4 Best Practices for Embedded System Software in Medical Devices
Introduction In the intricate realm of medical device development, the integration of...

Effective product design in the medical device industry is essential, as it directly influences patient safety and healthcare outcomes. By adhering to established best practices, teams can improve collaboration, optimize processes, and ultimately deliver innovative solutions to market more efficiently. However, the challenge lies in navigating the complexities of communication, regulatory compliance, and user feedback throughout the design process.
How can teams ensure they not only meet industry standards but also address the genuine needs of end-users?
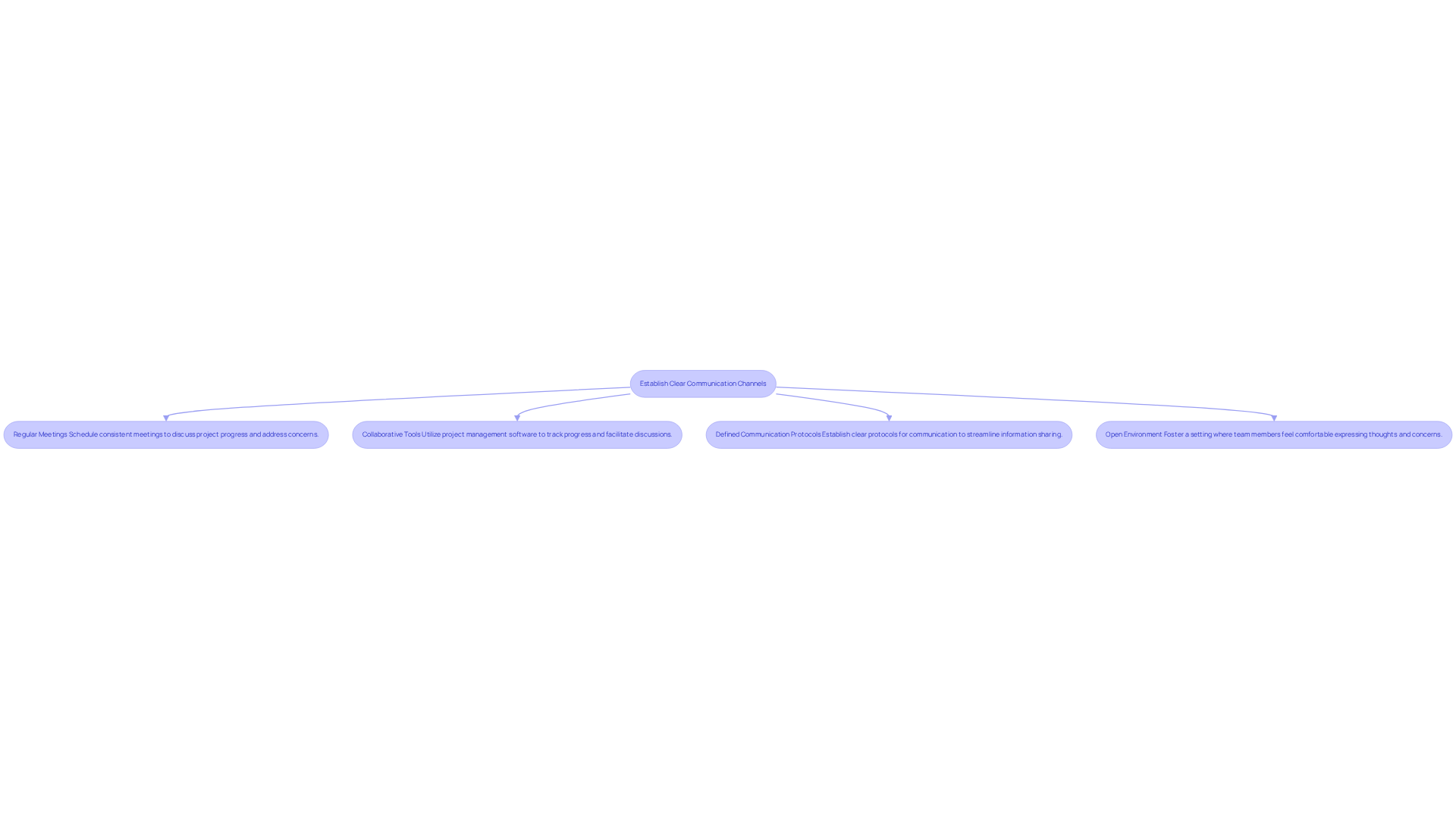
To ensure in medical devices, establishing clear communication channels among all team members - engineers, designers, regulatory experts, and marketing personnel - is essential. The following key steps can implement effective communication practices:
A study by CRICO Strategies reveals that over 7,000 out of 23,000 malpractice claims stemmed from communication failures, costing $1.7 billion and resulting in nearly 2,000 avoidable deaths. As noted by Steve Vlok, 'Standard healthcare communication platforms are often outdated and have limited capabilities, leading to low levels of quality patient care.' By adopting these effective communication methods, teams at [Voler Systems](https://volersystems.com) can address challenges more efficiently and enhance overall outcomes, ultimately facilitating the successful transfer of innovative healthcare technology to manufacturing through a [product design service](https://www.volersystems.com/blog/5-best-practices-for-prototype-3d-printing-in-medical-devices).
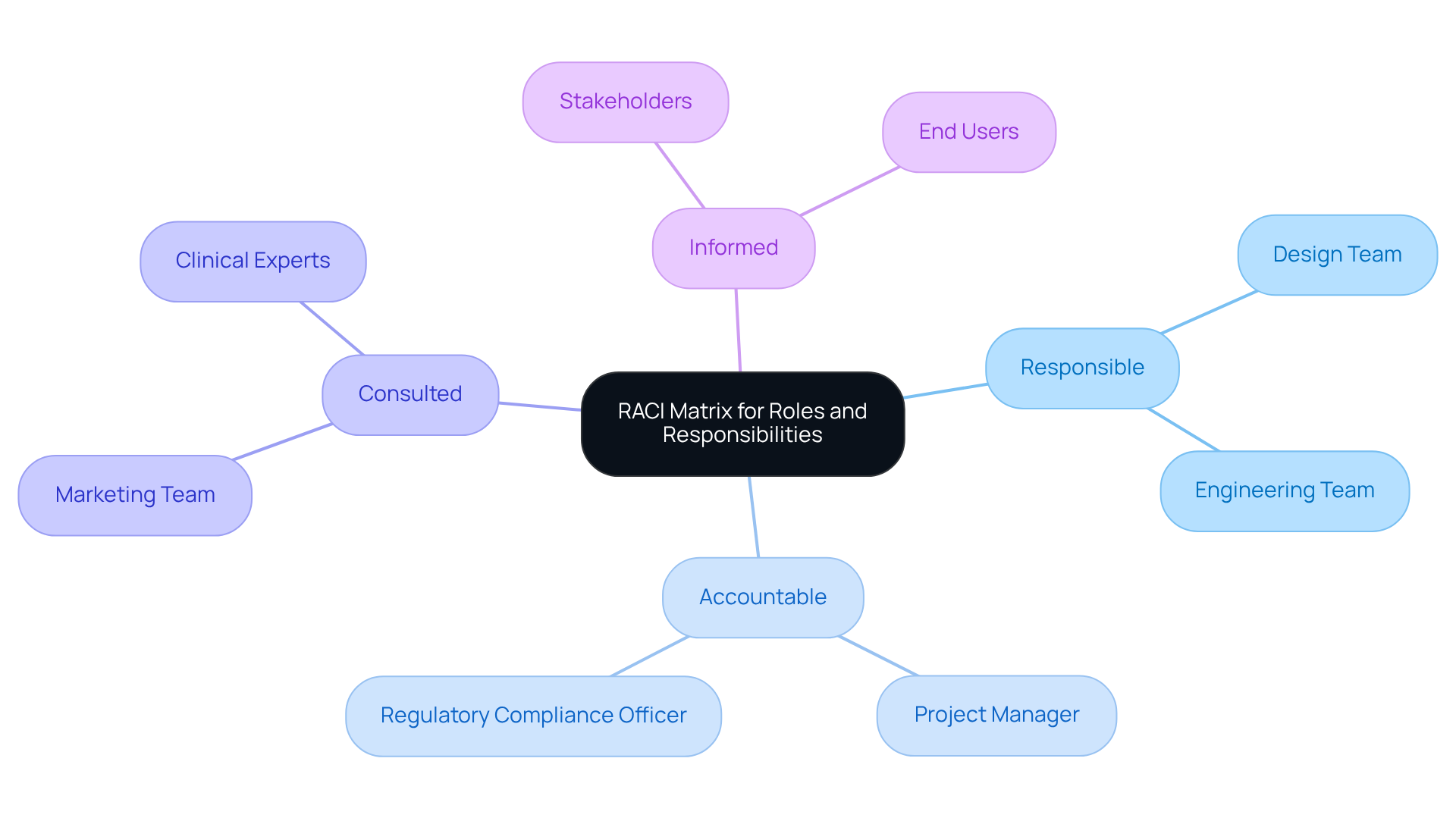
Clarifying roles and responsibilities within the group is essential for promoting effective collaboration in medical device development. Each group member must understand their specific contributions, which encompass design, engineering, regulatory compliance, and marketing.
Implementing a [RACI (Responsible, Accountable, Consulted, Informed) matrix](https://meegle.com/en_us/topics/raci-matrix/raci-matrix-case-studies) serves as an effective strategy to delineate these roles. This matrix specifies:
By establishing this clear structure, groups can significantly reduce confusion, enhance communication, and ensure that all elements are managed efficiently.
For instance, hospitals have successfully employed the RACI matrix to streamline processes, such as patient discharge, resulting in improved satisfaction and reduced delays. Regular updates to the matrix are crucial to reflect changes in scope or personnel structure, ensuring its ongoing relevance and effectiveness in fostering success.
These practices contribute to timely and cost-effective delivery, a hallmark of highly successful engineering design endeavors.
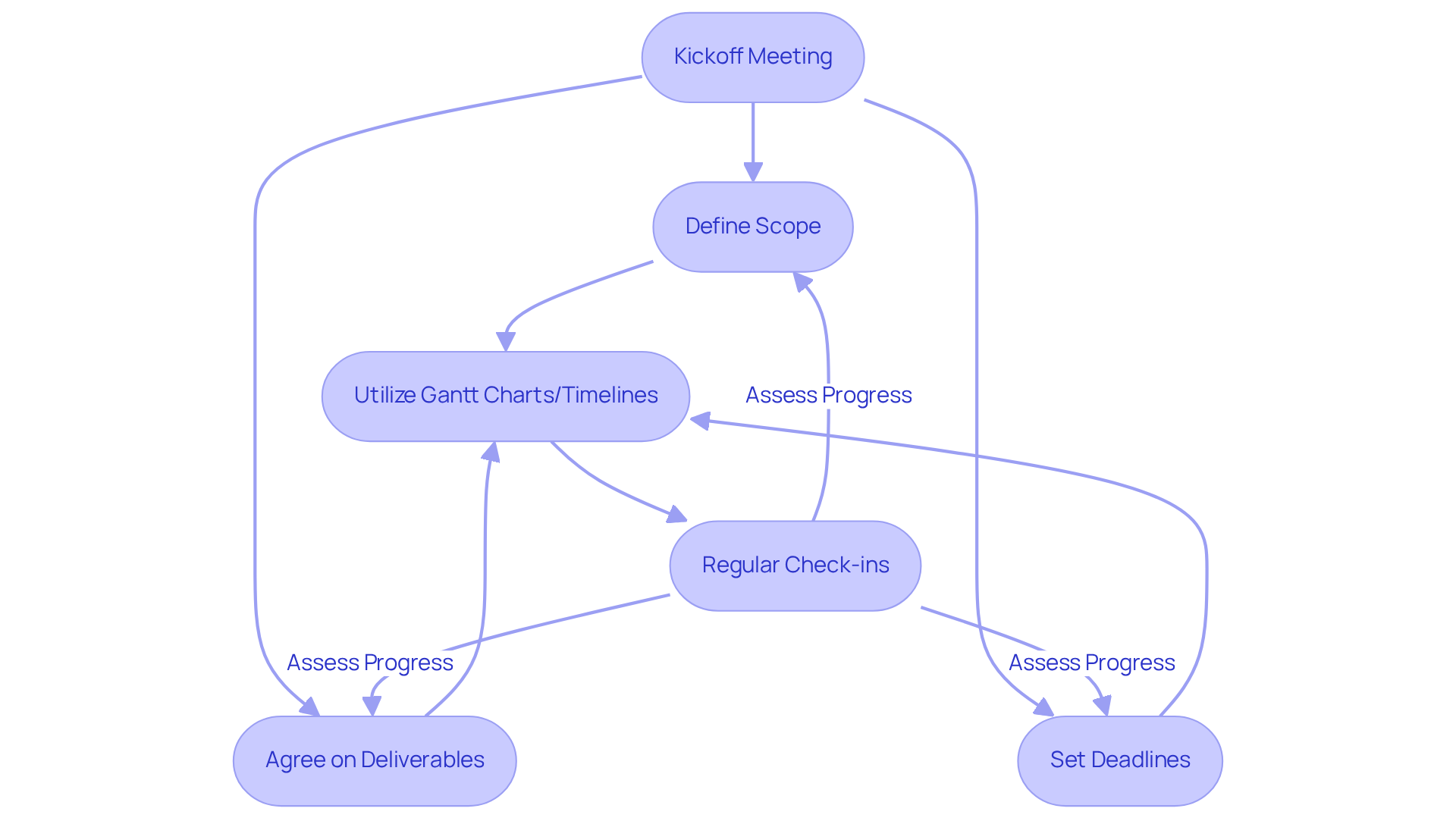
Aligning on goals and timelines is essential for ensuring that all team members work towards the same objectives. This alignment can be effectively established through [kickoff meetings](https://www.volersystems.com/blog/5-best-practices-for-selecting-embedded-software-products-in-medical-devices), where the scope, deliverables, and deadlines are clearly articulated and agreed upon. Utilizing tools such as or timelines provides a visual representation of the workflow, thereby promoting accountability among team members.
Regular check-ins are crucial for assessing progress against these established objectives, allowing for timely adjustments to keep the initiative on track. This organized approach not only enhances communication but also significantly impacts the outcomes of healthcare devices, ensuring that the [product design service](https://www.volersystems.com/blog/4-best-practices-for-machine-to-machine-communication-in-medical-devices) leads to projects being completed efficiently and in compliance with [industry standards](https://inloox.com/company/blog/articles/the-62-most-inspiring-project-management-quotes).
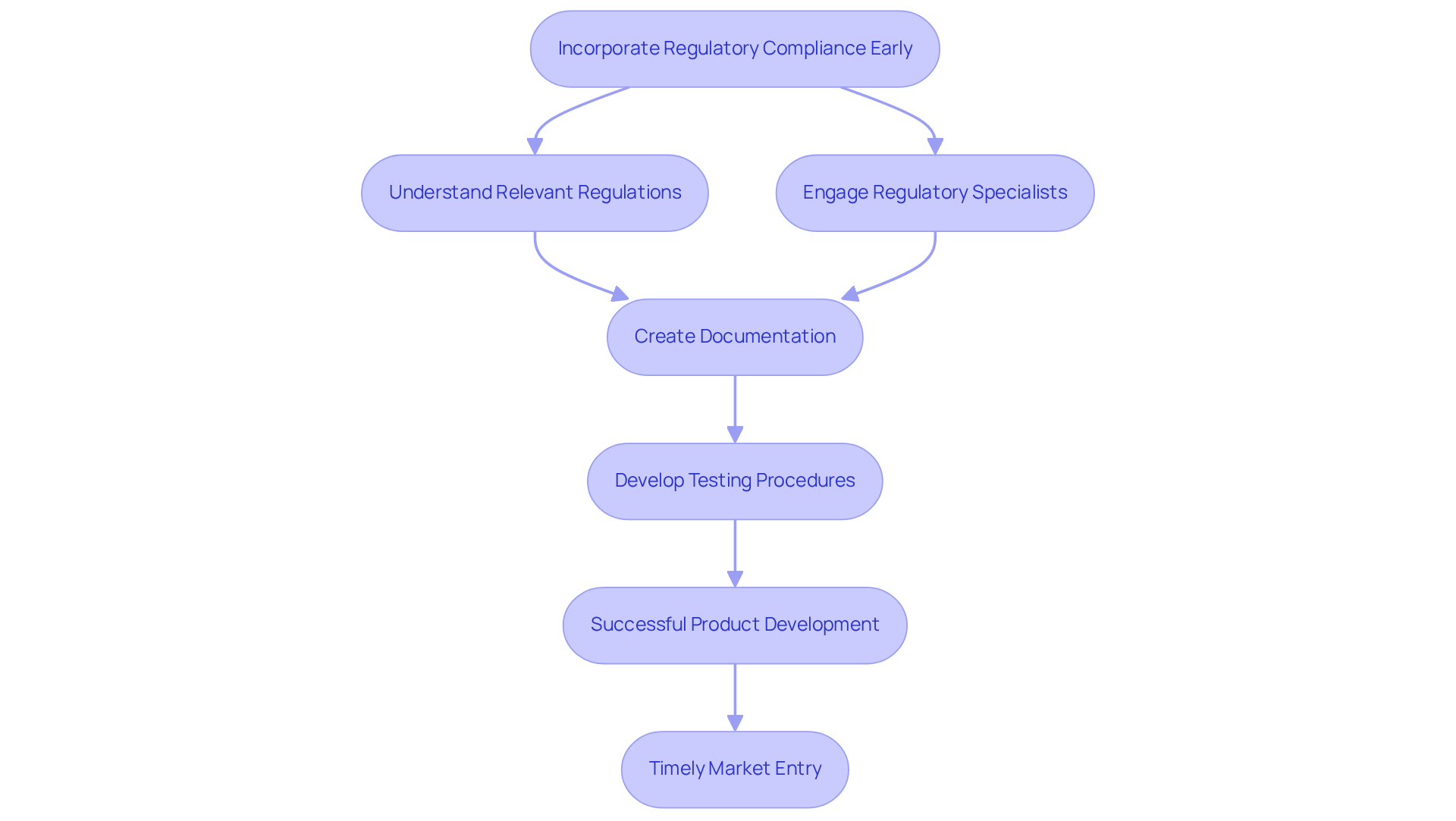
Incorporating regulatory compliance factors at the outset of device development is essential to mitigate potential issues later in the process. A thorough understanding of the relevant regulations and standards specific to the device is vital. Engaging regulatory specialists during the early planning stages allows teams to efficiently create necessary documentation and testing procedures. This proactive strategy not only but also significantly minimizes the risk of delays in bringing products to market.
For example, the FDA's recent guidance on Quality Management Systems underscores the significance of early compliance, which can expedite the approval process. Successful submissions, such as the IDx-DR, which received FDA clearance based on a well-documented clinical study, exemplify how early regulatory considerations can facilitate timely market entry. By prioritizing these elements, companies can improve their prospects for success in a competitive landscape.
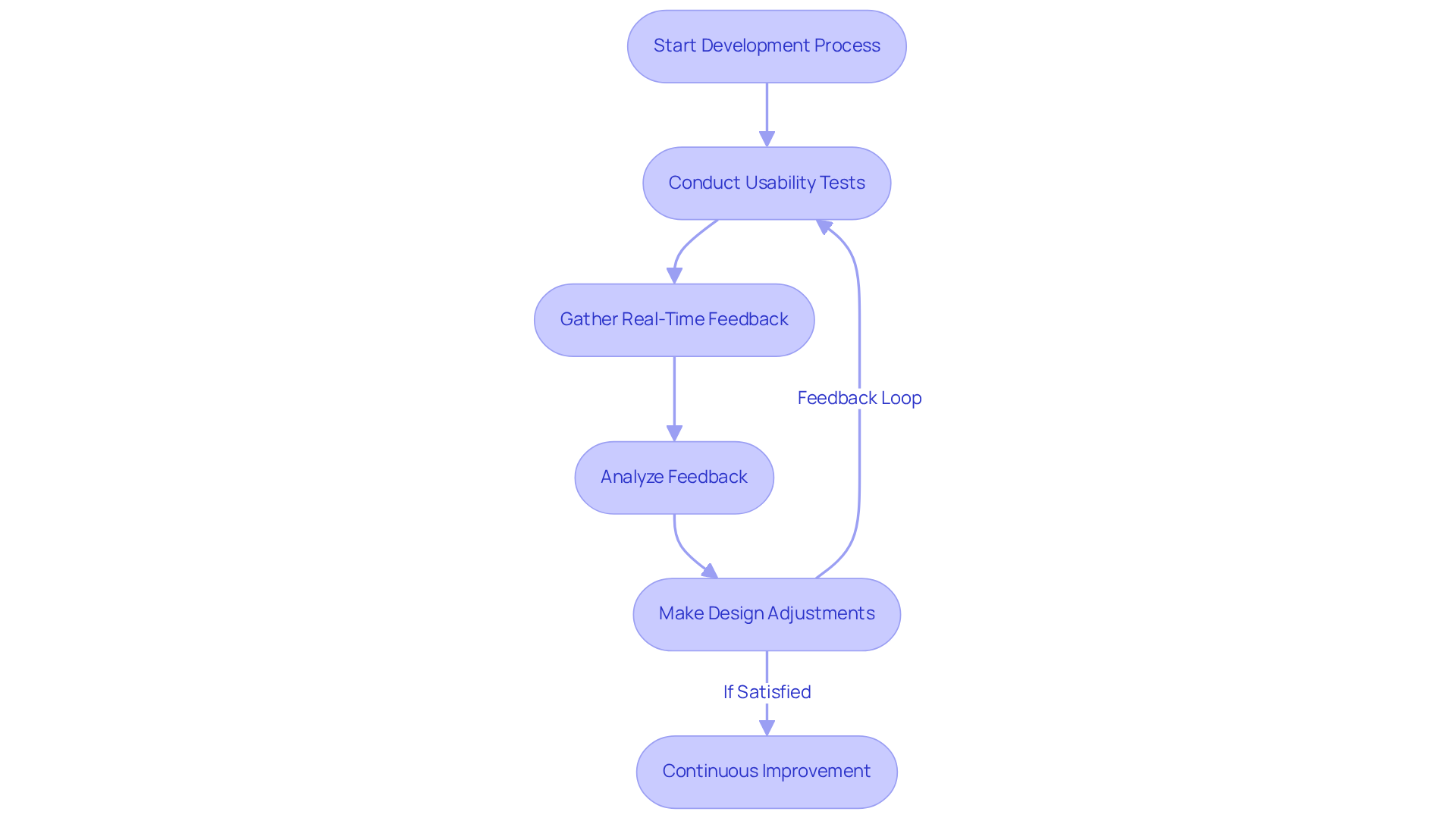
To ensure that medical devices effectively meet user needs, it is crucial to implement throughout the development process. Conducting usability tests at various phases of development allows teams to gather real-time feedback from end-users, facilitating prompt modifications to the design. Establishing a structured process for collecting and analyzing this feedback is essential for making informed design adjustments. By fostering a culture of continuous improvement, companies like [Voler Systems](https://volersystems.com) can significantly enhance product quality and user satisfaction, ultimately leading to improved patient outcomes.
For instance, iterative usability testing has been shown to reduce errors and increase user engagement, underscoring its vital role in the success of healthcare devices. Industry experts, including those at Voler Systems, emphasize that prioritizing user feedback not only streamlines the development process but also aligns products more closely with market needs, ensuring a higher rate of adoption and satisfaction. A notable example is the innovative calf-worn device designed for motion and circumference monitoring in knee replacement rehabilitation, which illustrates how user feedback can lead to compliant and reliable medical technology.
In the realm of medical device design, establishing effective product design services relies heavily on the integration of clear communication, defined roles, aligned goals, early regulatory compliance, and iterative feedback. Each of these core elements is crucial in driving successful outcomes, enabling teams to navigate the complexities of healthcare technology development with both precision and efficiency.
This article has highlighted several strategies, including:
These approaches not only streamline processes but also enhance collaboration among diverse team members. Ultimately, they lead to the development of innovative and compliant medical devices that meet user needs and adhere to regulatory standards.
The importance of implementing these best practices cannot be overstated. They contribute significantly to the timely delivery of high-quality products while fostering a culture of continuous improvement and accountability. For organizations engaged in medical device design, embracing these strategies is essential for maintaining competitiveness and ensuring that the innovations developed genuinely enhance patient care and safety.
Why is establishing clear communication channels important in medical device product design?
Establishing clear communication channels is essential to ensure successful product design service, as it allows team members-engineers, designers, regulatory experts, and marketing personnel-to collaborate effectively, address concerns, and enhance overall outcomes.
What are some key steps to implement effective communication practices?
Key steps include scheduling regular meetings to discuss project progress, utilizing collaborative tools like project management software, defining communication protocols for streamlined information sharing, and fostering an open environment for team members to express their thoughts and concerns.
What does the study by CRICO Strategies reveal about communication failures in healthcare?
The study reveals that over 7,000 out of 23,000 malpractice claims were due to communication failures, costing $1.7 billion and resulting in nearly 2,000 avoidable deaths.
How can the RACI matrix improve collaboration in medical device development?
The RACI matrix clarifies roles and responsibilities by specifying who is Responsible, Accountable, Consulted, and Informed for each task, reducing confusion and enhancing communication among team members.
Can you provide an example of how the RACI matrix has been used effectively?
Hospitals have successfully employed the RACI matrix to streamline processes, such as patient discharge, which has led to improved satisfaction and reduced delays.
Why is it important to regularly update the RACI matrix?
Regular updates to the RACI matrix are crucial to reflect changes in scope or personnel structure, ensuring its ongoing relevance and effectiveness in promoting successful collaboration and timely delivery.
