What Does DFM Stand for in Engineering? Key Insights for Medical Devices
The keyword 'what does dfm stand for in engineering' must be included in the summary.

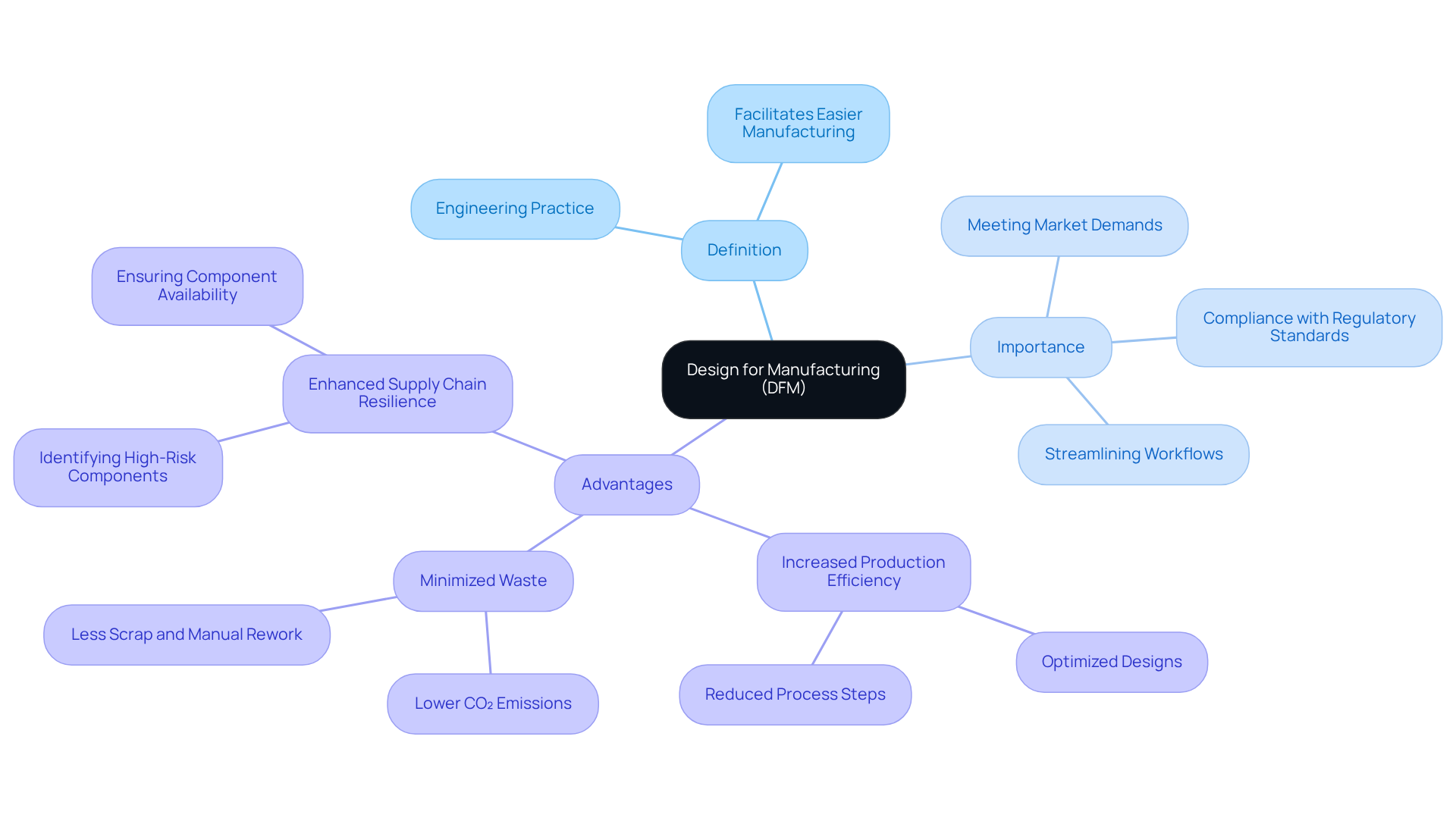
The integration of Design for Manufacturing (DFM) principles is crucial for navigating the complexities of medical device manufacturing. DFM prioritizes simplification and optimization from the outset of product development. Embracing DFM principles enhances production efficiency and improves product quality and regulatory compliance, which are critical in ensuring patient safety.
As the regulatory landscape evolves, manufacturers face the challenge of effectively integrating DFM principles. This integration is essential for maintaining competitiveness and responsiveness to market demands. Without effective DFM integration, manufacturers risk falling behind in a rapidly evolving market.
The dfm meaning highlights how Design for Manufacturing addresses the complexities of product design by prioritizing simplification and optimization from the outset. This approach is crucial in the medical equipment sector, ensuring compliance with regulatory standards and meeting market demands. Incorporating production considerations early helps identify potential challenges, streamlining workflows and improving efficiency. This proactive approach leads to significant cost savings and improved product quality. In 2026, the focus on DFM meaning becomes more pronounced as manufacturers aim for enhanced quality and reliability, ensuring that items are produced consistently and efficiently.
The key advantages of adopting a strategy that aligns with dfm meaning include:
Ultimately, understanding dfm meaning not only enhances operational efficiency but also positions manufacturers to thrive in an increasingly competitive market. Voler Systems utilizes specific methodologies in their DFM strategy, concentrating on early integration of production constraints and ongoing collaboration with stakeholders to ensure that items meet both quality standards and market demands.
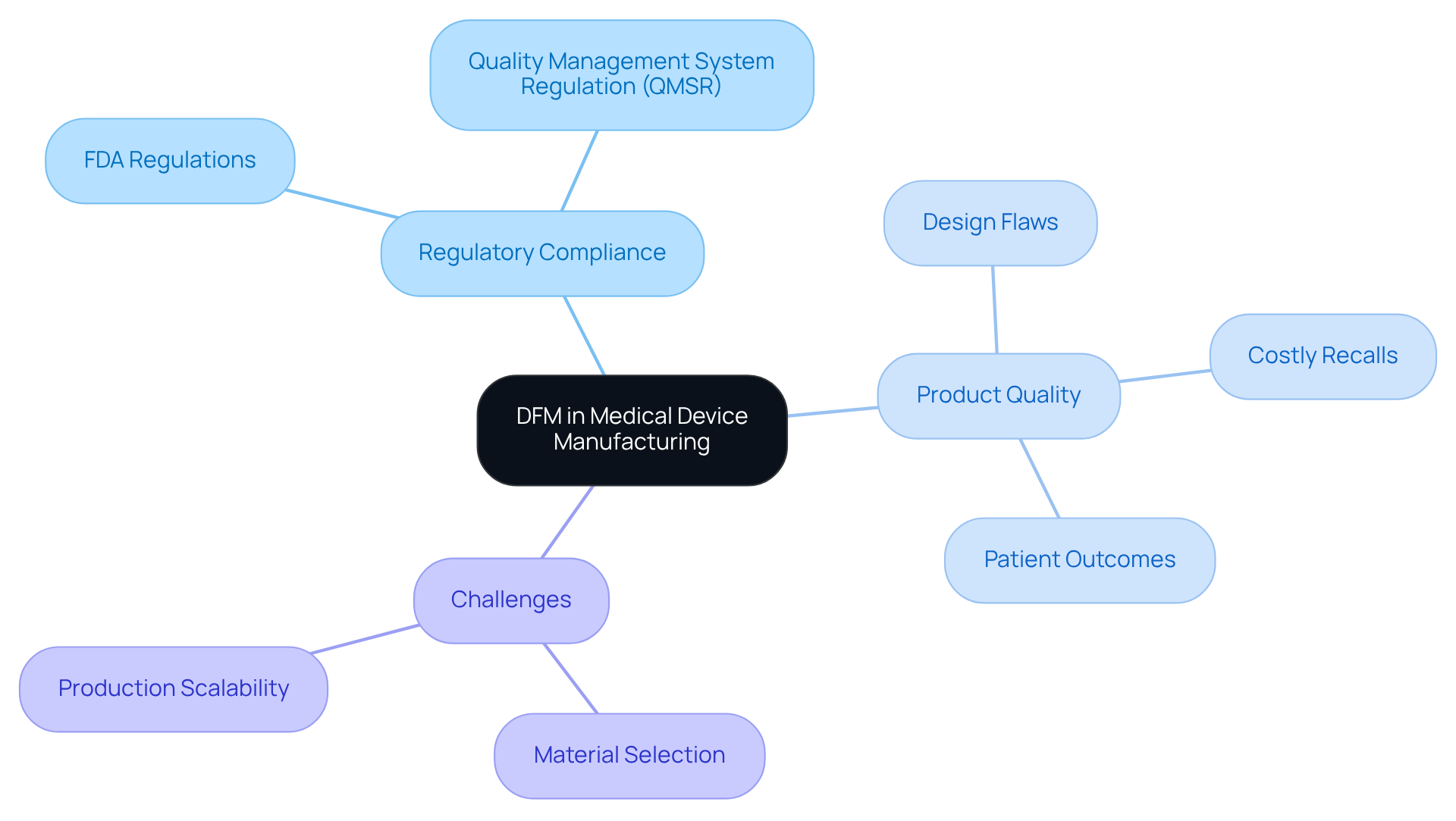
In the medical equipment industry, regulatory compliance poses significant challenges that necessitate effective strategies. The dfm meaning is crucial for ensuring that medical products are manufacturable, safe, and effective for end-users. Integrating DFM principles, which include dfm meaning, allows manufacturers to navigate FDA regulations effectively while addressing challenges like material selection and production scalability. The dfm meaning is crucial as it helps identify design flaws early, preventing costly recalls and compliance issues, thus safeguarding the manufacturer's reputation and patient health. This proactive approach is vital in the medical device sector, where precision and reliability directly impact patient outcomes.
Case studies show that effective DFM implementation enhances product quality and regulatory compliance, improving the manufacturing process. With the new Quality Management System Regulation (QMSR) in effect, the regulatory landscape is evolving. Understanding dfm meaning is increasingly important for addressing these challenges. Manufacturers who adopt DFM early are better positioned to meet FDA regulations and ensure the safety and effectiveness of their products.
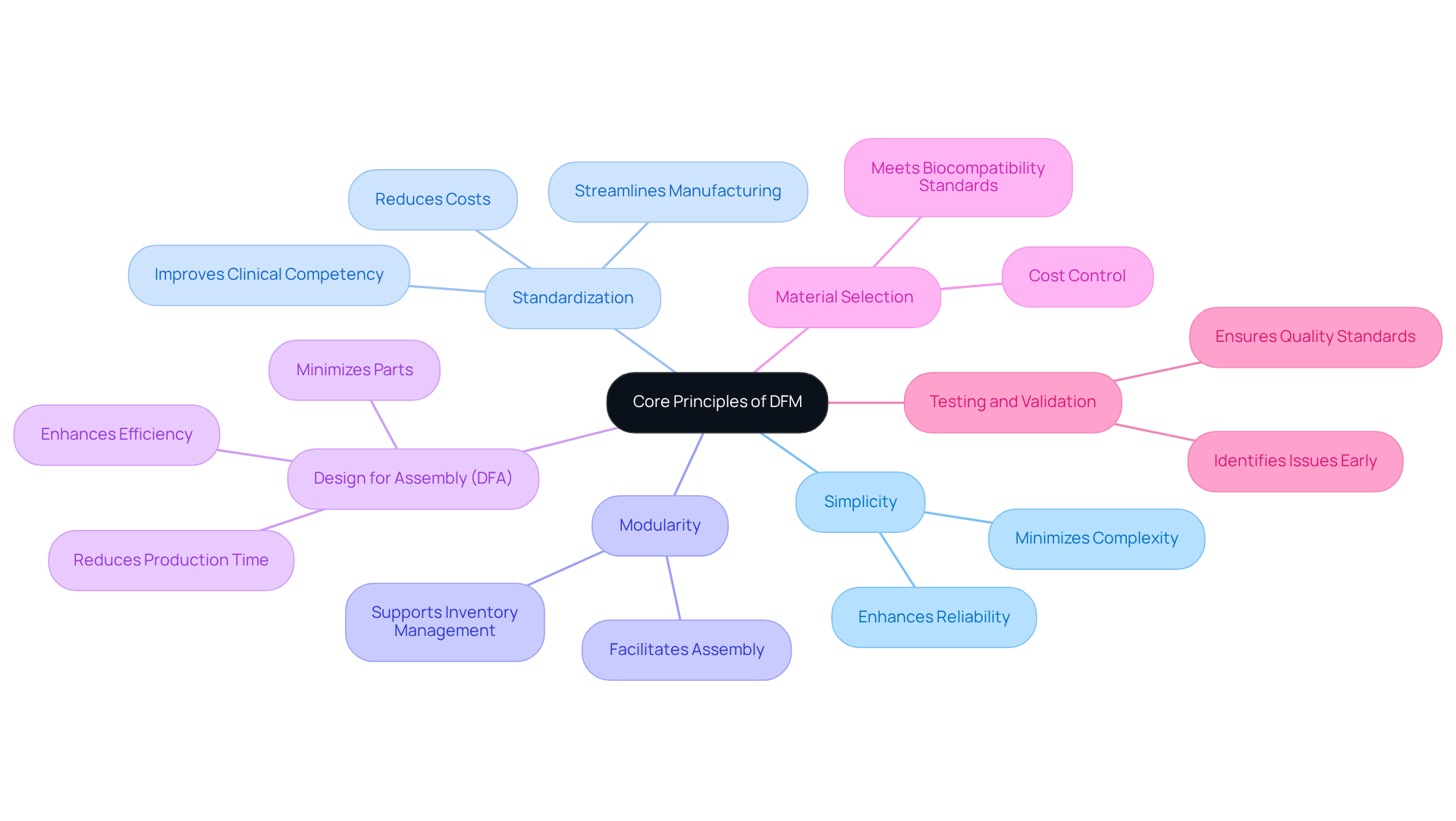
Understanding the DFM meaning is crucial for applying its principles to optimize medical device production and ensure compliance with quality and regulatory standards. Key principles include:
By following these principles, medical equipment manufacturers can create products that are not only manufacturable but also capable of meeting the rigorous demands of the healthcare industry. Ultimately, these principles empower manufacturers to enhance patient safety and satisfaction in a demanding healthcare landscape.
Understanding the dfm meaning is essential for medical device manufacturers seeking to implement Design for Manufacturing principles to enhance efficiency and reduce costs.
Real-world case studies illustrate the dfm meaning and demonstrate how DFM effectively enhances manufacturing processes. For instance, one study noted that effective DFM practices led to a 20% reduction in assembly components, further streamlining production and enhancing product reliability. Furthermore, cooperation with production engineers during the design phase has proven crucial for optimizing DFM results. The integration of DFM principles is not merely beneficial; it is imperative for maintaining a competitive edge in the evolving medical device landscape.
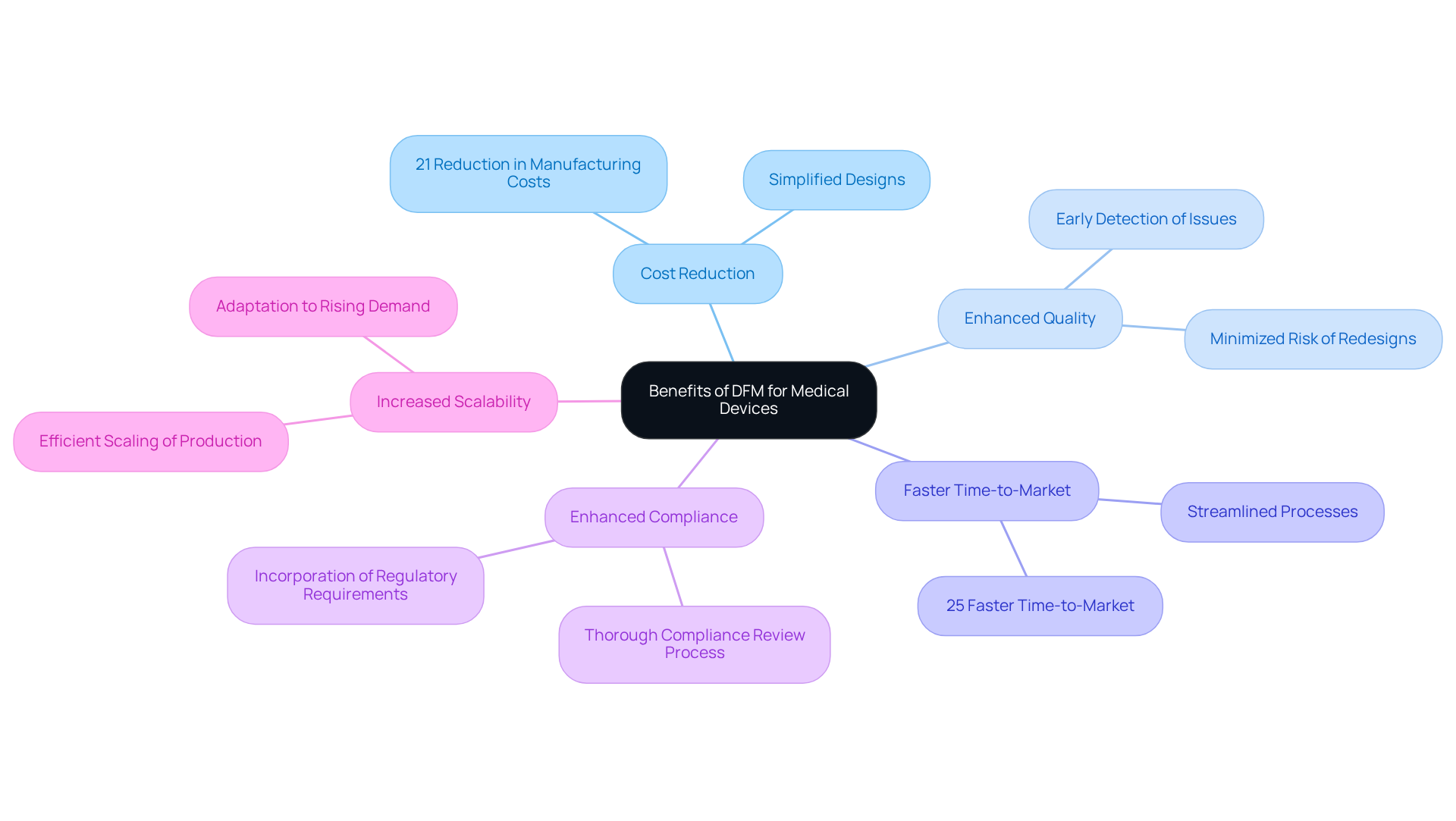
For medical device manufacturers, understanding Design for Manufacturing (DFM) is essential to overcoming production challenges and meeting regulatory standards. Focusing on simplification and optimization during the design phase allows manufacturers to enhance efficiency, lower costs, and provide safer, higher-quality products.
Throughout the article, we have explored key insights into DFM, including its core principles such as simplicity, standardization, and modularity. These principles facilitate smoother manufacturing processes and aid in the early detection of design flaws, thereby minimizing the risk of costly recalls. The benefits of adopting DFM practices are clear, with manufacturers experiencing reduced production costs, faster time-to-market, and improved compliance with regulatory requirements.
In conclusion, embracing DFM is not just a strategic advantage but a necessity for medical device manufacturers in a competitive landscape. By integrating DFM principles into their operations, companies can better navigate the complexities of product design and manufacturing, ensuring that they meet both market demands and regulatory standards. Integrating DFM principles is essential for manufacturers aiming to enhance product safety and meet the evolving demands of the medical device market.
What does Design for Manufacturing (DFM) mean?
Design for Manufacturing (DFM) refers to an approach that addresses the complexities of product design by prioritizing simplification and optimization from the outset, particularly in the medical equipment sector.
Why is DFM important in the medical equipment sector?
DFM is crucial in the medical equipment sector because it ensures compliance with regulatory standards and meets market demands by incorporating production considerations early in the design process.
What are the benefits of adopting a DFM strategy?
The key advantages of adopting a DFM strategy include increased production efficiency, minimized waste, and enhanced supply chain resilience.
How does DFM contribute to cost savings and product quality?
By identifying potential challenges early and streamlining workflows, DFM leads to significant cost savings and improved product quality.
What is the expected trend for DFM by 2026?
By 2026, the focus on DFM is expected to become more pronounced as manufacturers aim for enhanced quality and reliability, ensuring consistent and efficient production.
How does Voler Systems implement DFM in their strategy?
Voler Systems utilizes specific methodologies in their DFM strategy by concentrating on the early integration of production constraints and ongoing collaboration with stakeholders to ensure that items meet quality standards and market demands.
