4 Best Practices for Your Product Requirement Document in Medical Devices
Master the essentials of a product requirement document for successful medical device...

Crafting a Product Requirements Document (PRD) for medical devices is not merely a technical necessity; it stands as a strategic imperative that can significantly influence the success or failure of a product within a highly regulated market. By concentrating on essential components such as functional requirements, performance metrics, and regulatory compliance, teams can develop a comprehensive roadmap that directs development and guarantees adherence to industry standards.
However, a critical challenge persists: how can teams effectively integrate these elements while promoting collaboration and steering clear of common pitfalls? This article explores four fundamental practices that can elevate a PRD from a simple formality into a robust tool for innovation and compliance in the medical device sector.
To create an effective Product Requirements Document (PRD) for medical devices, it is essential to include the following core components:
By including these components, teams can ensure that the product requirements document example pdf acts as a comprehensive guide throughout the product development lifecycle. The significance of detailed documentation in the medical equipment sector is highlighted by a product requirements document example pdf, which effectively incorporates a comprehensive product overview and functional requirements, demonstrating its ability to enhance project timelines and lower expenses. Furthermore, specialist perspectives highlight that setting clear performance metrics is essential for confirming effectiveness and ensuring adherence to regulatory standards such as ISO 13485:2016.
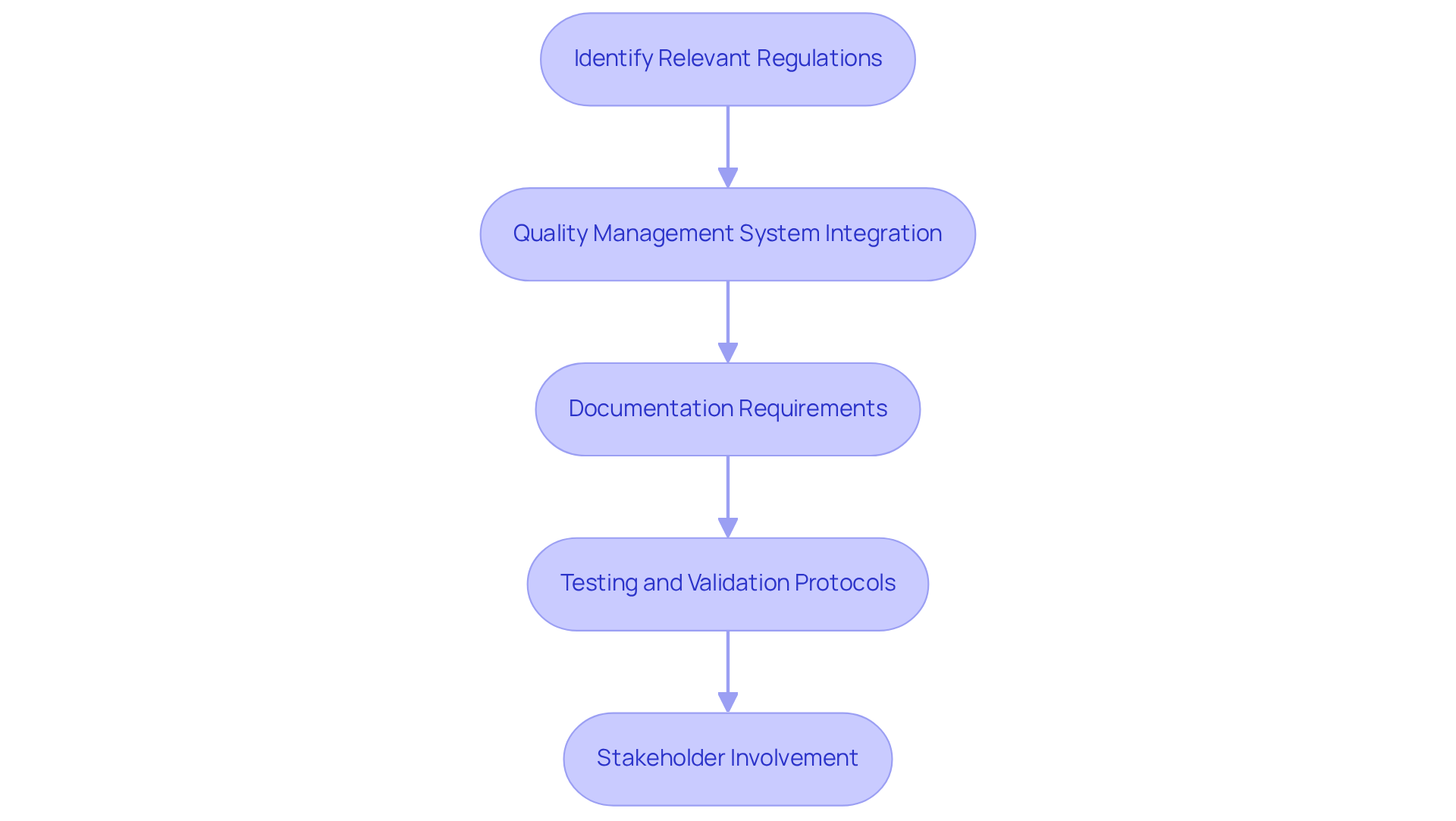
To ensure that a medical device meets regulatory compliance and quality standards, it is essential to integrate the following practices into the Product Requirements Document (PRD):
By embedding these elements into the product requirements document example pdf, teams can facilitate a smoother regulatory approval process while ensuring the safety and effectiveness of their medical devices. Additionally, being aware of common pitfalls in regulatory compliance can help avoid missteps in PRD development.
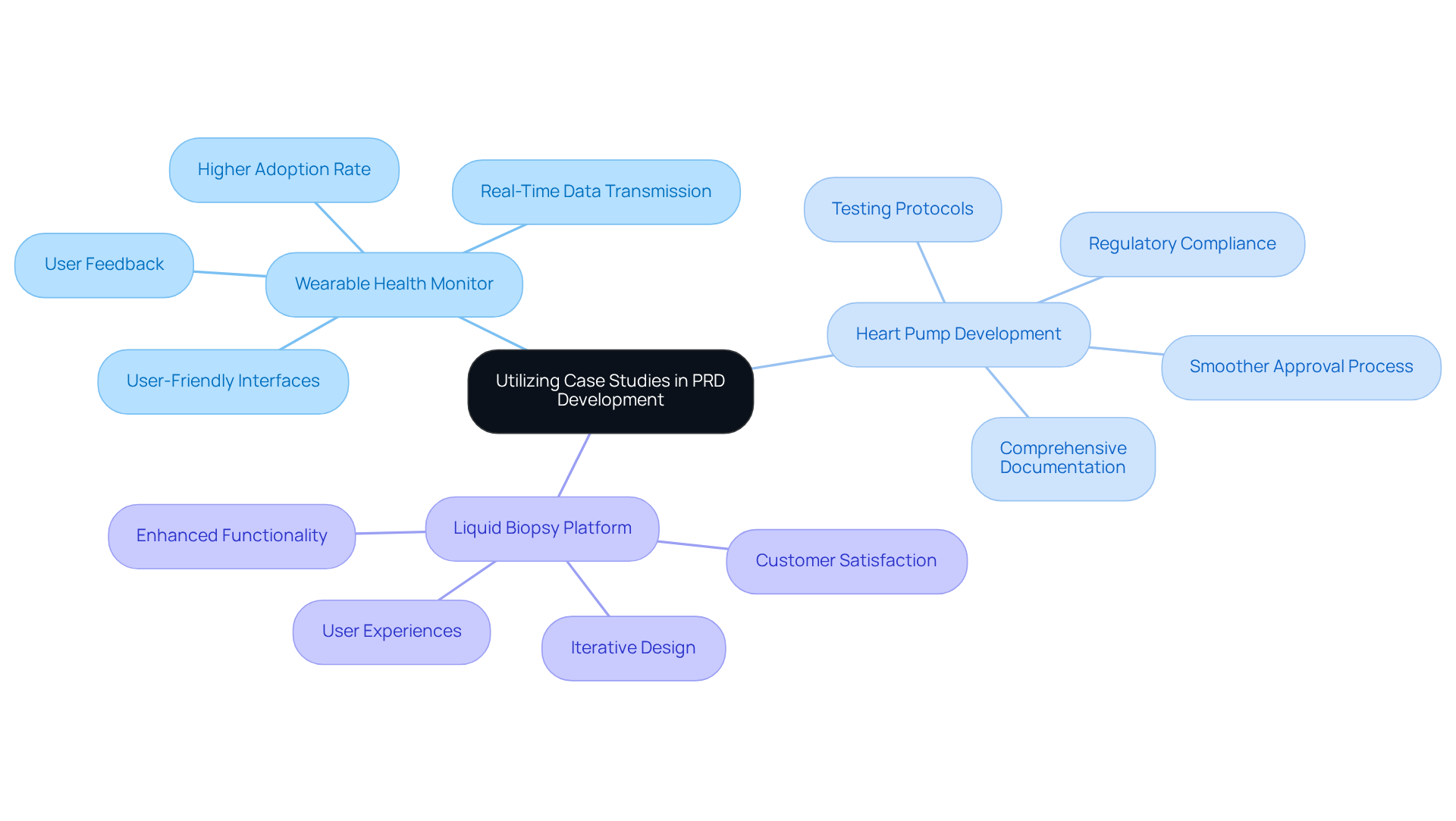
Incorporating case studies into the product requirements document example pdf process can significantly enhance the understanding and application of best practices. The following examples illustrate this point:
These examples demonstrate how leveraging case studies can inform the development of a product requirements document example pdf, resulting in more effective and user-centered medical devices.
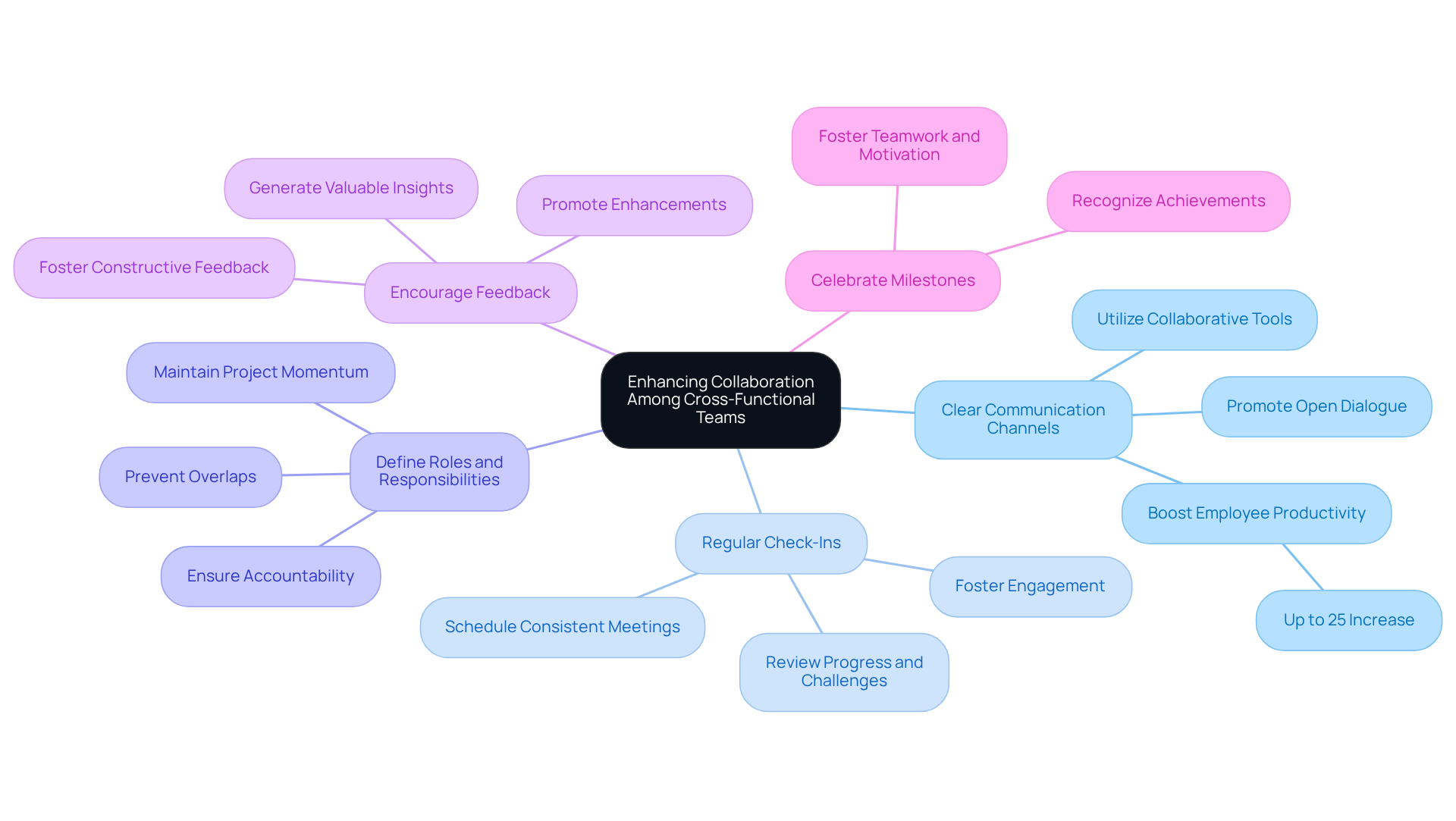
To enhance collaboration among cross-functional teams during the development of Product Requirements Documents (PRDs), consider implementing the following strategies:
By adopting these strategies, teams can significantly enhance collaboration, leading to a more effective PRD and ultimately contributing to the success of medical devices.
Creating a comprehensive Product Requirements Document (PRD) is essential for the successful development of medical devices. By concentrating on core components such as a clear product overview, functional requirements, performance metrics, user narratives, and out-of-scope items, teams can ensure that their PRD serves as a robust guide throughout the product lifecycle. This structured approach enhances communication among stakeholders and minimizes risks associated with regulatory compliance and quality standards, ultimately leading to improved product outcomes.
The article emphasizes the importance of integrating regulatory compliance practices, quality management systems, and thorough documentation into the PRD process. By identifying relevant regulations, outlining QMS integration, and defining testing protocols, teams can navigate the complex landscape of medical device approval more effectively. Real-world case studies illustrate the impact of these practices, demonstrating how user feedback and proactive planning can significantly enhance product development and market success.
In conclusion, the significance of a well-crafted PRD cannot be overstated. It is a vital tool that fosters collaboration among cross-functional teams, streamlines regulatory compliance, and ultimately enhances the effectiveness of medical devices. By adopting best practices and learning from real-world examples, organizations can improve their documentation processes and contribute to the advancement of healthcare technology. Embracing these strategies will pave the way for innovative solutions that meet the needs of users and regulatory bodies alike.
What is a Product Requirements Document (PRD) for medical devices?
A Product Requirements Document (PRD) for medical devices is a comprehensive guide that outlines the essential components, functionalities, and performance metrics necessary for the development of the device. It helps align the development team with the project's goals and ensures effective communication among stakeholders.
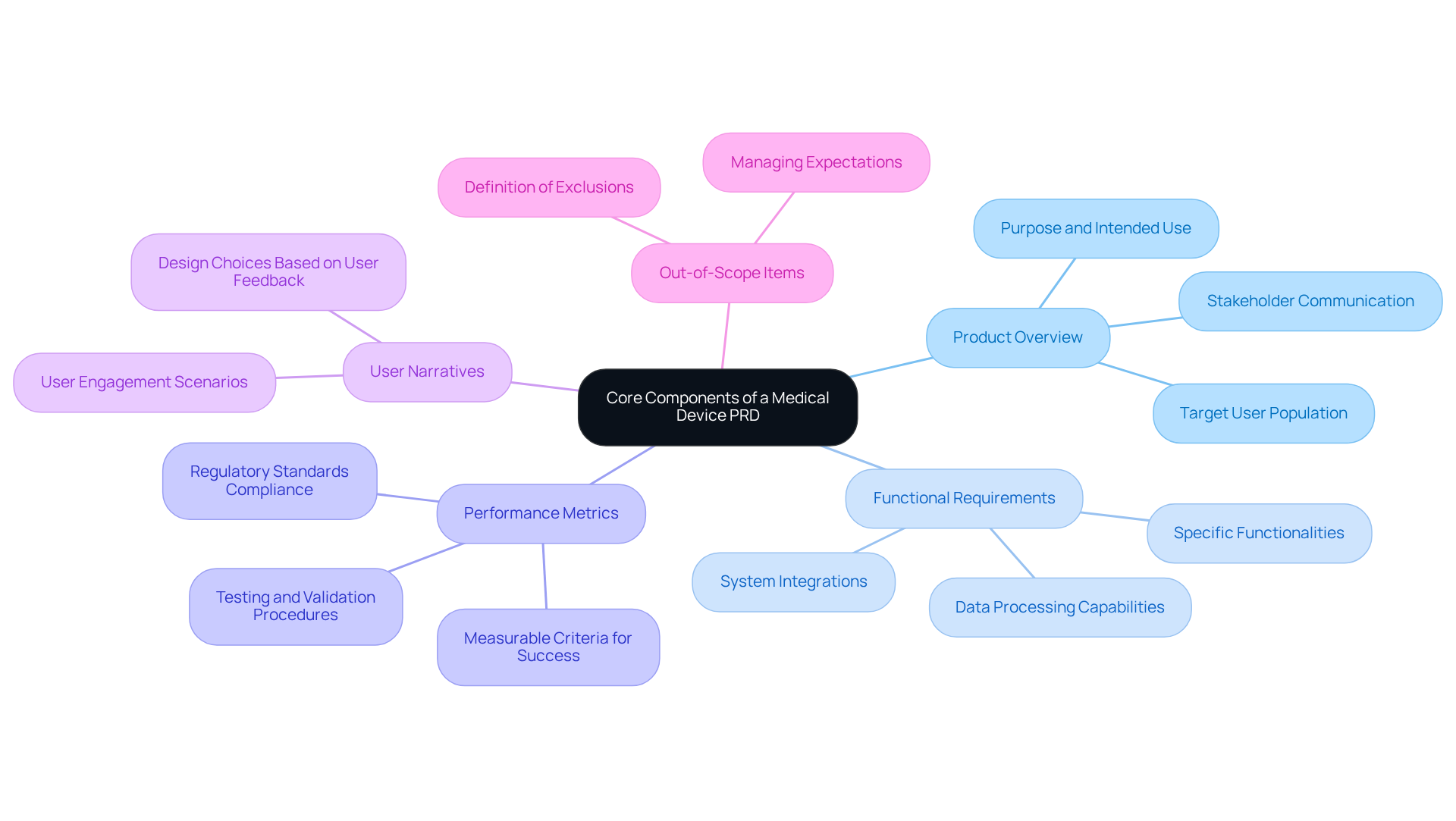
What are the core components of a medical device PRD?
The core components of a medical device PRD include: 1. Product Overview 2. Functional Requirements 3. Performance Metrics 4. User Narratives 5. Out-of-Scope Items
Why is the Product Overview important in a PRD?
The Product Overview is crucial as it clearly articulates the purpose of the device, its intended use, and the target user population. This sets the context for all subsequent requirements and enhances communication among stakeholders, streamlining the development process.
What should be included in the Functional Requirements section?
The Functional Requirements section should detail the specific functionalities the device must perform, including interactions, data processing capabilities, and essential integrations with other systems, such as real-time data transmission for a wearable health monitor.
What are Performance Metrics and why are they important?
Performance Metrics are measurable criteria for success, such as accuracy, reliability, and response times. They are important because they guide testing and validation procedures, ensuring the device meets industry standards and regulatory requirements.
How do User Narratives contribute to the PRD?
User Narratives illustrate how different personas will interact with the device, helping to understand user requirements and expectations. This approach improves user experience and guides design choices to make the device more intuitive and user-friendly.
What is the purpose of defining Out-of-Scope Items in a PRD?
Defining Out-of-Scope Items helps prevent scope creep and ensures that the project remains focused on essential features. It assists teams in managing expectations and effectively allocating resources.
How does a well-structured PRD impact the product development lifecycle?
A well-structured PRD acts as a comprehensive guide throughout the product development lifecycle, enhancing project timelines and lowering expenses. It emphasizes the importance of detailed documentation in the medical equipment sector.
