Introduction
Prototyping is fundamental to the development of medical devices, acting as a crucial link between innovative concepts and their practical applications. This process allows teams to identify design flaws, gather user feedback, and ensure compliance with regulatory standards. Consequently, prototyping not only improves product quality but also fosters cost efficiency and drives innovation.
Nevertheless, the transition from concept to a market-ready prototype presents numerous challenges. How can developers effectively navigate this complex landscape to create devices that are both functional and safe for users?
Understand the Importance of Prototyping in Medical Device Development
Prototyping plays a crucial role in the development of medical devices, fulfilling several essential functions:
- Identifying Design Flaws: Early prototypes are vital for uncovering potential design and functionality issues. This allows teams to address problems before production begins, significantly reducing the risk of costly recalls and enhancing overall product quality.
- User Feedback: Prototypes facilitate user evaluations, enabling developers to gather valuable insights from healthcare professionals and patients. Such feedback is critical for guiding further design enhancements and ensuring that the product aligns with real-world requirements.
- Regulatory Compliance: Prototyping is integral to meeting stringent regulatory standards. It allows for thorough testing that ensures the equipment is safe and effective, which is essential for securing FDA approval and complying with other international regulations. Voler Systems' expertise in managing compliance from prototype to production ensures that wearable health products, such as heart pumps and liquid biopsy platforms, meet all necessary standards.
- Cost Efficiency: By identifying issues early in the development process, prototyping can significantly lower costs associated with late-stage changes and potential recalls. For example, virtual prototyping dramatically reduces both time and costs by eliminating the need for repeated physical iterations. This efficiency is crucial for maintaining budgetary constraints and ensuring timely market entry, particularly when leveraging Voler Systems' AI-assisted engineering capabilities.
- Innovation: Prototyping encourages a culture of innovation, prompting teams to experiment with various designs and functionalities. Recent advancements in prototyping techniques, including virtual prototyping and the integration of augmented reality and artificial intelligence, have enhanced the ability to simulate real-world performance and optimize designs. This iterative process not only fosters creativity but also leads to the development of more efficient and user-friendly healthcare tools, a hallmark of Voler Systems' approach to embedded systems design.
In conclusion, understanding the significance of prototyping is essential for a successful medical equipment development process. It ensures that the final product is effective, innovative, and compliant with industry standards, ultimately safeguarding patient safety.
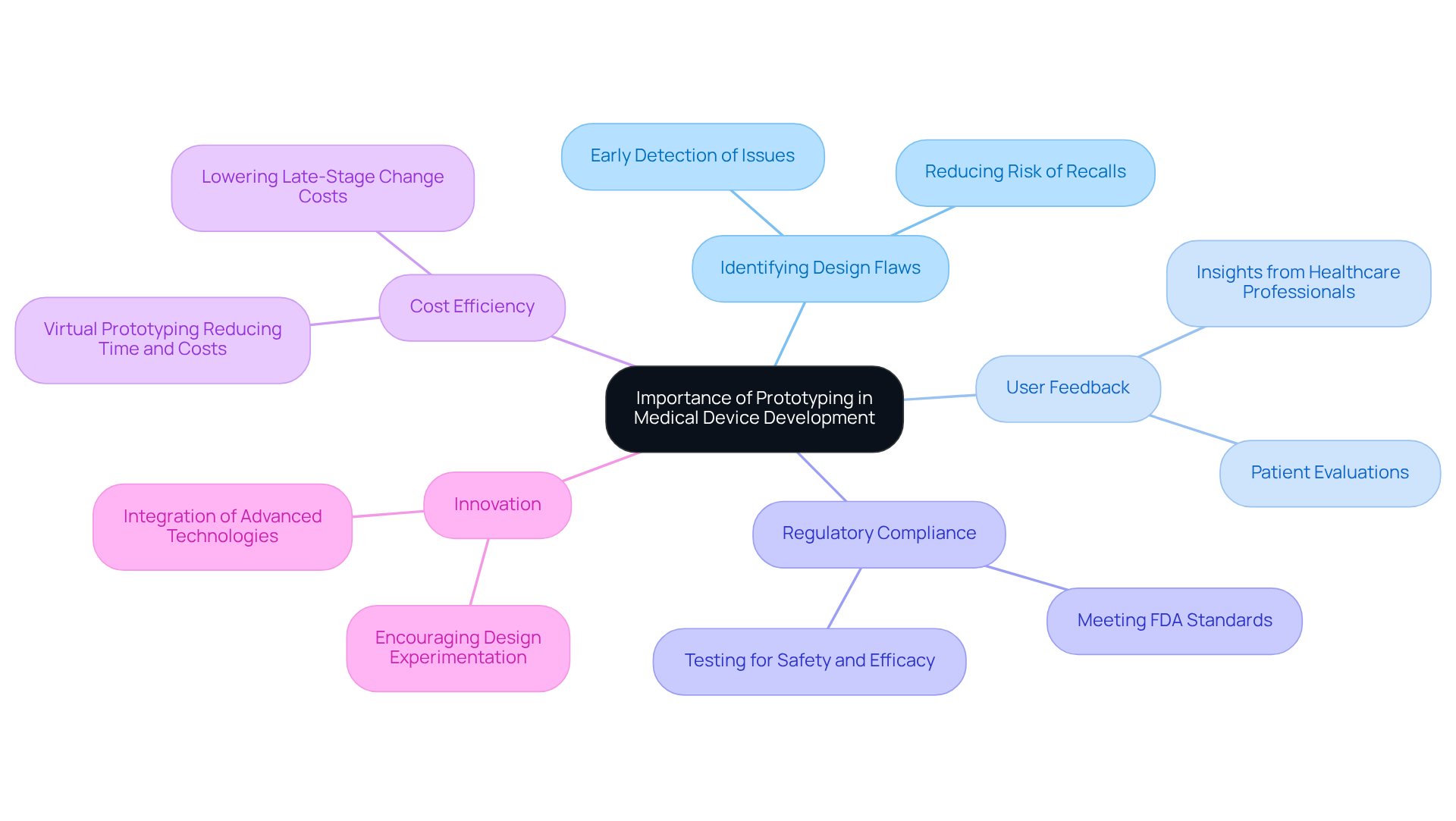
Follow Steps to Create an Effective Prototype
Creating an effective prototype involves several key steps:
- Define the Concept: Clearly articulate the issue your invention aims to solve. Outline the intended use, target users, and key functionalities. Engaging regulatory consultants early can streamline this phase, ensuring compliance with standards such as ISO 13485.
- Sketch Initial Designs: Create rough sketches or digital models of your creation. This visualization aids in refining the concept and serves as a reference for subsequent development stages.
- Select Materials: Choose materials based on the device's requirements, including biocompatibility, durability, and cost. Plastics are often favored for their lightweight and sterilizable properties, making them ideal for medical applications.
- Build the Model: Utilize techniques such as 3D printing, CNC machining, or traditional fabrication methods to create the model. Ensure that it closely resembles the final product in size and functionality, as this will facilitate more precise evaluation.
- Assemble Components: Integrate all necessary components, including sensors, circuits, and software. The assembly process should be efficient and replicable, which is crucial for scaling production.
- Initial Testing: Perform preliminary tests to evaluate the prototype's functionality, including stress tests, usability assessments, and performance evaluations to ensure the product meets user needs and regulatory standards. Prototyping assists in recognizing and addressing risks early through real-world evaluation and iteration.
- Document the Process: Maintain detailed records of the design and development process, including challenges faced and solutions implemented. This documentation is crucial for future iterations and regulatory submissions, as comprehensive documentation is a key requirement in healthcare product development.
By adhering to these steps, you can develop a prototype that accurately represents your medical apparatus concept and is prepared for additional evaluation and improvement. Engaging in iterative prototyping allows for gradual improvements, addressing technical complexities and enhancing the final product's reliability and compliance.
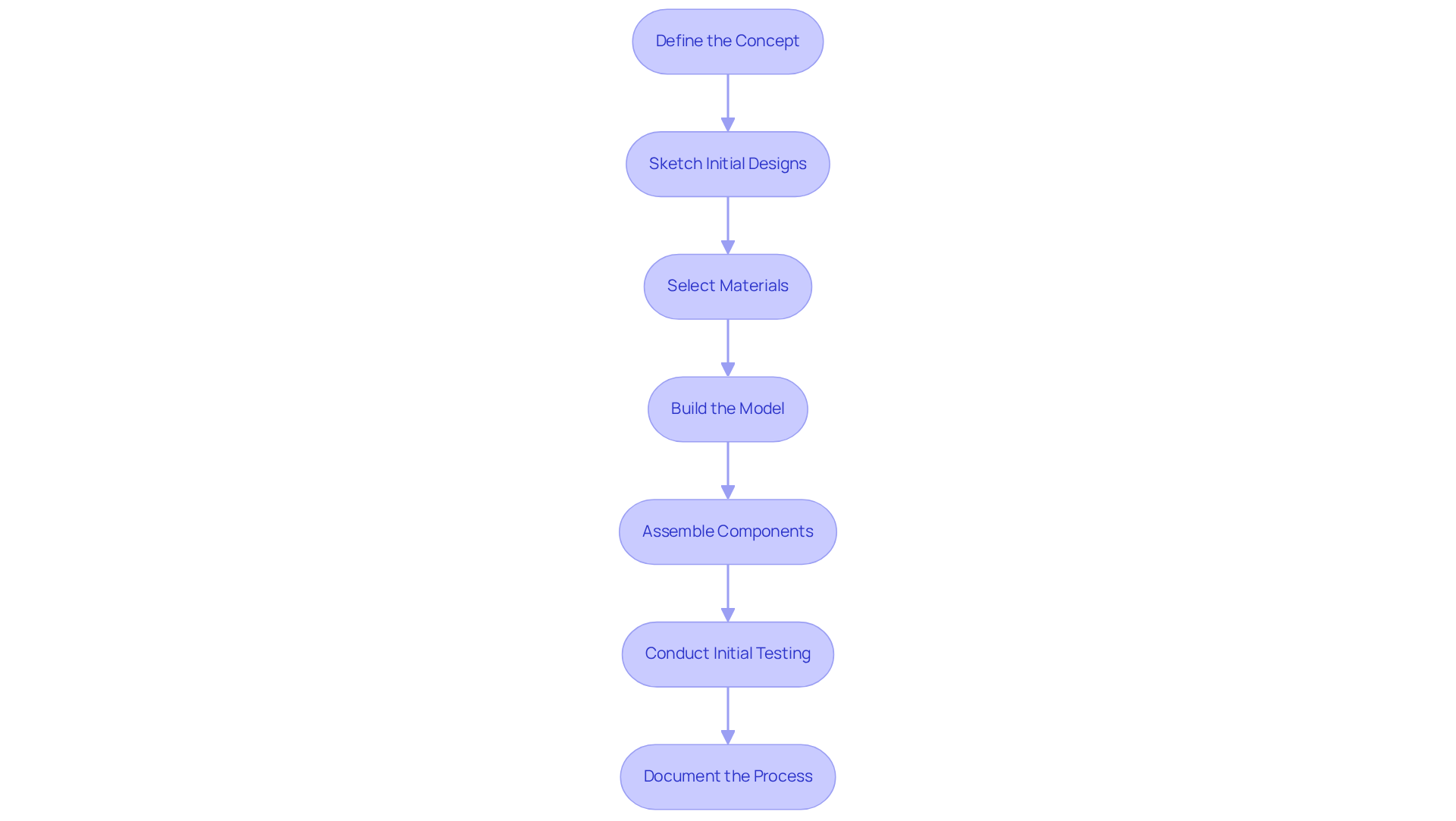
Conduct Thorough Testing to Validate Your Prototype
The phase to build and test prototype is critical in the prototyping process, ensuring that the device meets all necessary standards and functions as intended. Key testing methods to consider include:
- Functional Testing: This method assesses whether the prototype performs its intended functions under various conditions. It encompasses testing all features and functionalities to confirm they operate as designed.
- Usability Testing: Gathering feedback from potential users is essential for evaluating the device's ease of use. Observing users as they interact with the prototype allows for the collection of insights on the user interface and overall experience. Research indicates that a sample size of just 15 can uncover 90% to 97% of usability issues, highlighting the effectiveness of this testing method.
- Safety Testing: Conducting tests to ensure the equipment is safe for use is vital. This includes evaluating potential hazards such as electrical safety, mechanical safety, and biocompatibility, which are critical for patient safety and regulatory compliance.
- Regulatory Compliance Testing: Ensuring that the model meets all relevant regulatory standards, such as ISO and FDA requirements, is essential. This may involve specific tests for medical equipment, including performance and reliability evaluations, to demonstrate compliance with safety and effectiveness standards mandated by regulatory bodies.
- Iterative Evaluation: An iterative approach to assessment, where feedback from each round informs the next prototype iteration, is beneficial. This process aids in improving the design and addressing any problems that emerge, ultimately resulting in a more user-friendly and efficient product.
To avoid common errors in manufacturing assessments, manufacturers should implement best practices such as thorough documentation, regular training for evaluation personnel, and utilizing standardized assessment protocols.
Comprehensive evaluation not only allows us to build and test prototype but also fosters trust in the product's safety and efficacy, which is crucial for regulatory approval and market success. By prioritizing usability evaluations, manufacturers can significantly enhance patient safety and reduce the likelihood of costly recalls, making it a critical component of medical device development.
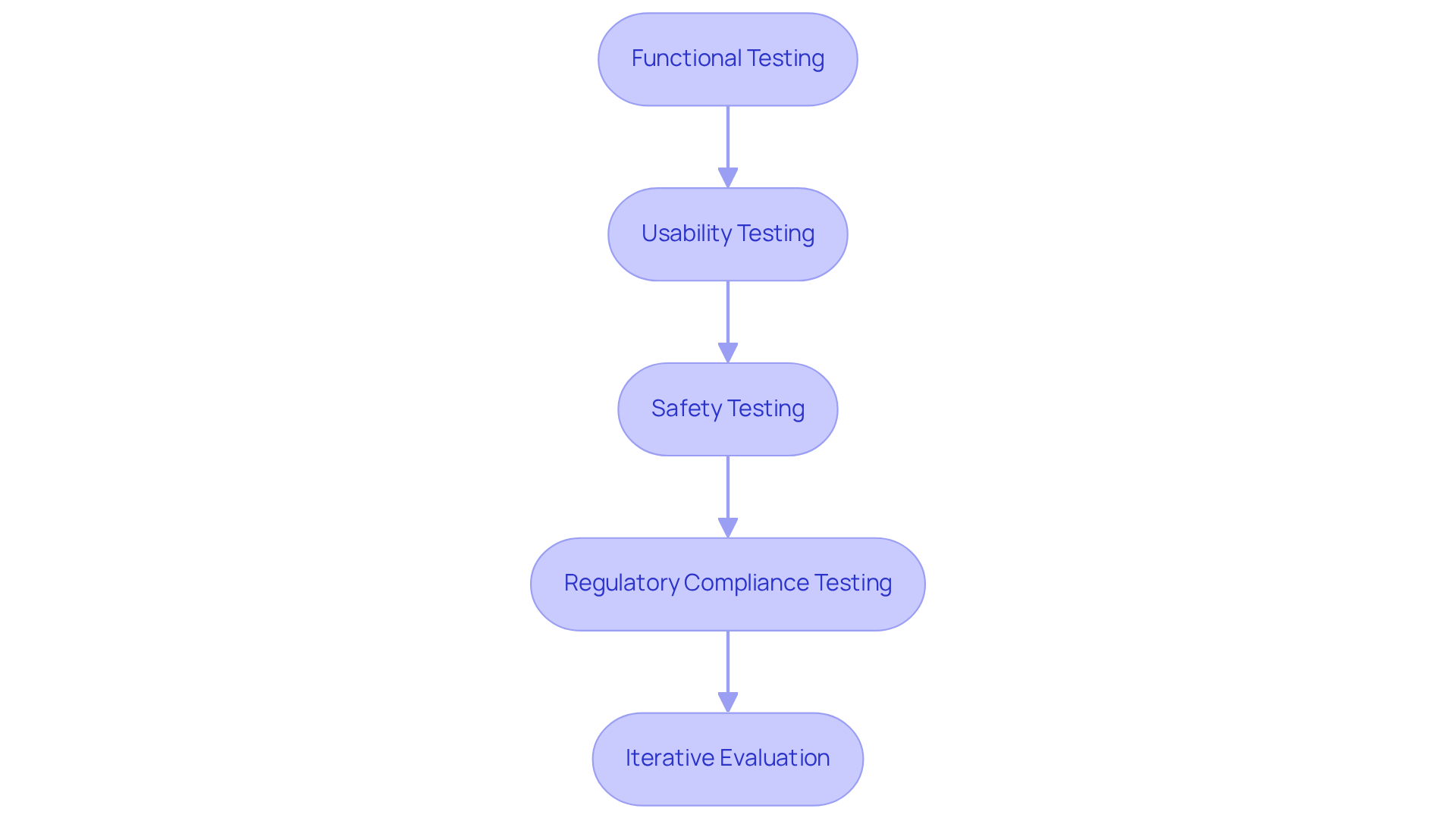
Iterate and Improve Based on Testing Feedback
Iteration is a crucial element in the prototyping process, as it enables teams to build and test prototype versions for continuous improvement through user feedback and evaluation results. Below is a structured approach to effectively iterate and refine your prototype:
- Analyze Feedback: After testing, systematically gather and analyze feedback from users and stakeholders. Identify recurring themes and issues that require attention.
- Prioritize Changes: Based on the feedback, determine which modifications are most critical to the product's success. Focus on aspects that impact safety, usability, and functionality, as these are paramount in the medical device sector.
- Make Adjustments: Implement the necessary modifications to the model. This may involve redesigning components, altering software, or selecting different materials to enhance performance.
- Retest the Prototype: Following adjustments, conduct another round of evaluation to assess the effectiveness of the changes. This step is vital to ensure that we can build and test prototype modifications that have adequately addressed the identified issues.
- Document Changes: Maintain comprehensive records of all changes made during the iteration process. This documentation is essential for regulatory submissions and future development phases, ensuring compliance with industry standards.
- Repeat the Process: Continue the cycle of testing and iteration until the prototype meets all design specifications and user requirements. This iterative approach not only enhances the product's effectiveness but also involves the process to build and test prototype, ensuring compliance with stringent regulatory standards.
By adopting an iterative mindset, developers can create medical devices that are innovative, reliable, and user-friendly, ultimately leading to improved product quality and safety.
Conclusion
Prototyping is a crucial element in the development of medical devices, acting as a foundational step that guarantees products are both innovative and safe. Engaging in the prototyping process allows developers to identify design flaws early, gather vital user feedback, and adhere to stringent regulatory standards. This systematic approach ultimately results in higher quality medical devices that effectively meet the needs of healthcare professionals and patients.
The article highlights the multifaceted benefits of prototyping, such as:
- Cost efficiency
- The encouragement of innovation
- The necessity of iterative testing
Each phase of the prototyping journey-from concept definition to comprehensive testing and iterative enhancements-plays a pivotal role in refining the final product. By following established practices and consistently seeking user input, developers can significantly improve the usability and performance of their medical devices.
As the healthcare landscape evolves, the importance of effective prototyping remains paramount. Adopting a robust prototyping strategy not only promotes innovation but also ensures patient safety and compliance with industry regulations. For those engaged in medical device development, prioritizing prototyping is essential for achieving success in a competitive market and ultimately enhancing patient outcomes.
Frequently Asked Questions
What is the role of prototyping in medical device development?
Prototyping plays a crucial role in identifying design flaws, gathering user feedback, ensuring regulatory compliance, promoting cost efficiency, and fostering innovation in medical device development.
How does prototyping help identify design flaws?
Early prototypes are vital for uncovering potential design and functionality issues, allowing teams to address problems before production begins, which reduces the risk of costly recalls and enhances product quality.
Why is user feedback important in the prototyping process?
Prototypes facilitate user evaluations, enabling developers to gather valuable insights from healthcare professionals and patients, which is critical for guiding design enhancements and ensuring alignment with real-world requirements.
How does prototyping relate to regulatory compliance?
Prototyping is integral to meeting stringent regulatory standards by allowing thorough testing to ensure that medical equipment is safe and effective, which is essential for securing FDA approval and complying with international regulations.
In what ways does prototyping contribute to cost efficiency?
Prototyping identifies issues early in the development process, significantly lowering costs associated with late-stage changes and potential recalls. Virtual prototyping, in particular, reduces both time and costs by eliminating the need for repeated physical iterations.
How does prototyping encourage innovation in medical device development?
Prototyping fosters a culture of innovation by prompting teams to experiment with various designs and functionalities. Recent advancements in techniques, such as virtual prototyping and the use of augmented reality and artificial intelligence, enhance the ability to simulate real-world performance and optimize designs.
What is the overall significance of understanding prototyping in medical device development?
Understanding the significance of prototyping is essential for ensuring that the final product is effective, innovative, and compliant with industry standards, ultimately safeguarding patient safety.
List of Sources
- Understand the Importance of Prototyping in Medical Device Development
- The Importance of Prototyping for Medical Devices - BDK (https://bdk.uk.com/2025/01/the-importance-of-prototyping-for-medical-devices)
- 11 food products which you can eat in a completely different way (https://flavoursofmekong.com.au/virtual-prototyping-accelerates-medical-device-development-2)
- Medical Device Prototyping In Accelerating Time To Market (https://europlaz.co.uk/medical-device-prototyping-in-accelerating-time-to-market)
- The Role of Rapid Prototyping in Fast-Tracking New Medical Technologies (https://gcmiatl.org/the-role-of-rapid-prototyping-in-fast-tracking-new-medical-technologies)
- Virtual Prototyping Accelerates Medical Device Development (https://quietude.com.au/virtual-prototyping-accelerates-medical-device-development-2?srsltid=AfmBOoqZACAGVyHbi-j3Rew2ATxny6UU4UzPKZRcGrw1fY5tIA3x7ojl)
- Follow Steps to Create an Effective Prototype
- Medical Device Prototyping: A Critical Step For Product Launch (https://opinc.com/medical-device-prototyping-a-critical-step-for-product-launch)
- Medical Device Prototyping: Stages, Process, Testing (https://arterexmedical.com/medical-device-prototyping)
- The four stages of medical device prototyping: who can help and how? (https://escatec.com/blog/the-four-stages-of-medical-device-prototyping)
- Best Practices for Developing Medical Devices: Overcoming Challenges (https://volersystems.com/blog/best-practices-for-developing-medical-devices-overcoming-challenges)
- Conduct Thorough Testing to Validate Your Prototype
- Key Benefits of Usability Testing in Medical Devices (https://emergobyul.com/resources/importance-usability-testing-medical-devices)
- Medical Device Usability Testing (https://exponent.com/capabilities/medical-device-usability-testing)
- Usability Testing for Medical Devices: Key Necessity - Freyr (https://freyrsolutions.com/blog/usability-testing-for-medical-devices-critical-necessity-or-mere-formality)
- Comprehensive Guide to Usability Testing for Medical Devices - MakroCare (https://makrocare.com/blog/usability-testing-in-medical-devices-a-comprehensive-guide)
- Medical Device Testing in Transition (https://mpo-mag.com/medical-device-testing-in-transition)
- Iterate and Improve Based on Testing Feedback
- Prototyping Insights from Initial Concept to Final Medical Device (https://aztechnica.com/resources/f/prototyping-insights-from-initial-concept-to-final-medical-device)
- Using Statistical Science to Optimize Quality across the Medical Device Life Cycle (https://exponent.com/article/using-statistical-science-optimize-quality-across-medical-device-life-cycle)
- Medical Device Prototyping In Accelerating Time To Market (https://europlaz.co.uk/medical-device-prototyping-in-accelerating-time-to-market)
- Usability Works at Carilion Clinic supports medical device manufacturers who need feedback on prototypes before they go to market (https://wfirnews.com/news/usability-works-at-carilion-clinic-supports-medical-device-manufacturers-who-need-feedback-on-prototypes-before-they-go-to-market)





