4 Steps to Develop Electronic Products for Medical Devices
Introduction The development of electronic products for medical devices presents...

Navigating the complex landscape of medical device development necessitates a strategic approach, with the Stage-Gate Process emerging as a vital framework. This structured methodology clearly outlines the journey from ideation to market launch, ensuring that each phase adheres to regulatory standards and meets market demands. As manufacturers pursue this path, they frequently face the challenge of balancing innovation with compliance. What essential steps and considerations can streamline this process and improve the likelihood of success?
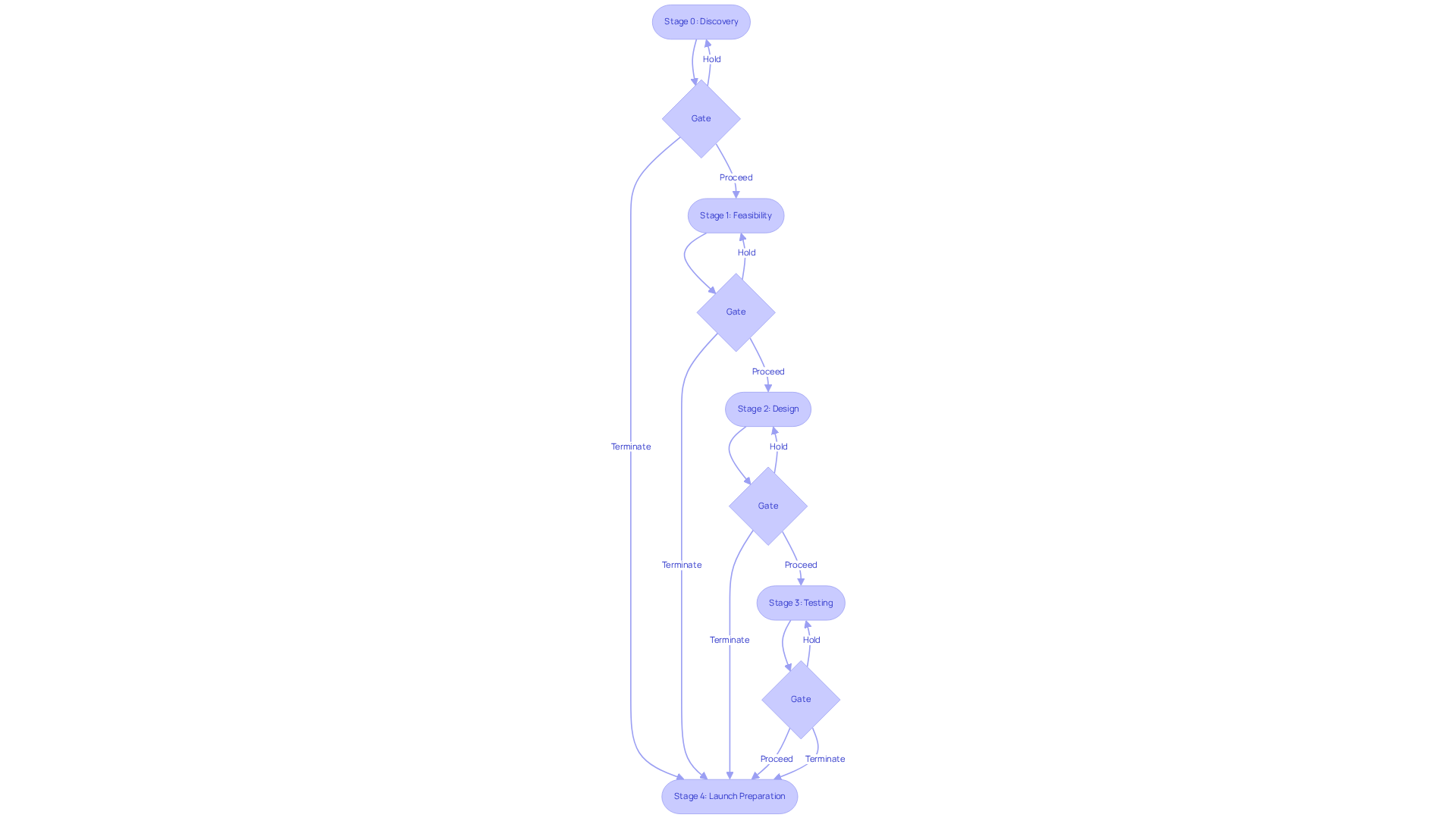
The Stage-Gate Process, also known as the Phase-Gate Process, is a structured project management methodology that divides the development of medical devices into distinct stages, each separated by critical decision points referred to as gates. This framework is vital for ensuring systematic progress and adherence to industry standards.
Stages: Each stage corresponds to a specific phase of development, which includes:
Gates: At each gate, a cross-functional team evaluates the project against established criteria, including market potential, technical feasibility, and regulatory compliance. The outcomes of these evaluations may include:
Documentation: Comprehensive documentation is required at each stage to support informed decision-making at the gates. This includes risk assessments, design specifications, and validation reports, which are crucial for maintaining compliance and ensuring project success.
Understanding this framework is essential for manufacturers aiming to while adhering to stringent industry regulations.
The Development Gate represents a pivotal stage in the creation of your healthcare apparatus. Here’s how to navigate this critical phase effectively:
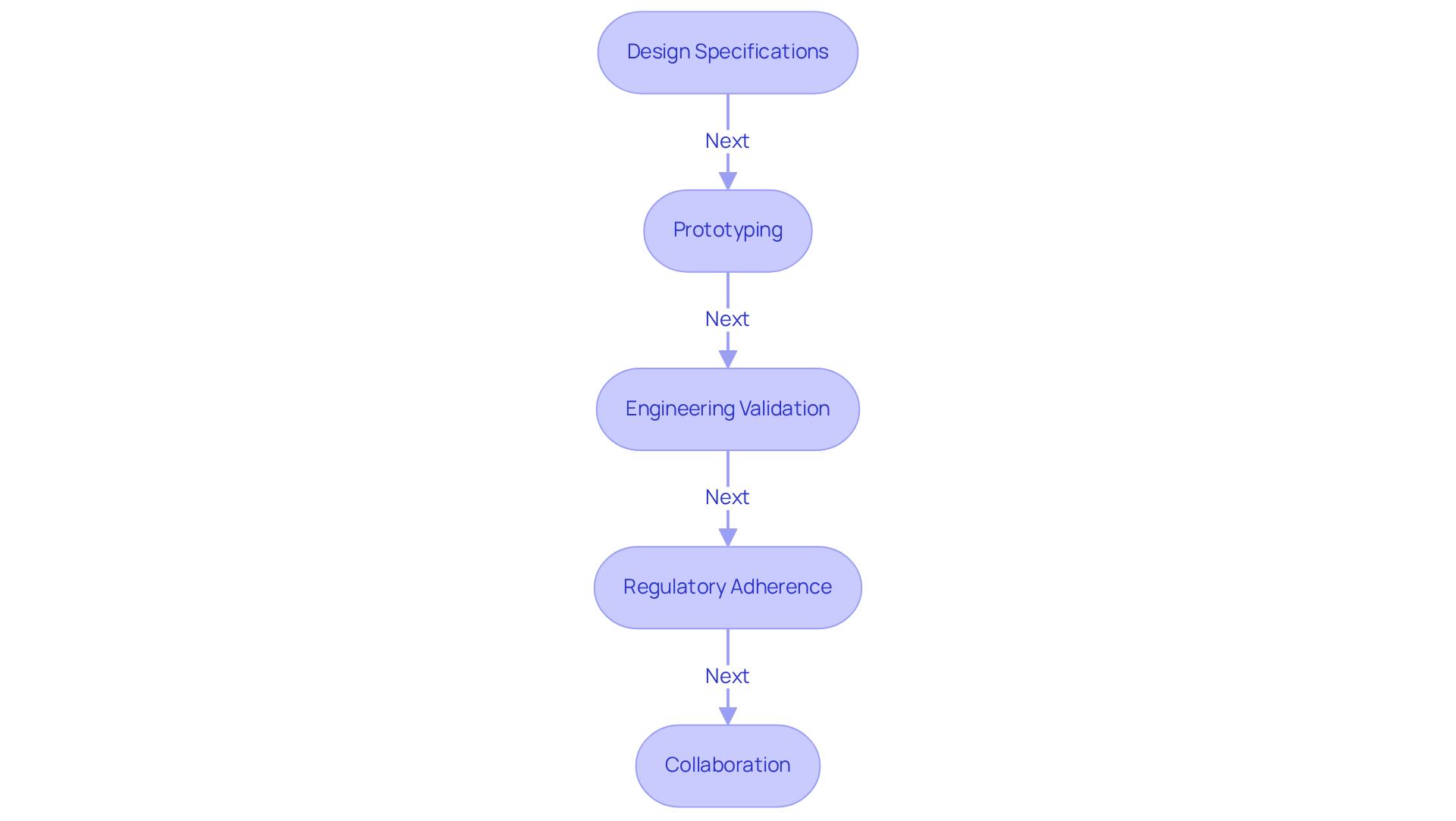
Design Specifications: Finalize your design specifications based on insights from earlier feasibility studies. Document all requirements meticulously to ensure compliance with regulatory standards. Skilled engineers at Voler Systems can assist in creating high-quality health products, including wearable technology, heart pumps, and liquid biopsy platforms, that meet these specifications within budget and on schedule.
Prototyping: Develop prototypes of your device using advanced techniques:
Engineering Validation: Validate the engineering aspects of your device through:
Regulatory Adherence: Maintain a strong focus on adherence to regulations throughout the development process. This includes:
Collaboration: Promote teamwork among cross-functional groups, including engineering, quality assurance, and compliance affairs. This guarantees that all elements of the apparatus align with project objectives and consumer requirements.
By following these steps, you can efficiently create a healthcare instrument that not only satisfies consumer needs but also adheres to strict regulatory standards, ultimately improving patient care and safety. Furthermore, the global rapid prototyping healthcare tools market is anticipated to reach $2.5 billion by 2025, with a CAGR of 5% from 2025 to 2033, underscoring the significance of these techniques in the development process.
Testing and validation are critical components of the , particularly for startups aiming to ensure compliance and reliability. To implement effective strategies, consider the following steps:
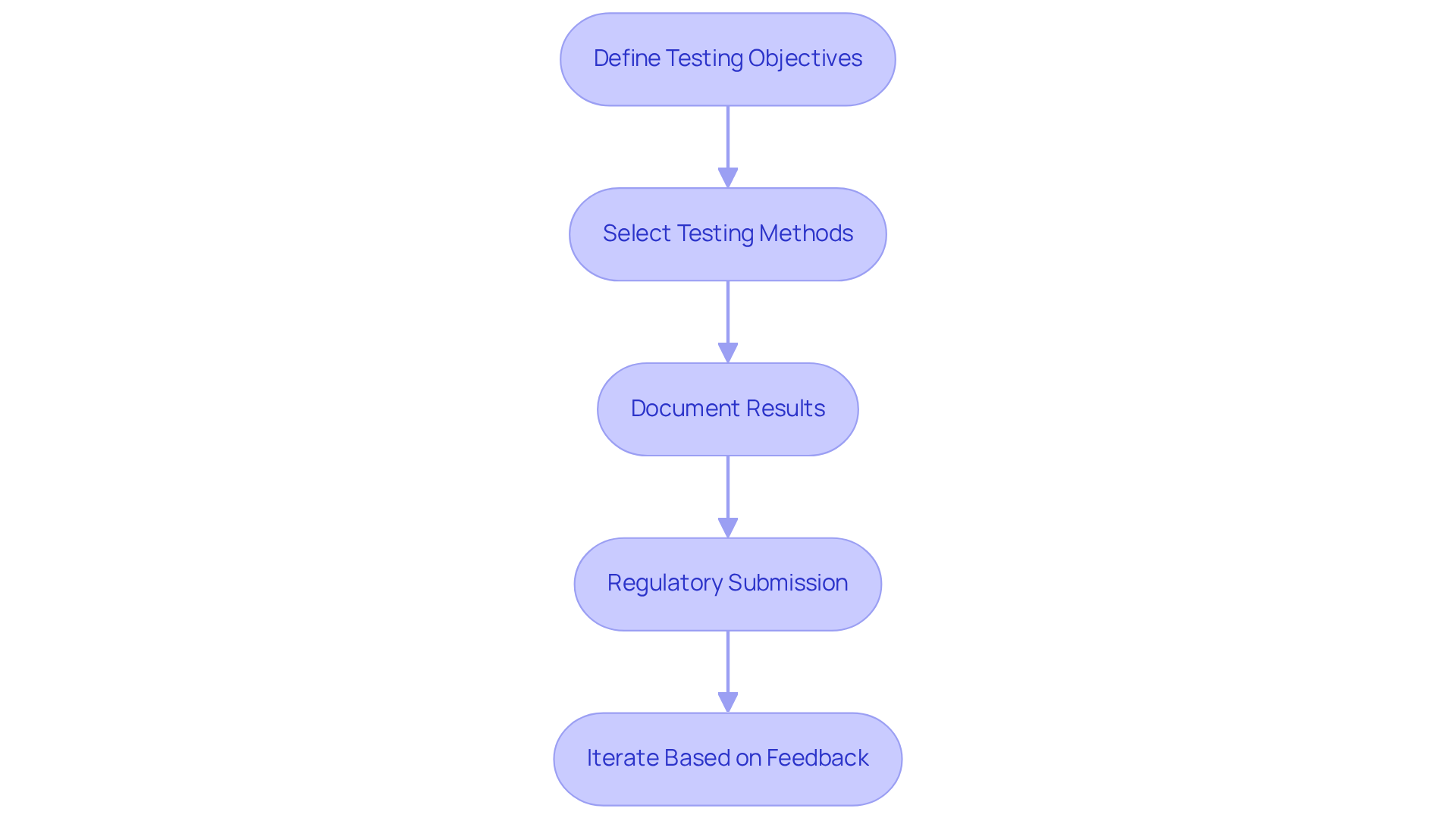
Define Testing Objectives: Clearly outline your testing needs, focusing on:
Select Testing Methods: Choose appropriate methods based on your objectives. Common approaches include:
Document Results: Maintain comprehensive documentation of all testing activities, including:
Regulatory Submission: Prepare your testing documentation for regulatory submission, which includes:
Iterate Based on Feedback: Utilize feedback from testing to make necessary design adjustments. This iterative process is vital for enhancing the apparatus and ensuring it meets all safety and efficacy standards, ultimately leading to successful clinical trials and readiness for market entry.
By applying these testing and validation approaches, you can enhance the likelihood of your healthcare product obtaining regulatory approval and successfully entering the industry, leveraging [Voler Systems](https://volersystems.com)' expertise in compliant and reliable electronic design.
The Launch Phase is critical for effectively presenting your medical device to consumers. To execute this phase successfully, consider the following steps:
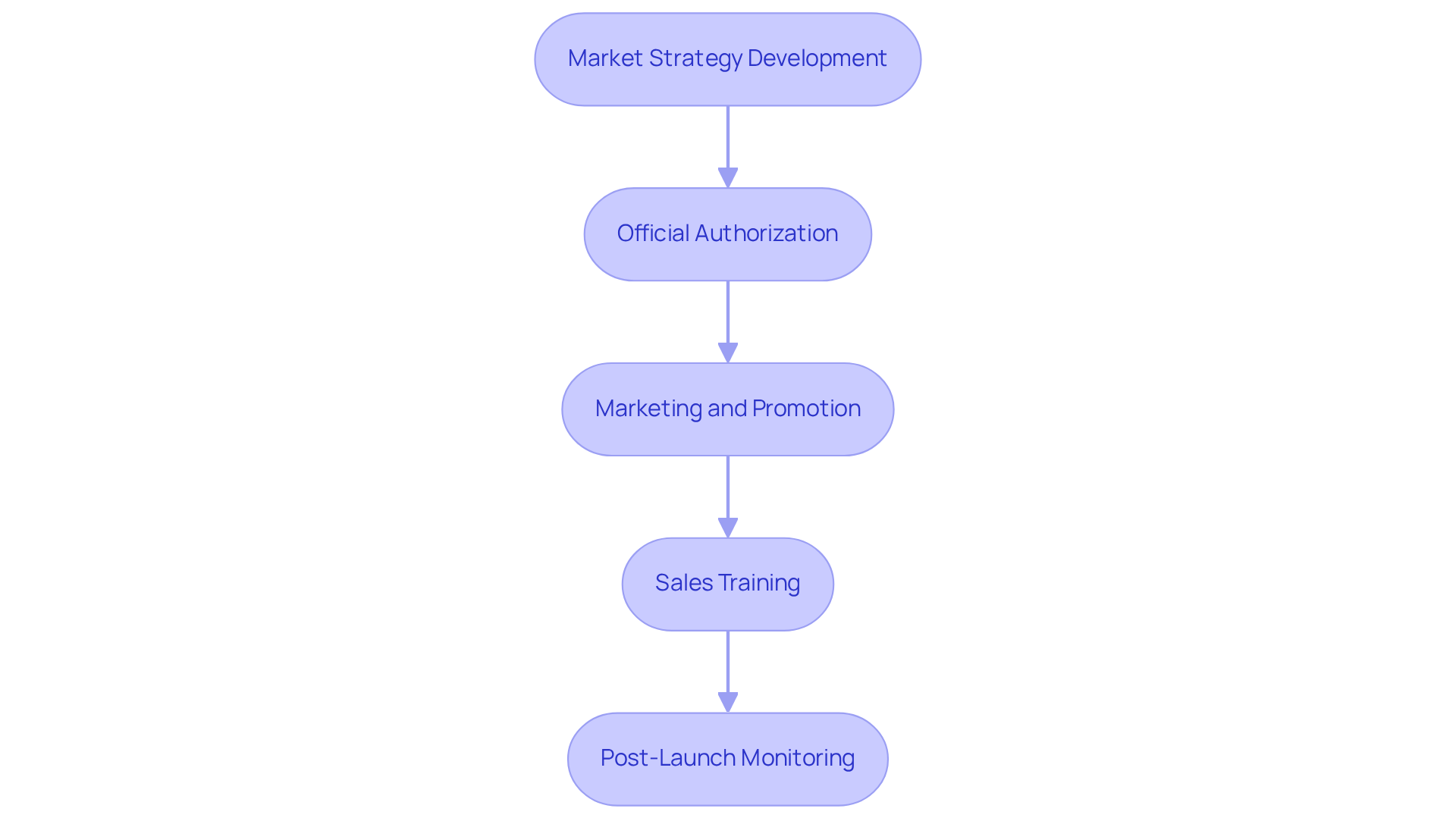
Market Strategy Development: Formulate a comprehensive market strategy that includes:
Official Authorization: Prioritize obtaining all necessary official approvals prior to the launch. This involves:
Marketing and Promotion: Develop a robust marketing plan that outlines your promotional strategies. Consider:
Sales Training: Equip your sales team with comprehensive training on the product's features and benefits. Ensure they are prepared to:
Post-Launch Monitoring: After the release, it is essential to observe the product's performance in the industry. This includes:
By meticulously following these steps, you can navigate the complexities of bringing your medical device to market, ensuring its long-term success and compliance with industry standards.
The journey of developing a medical device is intricate and multifaceted, necessitating a structured approach for successful navigation. The Stage-Gate Process serves as a crucial framework, segmenting development into manageable stages and gates that ensure comprehensive evaluation and compliance with industry standards. By adopting this methodology, manufacturers can significantly improve their chances of delivering innovative healthcare solutions that effectively address the needs of both patients and healthcare providers.
Throughout this article, the significance of each phase - from initial discovery to market launch - has been underscored. Essential steps such as:
are vital for the successful development of medical devices. Each of these components contributes to a streamlined process that not only fosters innovation but also prioritizes safety and efficacy.
In conclusion, the importance of a structured approach in medical device development cannot be overstated. By implementing the Stage-Gate Process and concentrating on meticulous planning, testing, and regulatory adherence, developers can not only meet compliance standards but also enhance the overall quality of healthcare products. As the medical technology landscape continues to evolve, leveraging these strategies will be essential for maintaining competitiveness and ensuring that new devices effectively tackle the challenges encountered in patient care.
What is the Stage-Gate Process Framework?
The Stage-Gate Process Framework, also known as the Phase-Gate Process, is a structured project management methodology that divides the development of medical devices into distinct stages, each separated by critical decision points called gates.
What are the stages involved in the Stage-Gate Process?
The stages include: - Stage 0: Discovery and opportunity identification, focusing on ideation and initial research. - Stage 1: Feasibility and concept development, where project requirements and stakeholder needs are defined. - Stage 2: Design and development, involving the creation of prototypes and initial designs. - Stage 3: Testing and validation, ensuring the product meets compliance standards and user needs. - Stage 4: Launch preparation, finalizing all aspects for introduction to consumers.
What happens at each gate in the Stage-Gate Process?
At each gate, a cross-functional team evaluates the project against established criteria, including market potential, technical feasibility, and regulatory compliance. Possible outcomes include proceeding to the next stage, holding the project for additional information or adjustments, or terminating the project if it fails to meet necessary standards.
What type of documentation is required in the Stage-Gate Process?
Comprehensive documentation is required at each stage, including risk assessments, design specifications, and validation reports. This documentation supports informed decision-making at the gates and is crucial for maintaining compliance and ensuring project success.
Why is the Stage-Gate Process important for manufacturers?
Understanding the Stage-Gate Process is essential for manufacturers to navigate the complexities of product development while adhering to stringent industry regulations.
