launches-medical-device-validation-program)
- Manufacturers Should Establish a Validation Plan (https://mddionline.com/manufacturing/manufacturers-should-establish-a-validation-plan)
- FDA releases new guidance on software validation for medical devices | Marcelo Trevino posted on the topic | LinkedIn (https://linkedin.com/posts/marcelo-trevino-medicaldevices_computer-software-assurance-for-production-activity-7376400250091143168-acFI)
- Adopt a User-Centered Design Approach
- Developing Evidence-Based, User-Centered Design and Implementation Guidelines to Improve Health Information Technology Usability | Digital Healthcare Research (https://digital.ahrq.gov/ahrq-funded-projects/developing-evidence-based-user-centered-design-and-implementation-guidelines)
- Medical Device Design Tips for Usability (https://acenth.com/blog/design-tips-to-make-medical-devices-easier-to-use)
- Roger Mazzella's 2026 MedTech Predictions: Usability Drives Future Devices | Tomas Yates posted on the topic | LinkedIn (https://linkedin.com/posts/tomas-yates-4602bb75_medtech-industry-predictions-for-2026-activity-7409159731182776320-b8FO)
- User-Centered Design in HealthTech in 2026: How UX Drives Safety, Compliance, and Adoption (https://linkedin.com/pulse/user-centered-design-healthtech-2026-how-ux-drives-safety-compliance-aj4pf)





