Best Practices in Electronic Product Design and Development for Medical Devices
Introduction The landscape of medical device development is rapidly evolving, propelled...

Creating printed circuit boards (PCBs) for medical devices is a complex endeavor that requires not only technical expertise but also a thorough understanding of regulatory standards and design best practices. As the healthcare industry continues to evolve, the demand for efficient, reliable, and compliant PCB designs becomes increasingly critical. This article explores four essential practices that engineers can adopt to enhance the effectiveness of their PCB designs. These practices range from mastering fundamental principles to leveraging advanced tools and iterative testing.
How can engineers effectively navigate the challenges of compliance and safety while simultaneously pushing the boundaries of innovation in medical device technology?
Creating efficient for requires a comprehensive understanding of key principles, particularly:
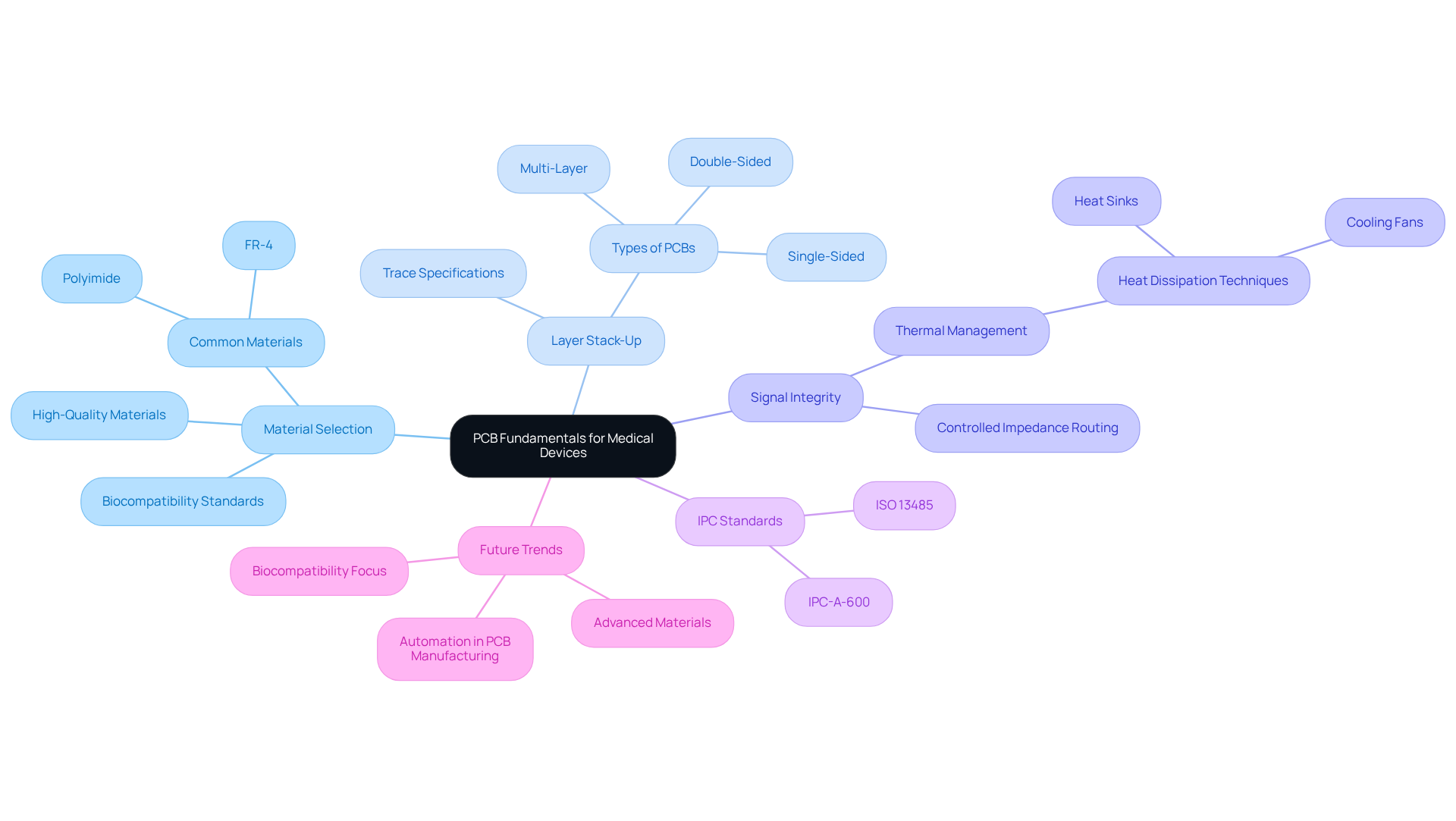
Each type of PCB - single-sided, double-sided, and multi-layer - has specific applications and limitations that must be evaluated within the medical context. Adhering to , is crucial for ensuring that designs meet stringent quality benchmarks. For example, utilizing like FR-4 or polyimide significantly enhances durability and performance, especially in devices subjected to varying environmental conditions.
Additionally, are vital to prevent overheating, particularly in continuously operating devices such as . By mastering these fundamentals, engineers can create PCBs that are not only functional but also reliable and compliant with rigorous .
Looking ahead to 2026, trends indicate a growing emphasis on and advanced materials, reflecting the industry's commitment to safety and innovation in healthcare technology.
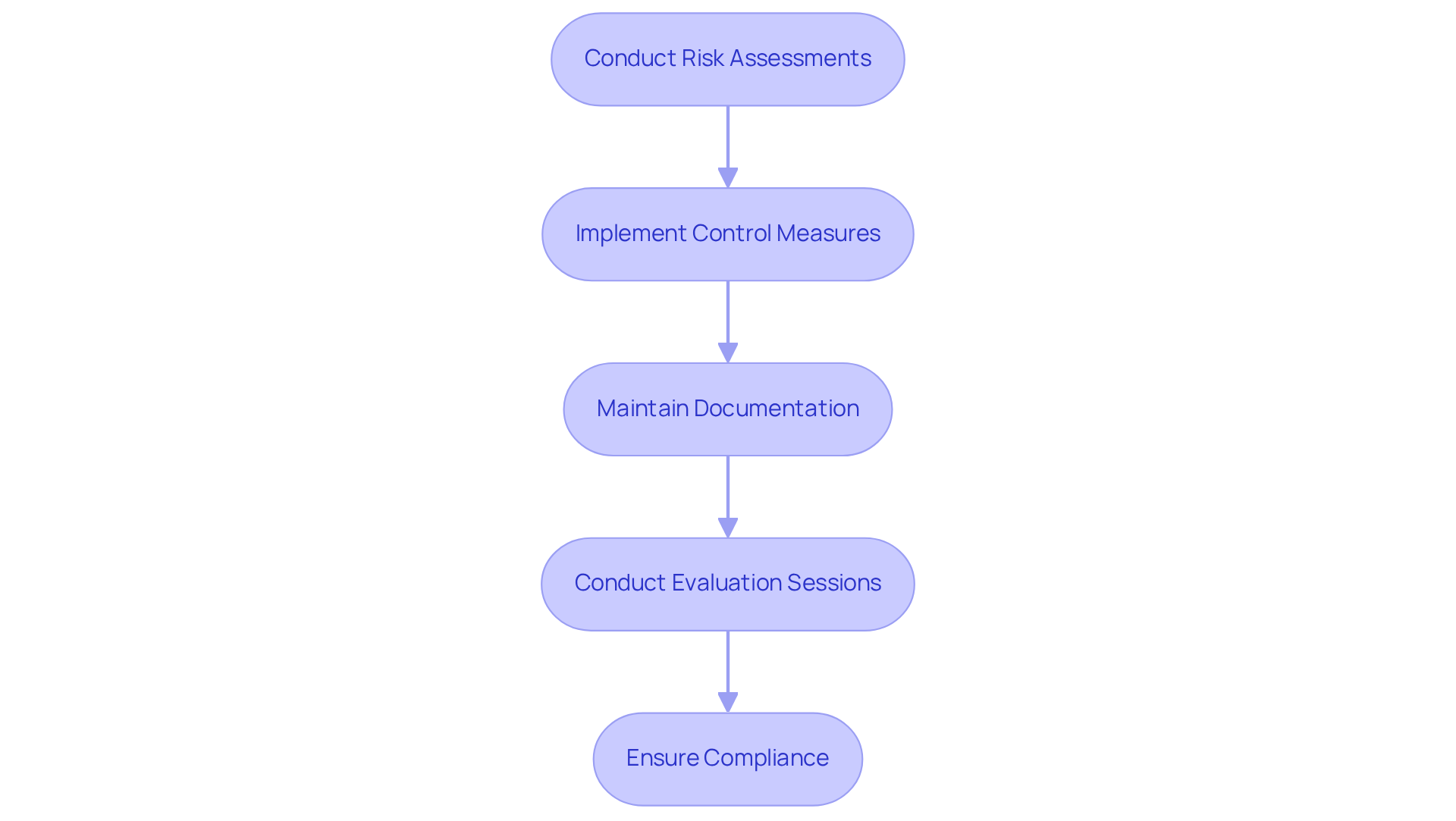
Designing for compliance and security requires the integration of at every stage of the PCB design process. must possess a thorough understanding of essential standards, such as , which governs quality management systems, and IEC 60601, which outlines for medical equipment.
Key steps in this process include:
By prioritizing adherence and safety, can ensure that their products are not only effective but also secure for end-users. This commitment ultimately leads to and increased customer trust.
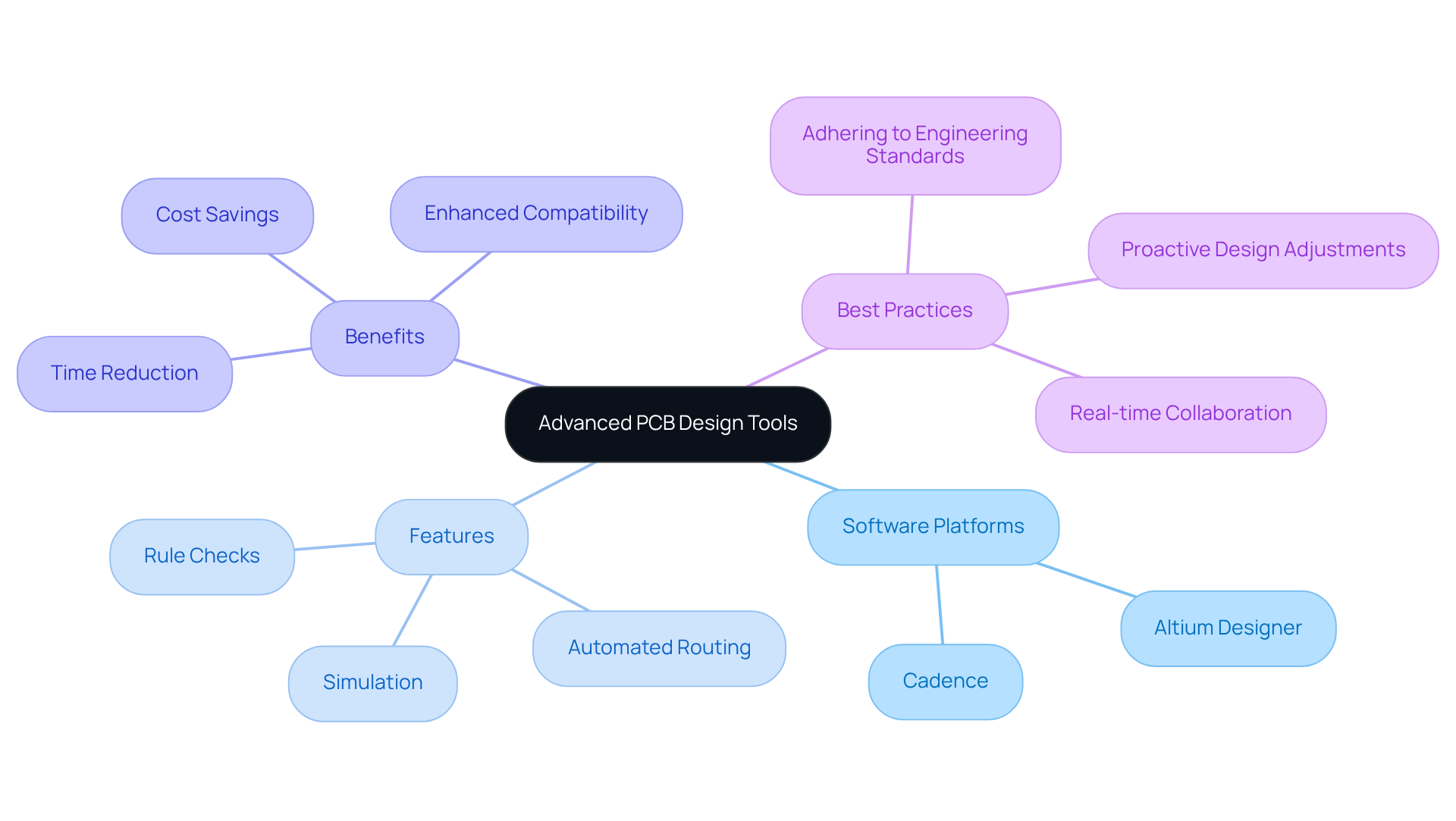
Utilizing advanced is crucial for improving the creation process in . Software platforms such as Altium Designer and Cadence provide essential features, including , which are necessary for complying with stringent industry standards. By adhering to optimal , - an essential factor for medical equipment manufacturers.
For instance, simulation tools can accurately predict issues before the physical prototypes are developed, significantly reducing both time and resource costs. Additionally, the integration of within equipment enclosures, enhancing compatibility and minimizing mechanical conflicts.
Moreover, the collaboration tools embedded in these development platforms facilitate , streamlining the workflow. By adopting these advanced tools and adhering to , engineers can markedly improve their productivity and elevate the overall quality of their healthcare product developments.
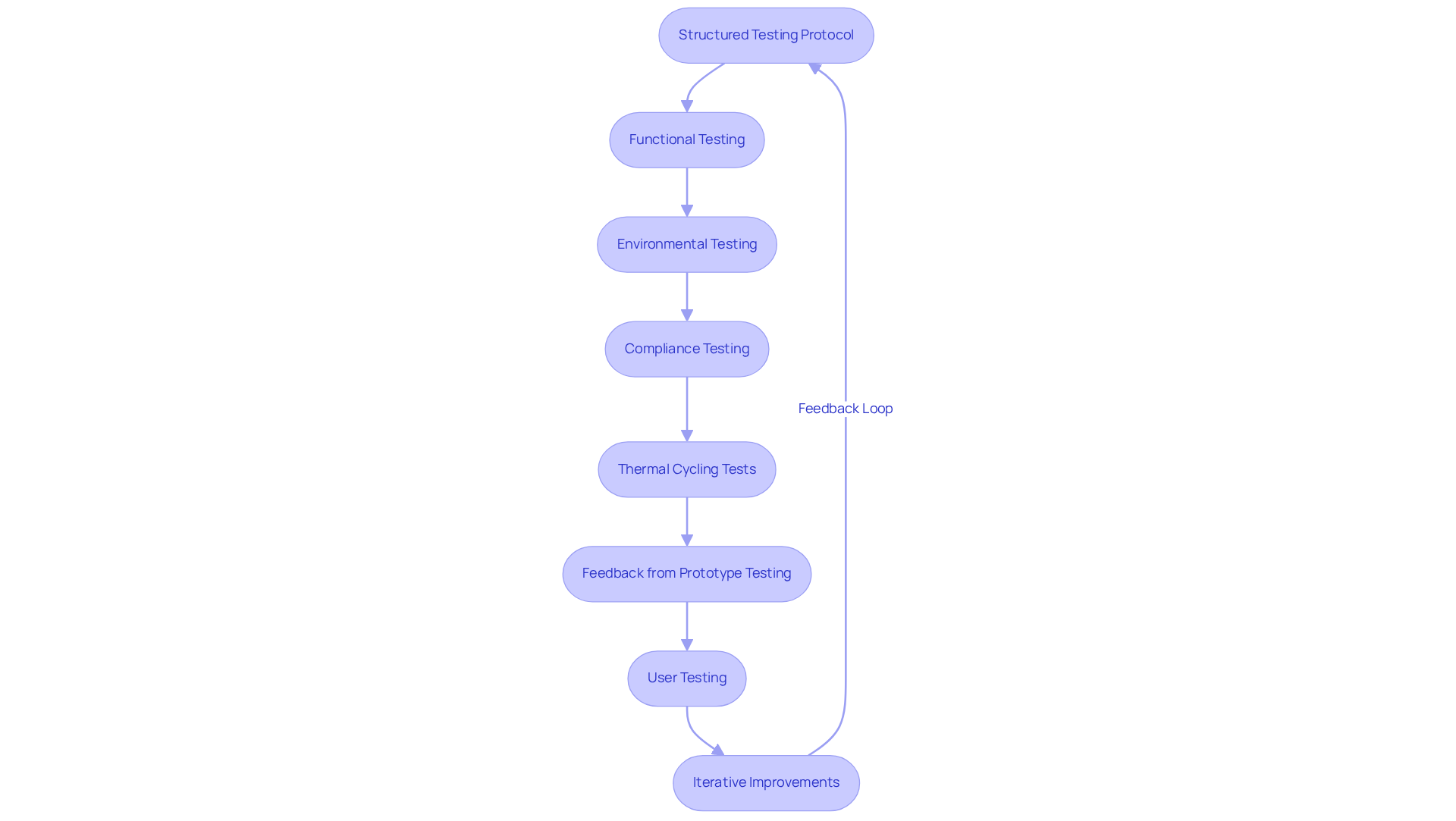
Iterative testing and validation are critical for the successful development of medical equipment PCBs. Engineers must implement a structured that includes functional, environmental, and . For instance, thermal cycling tests are vital for evaluating PCB performance under extreme temperature fluctuations, which is essential for equipment functioning in diverse environments. These tests significantly impact , as they simulate real-world conditions that the equipment may encounter.
Moreover, leveraging feedback from prototype testing promotes , enabling engineers to address potential issues prior to mass production. Engaging in into the product's usability and functionality, ensuring it meets the needs of healthcare professionals and patients. A recent case analysis highlights how assisted a healthcare equipment company in enhancing their legacy testing tools, underscoring the importance of managing compliance throughout the development process.
By adopting an , engineers can improve the reliability and safety of their , ultimately leading to better patient outcomes.
Creating effective printed circuit boards (PCBs) for medical devices requires a comprehensive understanding of foundational principles and compliance requirements. By concentrating on critical aspects such as material selection, safety design, and advanced tools, engineers can ensure their designs not only meet regulatory standards but also improve patient outcomes. Integrating these best practices is essential for fostering innovation and reliability in healthcare technology.
This article explored key insights, including the necessity of mastering PCB fundamentals, implementing rigorous compliance and safety measures, utilizing advanced design tools, and conducting thorough iterative testing. Each of these elements is vital to the overall success of medical device development, ensuring that products are effective and safe for end-users. The emphasis on continuous improvement through feedback and testing underscores the commitment to quality and innovation in the field.
As the healthcare technology landscape evolves, embracing these best practices will be crucial for engineers and manufacturers. Prioritizing compliance, safety, and advanced design methodologies will enhance product reliability and build trust with customers and stakeholders. The future of medical devices depends on a dedication to excellence in PCB design, ultimately leading to improved patient care and outcomes.
What are the key principles to understand when creating PCBs for medical devices?
The key principles include material selection, layer stack-up, and signal integrity.
What types of PCBs are commonly used in medical devices?
The commonly used types of PCBs in medical devices are single-sided, double-sided, and multi-layer PCBs.
Why is adhering to IPC standards important in PCB design for medical devices?
Adhering to IPC standards, such as IPC-A-600, is crucial for ensuring that designs meet stringent quality benchmarks required in the medical field.
What materials are recommended for enhancing the durability and performance of PCBs in medical devices?
High-quality materials such as FR-4 or polyimide are recommended for enhancing durability and performance, especially in devices exposed to varying environmental conditions.
Why is thermal management important in PCBs for medical devices?
Effective thermal management is vital to prevent overheating, particularly in continuously operating devices like wearable sensors.
What is the significance of mastering PCB fundamentals for engineers in the medical device field?
Mastering these fundamentals enables engineers to create PCBs that are functional, reliable, and compliant with rigorous medical device regulations.
What trends are expected in PCB design for medical devices by 2026?
By 2026, there is expected to be a growing emphasis on biocompatibility and advanced materials, reflecting the industry's commitment to safety and innovation in healthcare technology.
