Understanding DFM Meaning: Key Insights for Medical Device Manufacturers
Discover the dfm meaning and its impact on medical device manufacturing efficiency and...

A comprehensive understanding of Design for Manufacturing (DFM) is essential in the engineering landscape, particularly in the healthcare sector where precision is paramount. This strategic approach not only streamlines production processes but also enhances product quality and compliance with stringent regulations.
Manufacturers often struggle to balance innovation with cost control while adhering to DFM principles, raising the question of how to effectively integrate these principles to enhance efficiency and safety in medical device production.
This article examines the significance of DFM, its core principles, historical evolution, and real-world applications that underscore its impact on the industry.
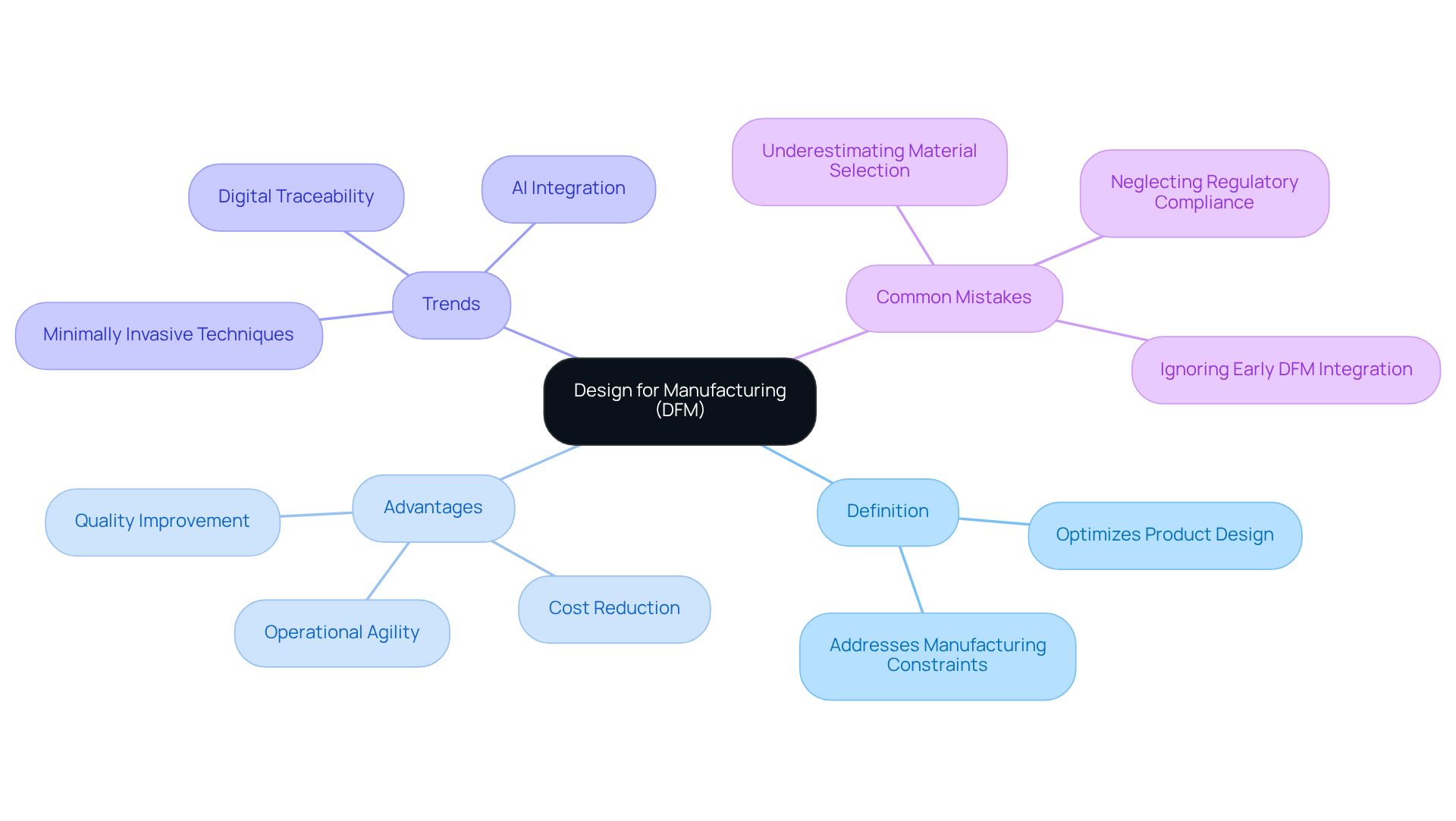
In engineering, what does DFM stand for in engineering is an essential practice that optimizes product design for efficient manufacturing processes. This approach focuses on reducing production costs and enhancing product quality by addressing manufacturing constraints during the design phase. DFM prioritizes the design of parts that facilitate easy production, assembly, and testing, resulting in more efficient production cycles and improved quality outcomes. In the healthcare equipment sector, it is crucial to understand what does dfm stand for in engineering, as it ensures that products not only meet stringent regulatory standards but are also produced reliably and economically.
The advantages of DFM in healthcare product manufacturing are significant. By integrating DFM principles early in the design process, manufacturers can markedly improve product quality and control costs, ultimately demonstrating what does dfm stand for in engineering and leading to successful product launches in a competitive market. For instance, companies that adopt DFM practices can eliminate unnecessary steps and optimize material usage, allowing them to offer high-quality products at competitive prices. This is crucial as the industry shifts focus from mere compliance to operational performance and agility.
Recent trends underscore the importance of DFM in the evolving landscape of healthcare equipment. As manufacturers encounter pressures from regulatory bodies and market demands, it becomes essential to understand what does dfm stand for in engineering to navigate these challenges effectively. For example, the integration of digital traceability and AI insights into DFM practices can enhance operational agility and ensure compliance with evolving standards. Moreover, the growth of minimally invasive techniques has increased the demand for sophisticated healthcare tools, positioning DFM as a critical element in addressing these market requirements.
Common mistakes in production evaluations can significantly impact the success of healthcare instruments. By addressing these errors through DFM, manufacturers can enhance testing efficiency and product reliability. What does dfm stand for in engineering is crucial as it not only ensures compliance with regulatory standards but also drives innovation and efficiency in healthcare product manufacturing. By prioritizing DFM, companies can not only meet regulatory demands but also drive innovation and efficiency in their manufacturing processes.
In the healthcare device sector, it is essential to know what DFM stands for in engineering, as its absence can lead to significant production challenges and regulatory hurdles. By embedding principles that answer what DFM stands for in engineering early in the design process, engineers can foresee potential manufacturing challenges, leading to designs that are not only functional but also easy to produce. This proactive approach significantly reduces the risk of expensive redesigns and production delays, which can lead to significant setbacks in the highly regulated healthcare sector. Research indicates that effective DFM practices can reduce redesigns by up to 30%. This reduction directly influences how quickly products can reach the market.
Integrating DFM, which is what DFM stands for in engineering, can improve adherence to strict regulatory standards, ensuring that healthcare products are produced consistently and safely. Additionally, DFM ensures that design intent aligns closely with clinical realities, a factor vital for patient safety and product effectiveness. Voler Systems plays a crucial role in assisting healthcare product startups by offering thorough documentation compliance support, including advice on regulatory submissions and quality management systems, aiding them in navigating the intricate regulatory environment.
The integration of DFM, which is what DFM stands for in engineering, not only streamlines the development process but also strengthens regulatory documentation, making it easier to meet compliance requirements. As the FDA has increased examinations of healthcare instruments, with a significant 46% increase in yearly inspections from 2007 to 2017, the significance of DFM in upholding high compliance standards cannot be emphasized enough. By adopting DFM principles, manufacturers of healthcare products can enhance product quality, reduce time to market, and ultimately improve patient outcomes, all while benefiting from Voler Systems' expertise in compliant and reliable electronic solutions. Manufacturers who overlook DFM risk not only their market position but also the safety and efficacy of their products.
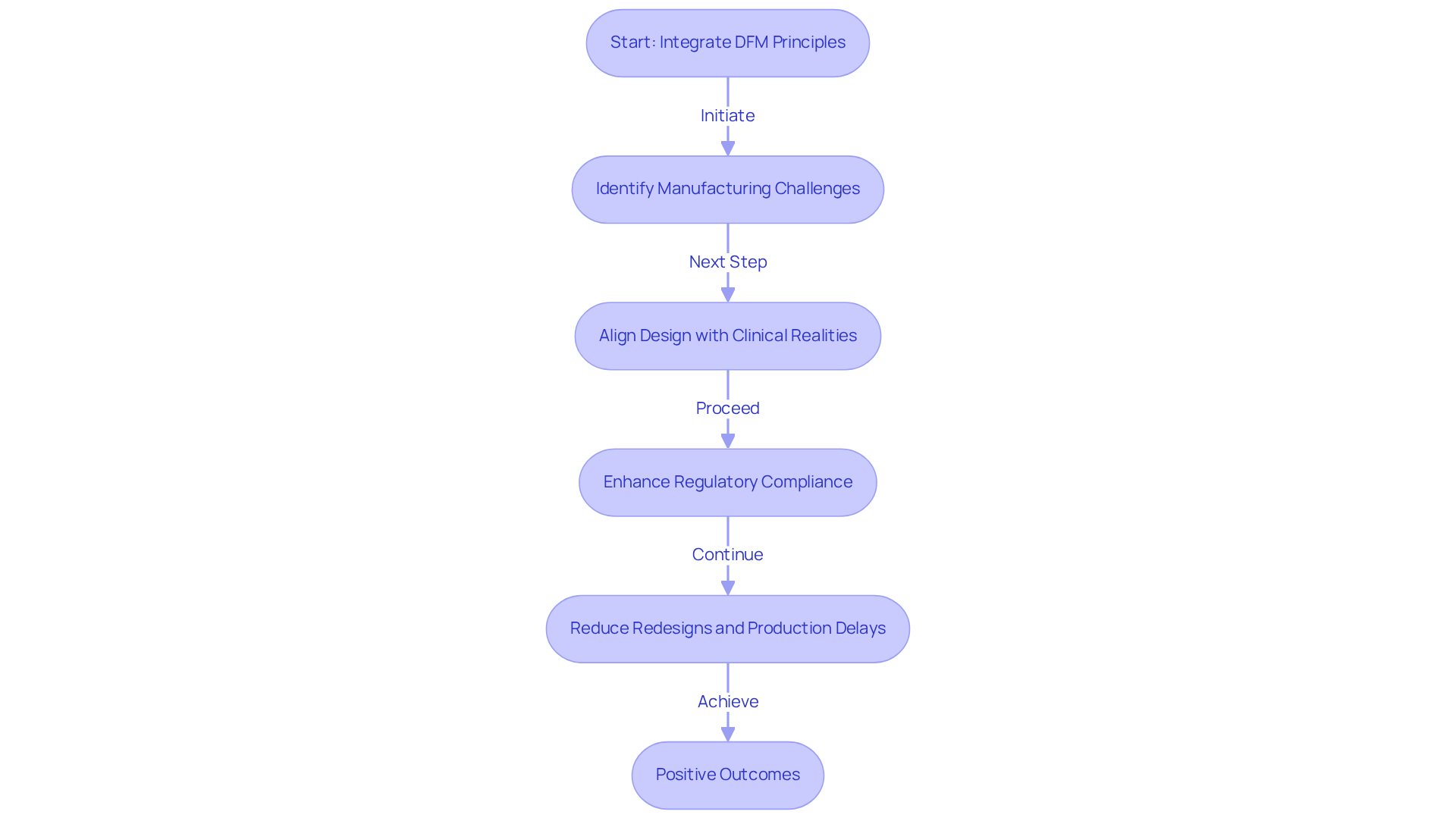
The evolution of Design for Manufacturing (DFM), which raises the question of what does DFM stand for in engineering, reflects the increasing complexity of production processes in the modern era. Since its inception in the early 20th century, DFM has significantly progressed, raising the question of what does DFM stand for in engineering, while paralleling advancements in manufacturing technology.
Initially, manufacturing was predominantly craft-based, but as industries expanded, the necessity for standardized production methods became evident. The advent of mass production techniques in the mid-1900s marked a pivotal shift, prompting a focus on designing products for efficient large-scale manufacturing.
Over the decades, DFM has integrated modern technologies and methodologies, notably computer-aided drafting (CAD) and lean manufacturing principles. CAD has improved design accuracy, and lean manufacturing has streamlined production, minimizing waste and enhancing efficiency. These advancements have turned DFM, which refers to what does DFM stand for in engineering, into an essential element of product development, particularly in the healthcare equipment sector, where the smooth integration of planning and manufacturing processes is critical for meeting rigorous compliance standards and guaranteeing product quality.
Furthermore, the application of DFM principles has been shown to lead to a 20% reduction in assembly components and a 21% reduction in manufacturing costs, illustrating its measurable advantages. Consequently, DFM now fulfills a vital function in the successful marketing of healthcare products, conforming to regulatory standards and improving patient safety and clinical results.
Voler Systems exemplifies leadership in this evolving landscape through its commitment to compliant and reliable electronic device development, particularly in the field of wearable medical equipment that tracks continuous vital signs. As the healthcare industry continues to advance, the role of DFM in ensuring product efficacy and safety will only become more pronounced.
Understanding what does dfm stand for in engineering is essential for optimizing production efficiency in the medical device industry through key principles of Design for Manufacturing (DFM).
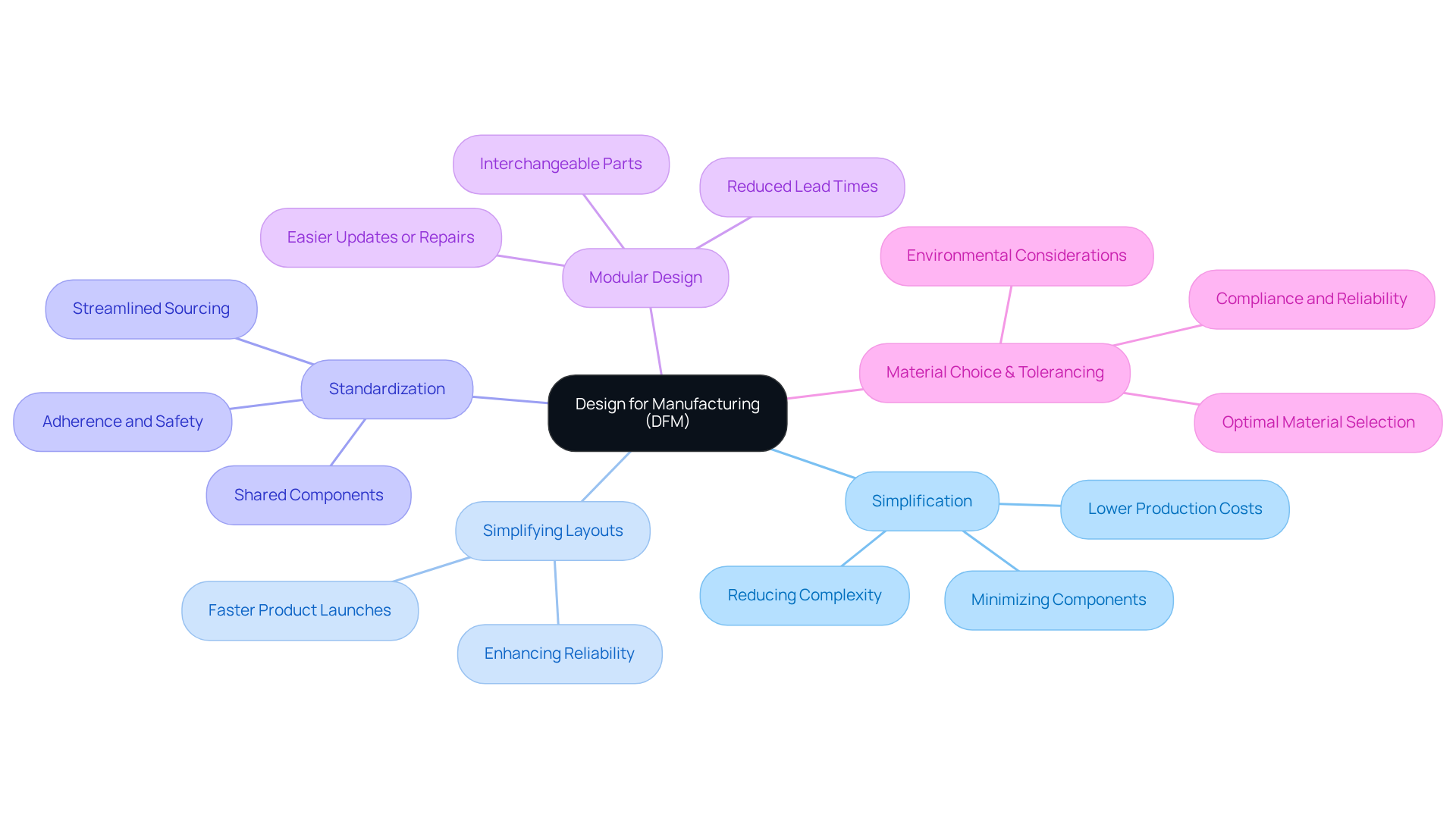
In the medical equipment industry, manufacturers often grapple with high production costs and the need for reliable devices. For instance, understanding what does DFM stand for in engineering and applying those principles in the design of a wearable health monitor allows for the use of standardized components, which simplifies assembly and reduces costs. A notable case study on a cardiac instrument revealed that implementing principles related to what does DFM stand for in engineering resulted in a 30% reduction in production costs and improved instrument reliability.
Moreover, knowing what does DFM stand for in engineering allows practices to effectively identify potential manufacturing issues early in the design process, enabling timely adjustments that ensure compliance with regulatory standards. By prioritizing DFM, which leads us to consider what does DFM stand for in engineering, manufacturers of healthcare equipment can achieve faster time-to-market, improved product quality, and enhanced patient safety.
Statistics indicate that implementing what does DFM stand for in engineering can lead to a 21% reduction in manufacturing costs, underscoring its value in the competitive landscape of medical device production. Understanding what does DFM stand for in engineering is essential, as the strategic implementation of DFM is not merely beneficial but crucial for maintaining competitiveness in the medical device market.
In the competitive landscape of medical device manufacturing, the absence of Design for Manufacturing (DFM) can lead to significant operational challenges. DFM is essential in medical device engineering, ensuring products are designed for both functionality and efficient production. Emphasizing DFM principles early in the design phase leads to improvements in product quality, cost management, and compliance with regulatory standards. This proactive approach enhances manufacturability and aids manufacturers in addressing healthcare challenges.
Throughout the article, key insights reveal the multifaceted advantages of DFM, including:
Real-world applications demonstrate how DFM can result in substantial cost savings and improved reliability in medical devices. The integration of modern technologies and methodologies further underscores the importance of DFM in achieving operational efficiency and compliance in an increasingly competitive market.
As the healthcare industry evolves, the importance of DFM becomes increasingly clear. It represents a strategic imperative for manufacturers aiming to deliver high-quality, safe, and effective medical devices. By embracing DFM principles, stakeholders can meet regulatory demands while fostering innovation and efficiency, ultimately leading to better patient outcomes. Embracing DFM is crucial for the future success of medical device manufacturing in a rapidly changing environment.
What does DFM stand for in engineering?
DFM stands for Design for Manufacturing, which is an essential practice that optimizes product design for efficient manufacturing processes.
What are the main goals of DFM?
The main goals of DFM are to reduce production costs and enhance product quality by addressing manufacturing constraints during the design phase, leading to efficient production cycles and improved quality outcomes.
Why is DFM important in the healthcare equipment sector?
DFM is crucial in the healthcare equipment sector because it ensures that products meet stringent regulatory standards while being produced reliably and economically.
How can DFM benefit healthcare product manufacturing?
By integrating DFM principles early in the design process, manufacturers can improve product quality, control costs, eliminate unnecessary steps, and optimize material usage, leading to successful product launches.
What recent trends highlight the importance of DFM?
Recent trends include pressures from regulatory bodies and market demands, the integration of digital traceability and AI insights, and the growth of minimally invasive techniques, all of which position DFM as a critical element in healthcare manufacturing.
What common mistakes in production evaluations can DFM help address?
DFM can help address errors that impact the success of healthcare instruments, enhancing testing efficiency and product reliability.
How does DFM relate to regulatory compliance?
DFM ensures compliance with regulatory standards and drives innovation and efficiency in healthcare product manufacturing, making it essential for meeting regulatory demands.
What are the consequences of not implementing DFM in the healthcare sector?
Not implementing DFM can lead to significant production challenges, regulatory hurdles, expensive redesigns, production delays, and risks to product safety and efficacy.
How does DFM affect the time to market for healthcare products?
Effective DFM practices can reduce redesigns by up to 30%, directly influencing how quickly products can reach the market.
What role does Voler Systems play in relation to DFM?
Voler Systems assists healthcare product startups by offering documentation compliance support, advice on regulatory submissions, and quality management systems to navigate the regulatory environment effectively.
