Introduction
The complexity of medical device development presents significant challenges that require a strategic approach to navigate effectively. A thorough understanding of product development steps is crucial for ensuring compliance and meeting user needs, which ultimately enhances patient care. The complexity of regulatory compliance and user needs creates significant challenges for development teams. Failure to address these challenges can lead to costly delays and compromised patient care.
Understand the Fundamentals of Product Development Plans for Medical Devices
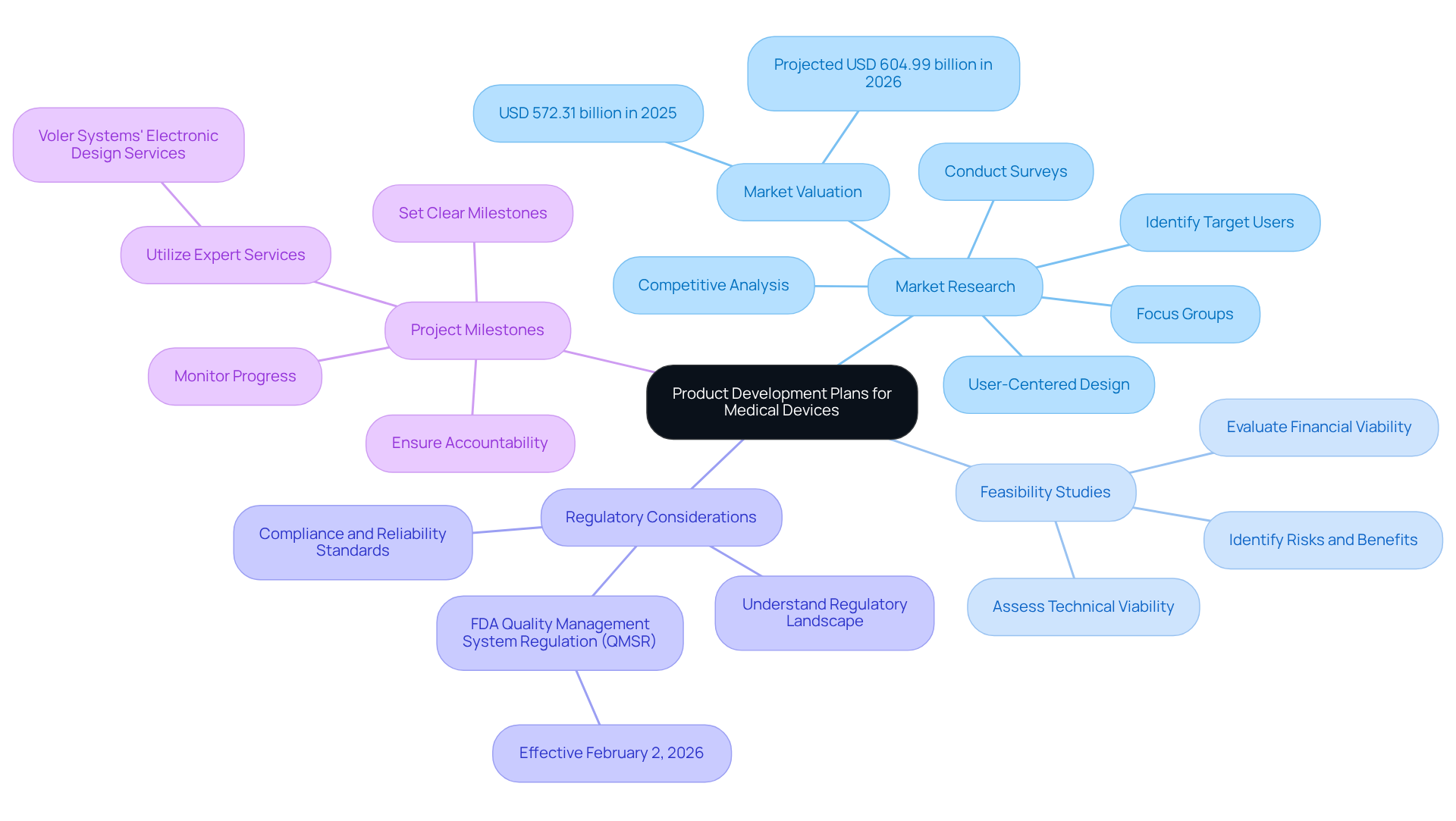
Navigating the complexities of healthcare equipment development requires a deep understanding of product development plans. These strategies serve as blueprints outlining the goals, methods, and timelines necessary for product development plans to transition healthcare products, such as heart pumps or liquid biopsy platforms, from concept to market. Key elements include:
- Market Research: Identifying target users and understanding their needs is critical. Conduct surveys, focus groups, and competitive analysis to gather insights. The global medical equipment market was valued at USD 572.31 billion in 2025 and is expected to expand to USD 604.99 billion in 2026, highlighting the significance of user-centered designs in medical tools.
- Feasibility Studies: Assess the technical and financial viability of the proposed device. This includes evaluating potential risks and benefits.
- Regulatory Considerations: Familiarize yourself with the regulatory landscape, including the FDA's Quality Management System Regulation (QMSR), effective February 2, 2026. Adhering to these stringent standards is crucial for successful product creation. Voler Systems' expertise in navigating these regulations ensures that your products meet necessary compliance and reliability standards.
- Project Milestones: Set clear milestones to monitor progress and ensure accountability throughout the creation process. Utilizing Voler Systems' expert electronic design services can expedite the creation of wearable and IoT solutions, ensuring that your project remains on course and benefits from innovative, AI-assisted engineering strategies.
By mastering these fundamentals, you position yourself to develop products that not only meet industry standards but also resonate with user needs, ultimately enhancing patient care.
Identify the Key Stages in Product Development Planning
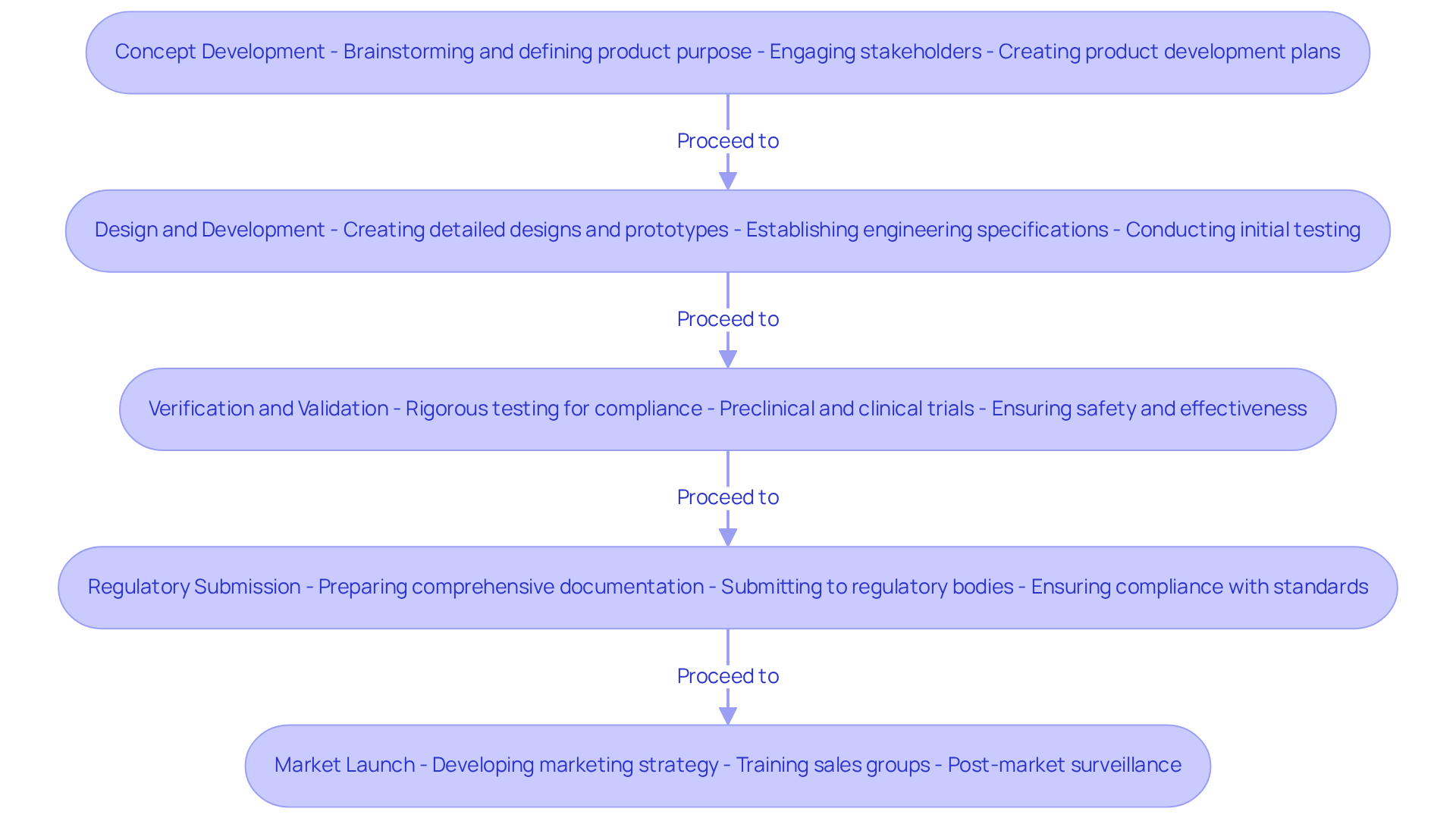
The product development process for medical devices is a structured journey that guides teams from initial concept to market launch, ensuring compliance and effectiveness at every stage:
- Concept Development: This foundational stage involves brainstorming and defining the product's purpose. Engaging stakeholders is crucial to refine the concept based on user needs and market demands. Thorough product development plans significantly shorten time to market and minimize costly errors, as successful teams prioritize careful planning.
- Design and Development: In this phase, detailed designs and prototypes are created. This includes establishing engineering specifications, user interface design, and conducting initial testing to validate concepts. Rapid prototyping techniques, such as 3D printing, facilitate faster iterations, allowing teams to identify design flaws early.
- Verification and Validation: Rigorous testing is conducted to ensure the apparatus meets design specifications and regulatory requirements. Achieving compliance with design specifications and regulatory requirements is a challenging endeavor that requires rigorous testing. This includes preclinical and clinical trials, which are essential for demonstrating safety and effectiveness. Verification confirms that the apparatus is constructed correctly, while validation ensures it meets real-world use conditions.
- Preparing and submitting comprehensive documentation to regulatory bodies is critical for the approval of product development plans. This submission must include descriptions of the equipment, intended use, risk management files, and testing data, ensuring compliance with standards such as ISO 13485 and FDA regulations. Startups benefit from tailored compliance documentation support, which is crucial for navigating regulatory complexities. Review timelines can vary significantly, with FDA 510(k) reviews averaging several months.
- Market Launch: Developing a robust marketing strategy and preparing for product launch are vital. This encompasses training sales groups and ensuring supply chain preparedness to meet market demands. Post-market surveillance is also essential for monitoring product performance and compliance after launch.
By adhering to these structured phases, teams not only mitigate risks but also position themselves for success in a highly competitive healthcare market.
Incorporate Regulatory Compliance and Quality Assurance into Your Plan
Integrating regulatory compliance and quality assurance into the medical device development process is crucial for success. Here’s how to effectively incorporate these elements into your product development plan:
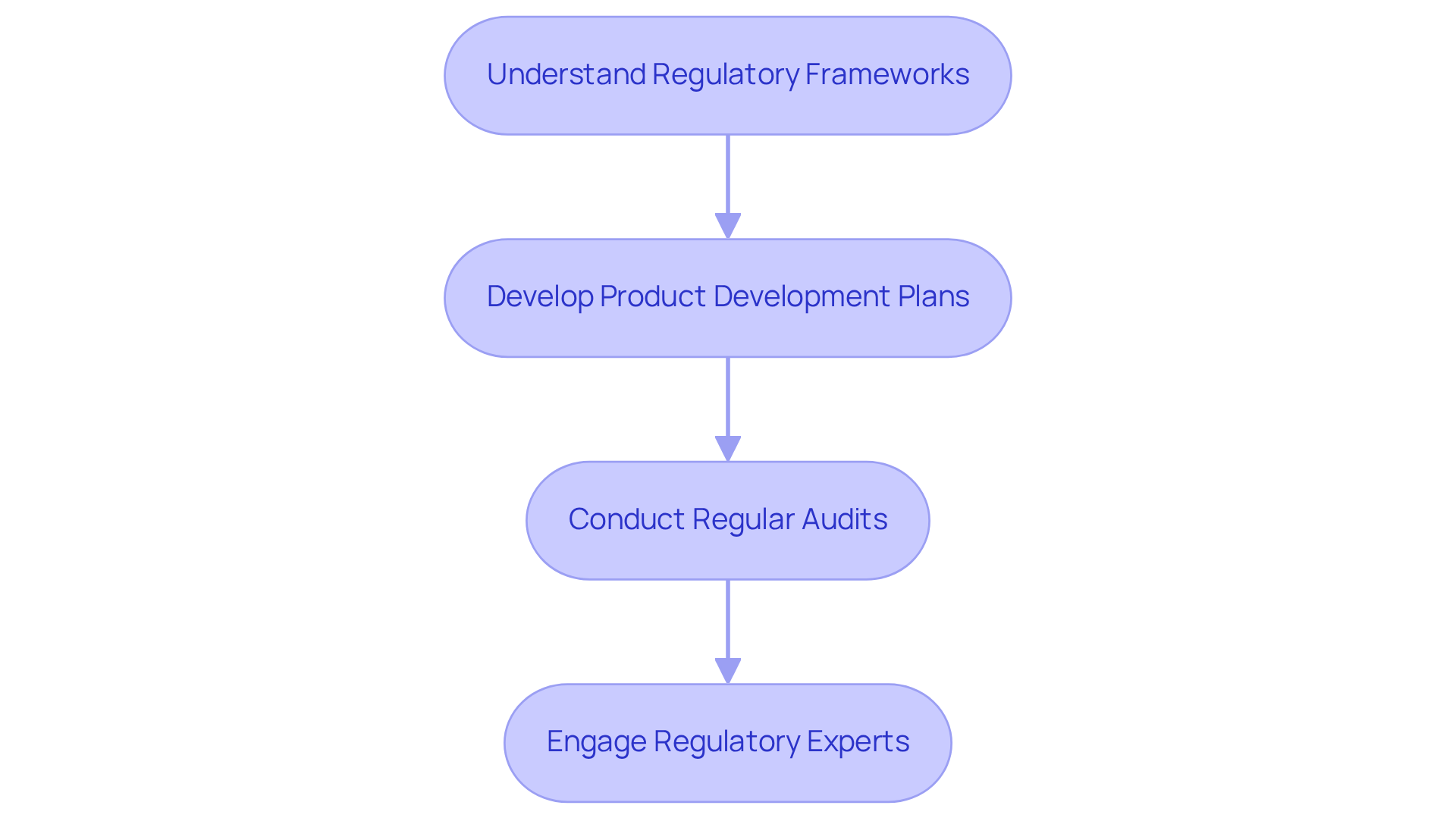
- Understand Regulatory Frameworks: Familiarize yourself with critical components of medical device development, including FDA guidelines and ISO 13485. This knowledge informs design and testing, ensuring compliance with industry standards.
- Develop product development plans: Establish comprehensive product development plans that outline procedures for quality control, risk management, and documentation. This system should be integrated into every stage of product development plans, fostering a commitment to quality and compliance throughout the organization.
- Conduct Regular Audits: Implement a schedule for internal audits to verify adherence to established protocols and identify areas for enhancement. Maintaining compliance can be challenging without regular checks and balances, allowing for timely corrective actions.
- Engage Regulatory Experts: Collaborate with regulatory affairs specialists to navigate the complexities of submission processes. Their expertise ensures that all documentation is thorough and accurate, facilitating smoother interactions with regulatory bodies.
This strategic integration not only enhances product reliability but also streamlines the path to regulatory approval, ultimately safeguarding patient health.
Foster Collaboration Among Cross-Functional Teams
Effective collaboration among cross-functional teams is crucial for the successful development of medical devices. Here are key strategies to enhance teamwork:
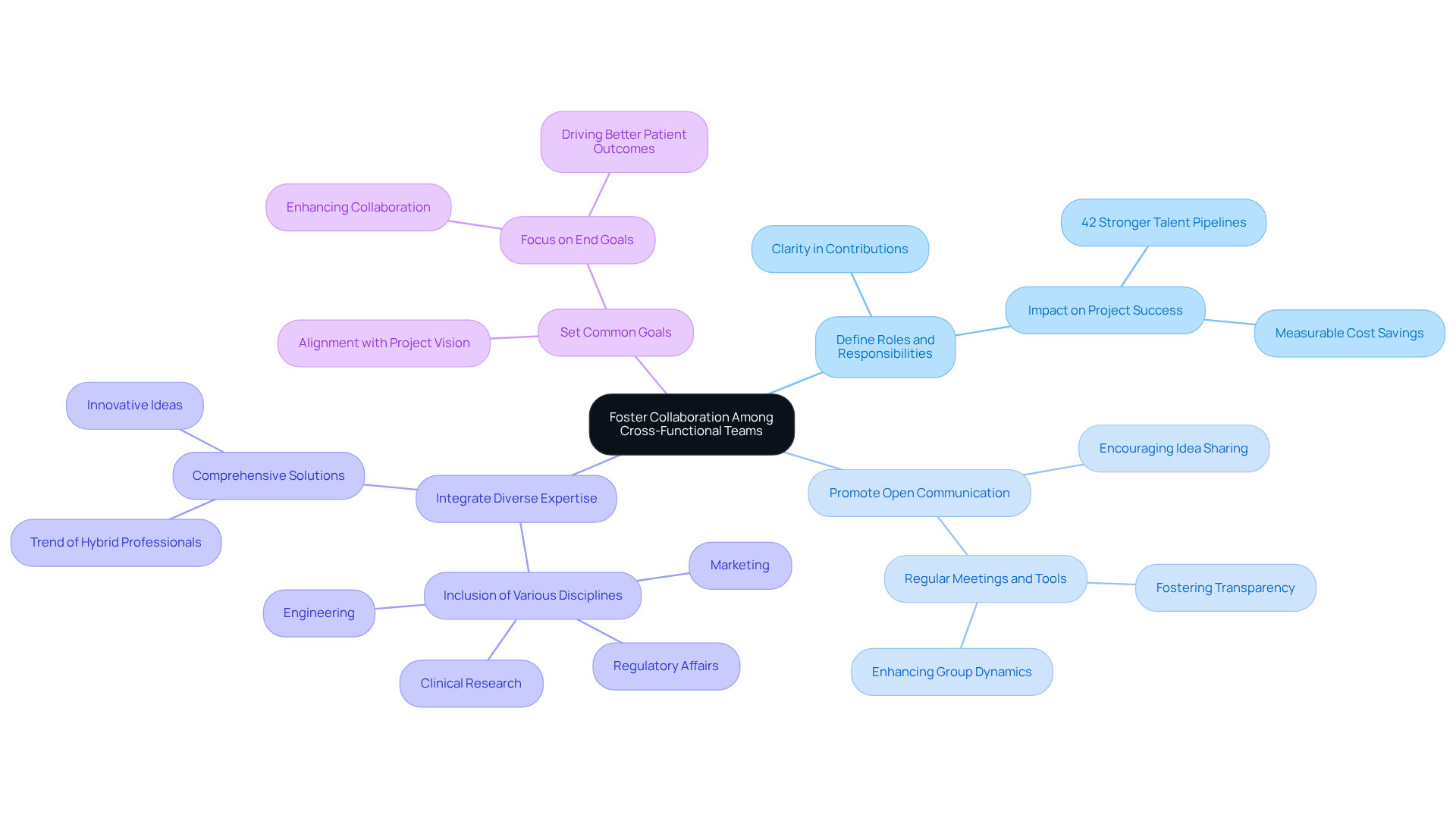
- Define Roles and Responsibilities: Clearly delineate the roles of each team member to ensure everyone understands their contributions. This clarity is vital; defined roles significantly impact project success, with organizations using cross-functional teams reporting 42% stronger talent pipelines and measurable cost savings.
- Promote Open Communication: Establish a setting where members feel at ease sharing ideas and feedback. Regular meetings and collaborative tools enhance communication, fostering transparency and enhancing group dynamics.
- Integrate Diverse Expertise: Include members from various disciplines such as engineering, regulatory affairs, marketing, and clinical research. This diversity leads to more comprehensive solutions and innovative ideas, aligning with the trend of hybrid professionals in MedTech who possess both clinical and technical skills.
- Set Common Goals: Establish shared objectives that align with the overall project vision. This alignment helps bring people together and keeps everyone focused on the end goal, enhancing collaboration and driving better patient outcomes.
By implementing these strategies, teams can effectively harness their collective expertise to overcome challenges and achieve project success.
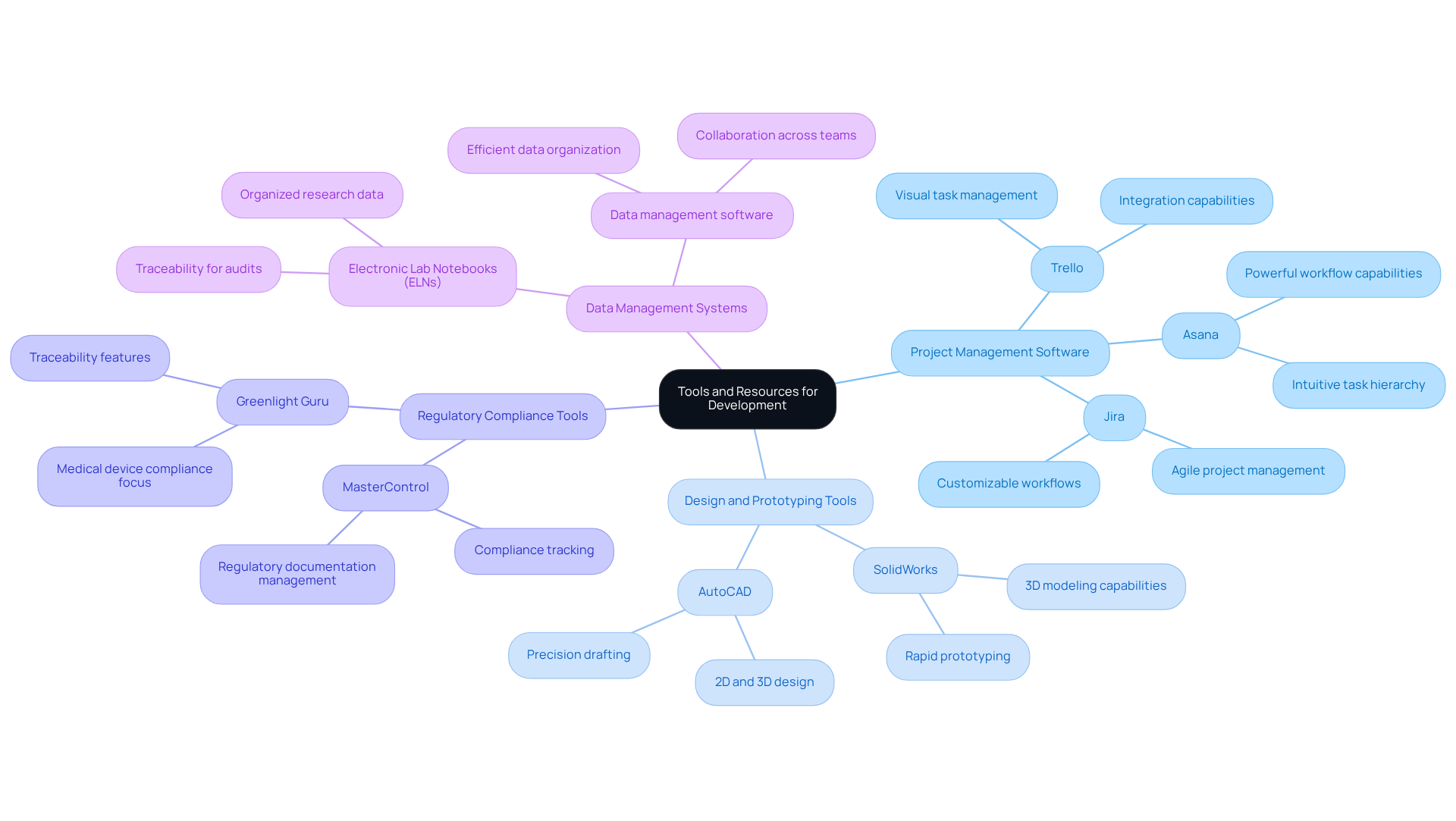
The medical device development process can be complex and fraught with challenges, making the right tools essential for success. Consider integrating the following tools and resources to enhance efficiency:
- Project Management Software: Utilize platforms like Trello, Asana, or Jira to effectively manage tasks, timelines, and team collaboration. These tools not only enhance accountability but also keep all team members aligned with project objectives, fostering a collaborative environment. For instance, Asana's intuitive task hierarchy and powerful workflow capabilities have proven beneficial in managing compliance and operational tasks in healthcare settings.
- Design and Prototyping Tools: Leverage advanced software such as SolidWorks or AutoCAD for design and prototyping. These tools facilitate rapid iteration and testing of design concepts, significantly impacting project timelines. In 2026, the incorporation of AI-driven features in these tools is anticipated to further improve design efficiency, enabling groups to adapt quickly to evolving requirements.
- Regulatory Compliance Tools: Implement software solutions like MasterControl or Greenlight Guru that assist with regulatory documentation and compliance tracking. These platforms simplify the submission process, ensuring adherence to industry standards and reducing the risk of delays. Companies utilizing these tools have reported improved compliance rates and faster approval times.
- Data Management Systems: Employ electronic lab notebooks (ELNs) and data management systems to organize research and testing data efficiently. This ensures easy access and traceability, which are critical for maintaining compliance and facilitating audits. The use of structured data management has been shown to enhance project visibility and coordination across teams.
Ultimately, the strategic integration of these tools into product development plans can transform the development landscape, enabling companies to bring innovative medical devices to market more swiftly and efficiently.
Conclusion
Navigating the complexities of healthcare innovation requires a mastery of product development plans for medical devices. Understanding the key components of these plans enables stakeholders to turn concepts into market-ready solutions that comply with regulations and meet user needs, thereby enhancing patient care.
Throughout the article, critical steps such as:
- Conducting thorough market research
- Engaging in feasibility studies
- Ensuring regulatory compliance
have been emphasized as essential for successful medical device development. The importance of structured stages - from concept development to market launch - has been highlighted, illustrating how each phase contributes to a cohesive and efficient product development process. Additionally, collaboration among cross-functional teams, supported by data analytics and project management tools, can significantly improve project outcomes, streamline workflows, and mitigate risks.
In light of these insights, it is clear that a strategic approach to product development planning is vital for any organization aiming to thrive in the competitive medical device sector. Ultimately, organizations that prioritize strategic planning will not only enhance their market position but also contribute to significant advancements in patient care.
Frequently Asked Questions
What is a product development plan for medical devices?
A product development plan serves as a blueprint outlining the goals, methods, and timelines necessary to transition healthcare products from concept to market. It includes key elements like market research, feasibility studies, regulatory considerations, and project milestones.
Why is market research important in product development for medical devices?
Market research is critical for identifying target users and understanding their needs. It involves conducting surveys, focus groups, and competitive analysis to gather insights, which are essential for creating user-centered designs in medical tools.
What are feasibility studies in the context of medical device development?
Feasibility studies assess the technical and financial viability of a proposed medical device, evaluating potential risks and benefits to determine if the project should proceed.
What regulatory considerations should be taken into account for medical device development?
It is important to familiarize oneself with the regulatory landscape, including the FDA's Quality Management System Regulation (QMSR). Adhering to these standards is crucial for successful product creation and compliance.
What are project milestones in product development?
Project milestones are clear goals set to monitor progress and ensure accountability throughout the creation process. They help teams stay on track and measure success at various stages of development.
What are the key stages in the product development process for medical devices?
The key stages include: 1. Concept Development: Brainstorming and defining the product's purpose. 2. Design and Development: Creating detailed designs and prototypes. 3. Verification and Validation: Conducting rigorous testing to ensure compliance with design specifications and regulatory requirements. 4. Market Launch: Developing a marketing strategy and preparing for product launch, including post-market surveillance.
What is involved in the verification and validation stage?
This stage involves rigorous testing to ensure that the device meets design specifications and regulatory requirements. It includes preclinical and clinical trials to demonstrate safety and effectiveness, as well as ensuring compliance with standards like ISO 13485 and FDA regulations.
What documentation is required for regulatory approval?
Comprehensive documentation must be prepared and submitted to regulatory bodies, including descriptions of the equipment, intended use, risk management files, and testing data to ensure compliance with relevant standards.
How does post-market surveillance contribute to product development?
Post-market surveillance is essential for monitoring product performance and compliance after launch, allowing teams to address any issues that may arise and ensuring ongoing safety and effectiveness in the market.
List of Sources
- Understand the Fundamentals of Product Development Plans for Medical Devices
- 2026 Forecast: Medical manufacturing industry outlook (https://todaysmedicaldevelopments.com/article/2026-forecast-todays-medical-developments-industry-outlook)
- 5 Trends Shaping the Medical Device Industry in 2026 (https://ecinnovations.com/blog/medical-device-trends)
- MedTech Investment Trends & Predictions for 2026 (https://htdhealth.com/insights/medtech-investment-trends-predictions-for-2026)
- 2026 Medical Device Manufacturing: Optimism Amid Uncertainty (https://mddionline.com/manufacturing/2026-medical-device-manufacturing-outlook-optimism-grows-despite-lingering-uncertainty)
- The medical device industry outlook for 2026 | Medical Economics (https://medicaleconomics.com/view/the-medical-device-industry-outlook-for-2026)
- Identify the Key Stages in Product Development Planning
- Medical Product Development Trends - 2026 | IDP (https://innovativedesignproducts.com/medical-product-development-trends-2026)
- Top Medical Device Regulations to Watch in 2026 (https://xtalks.com/top-medical-device-regulations-to-watch-in-2026-4549)
- Medical Device Product Development Timeline (https://huimedicalcarts.com/blog/medical-device-development)
- MedTech Product Development Trends in 2026 (https://enlil.com/blog/medtech-product-development-trends-in-2026)
- Medtech trends 2026: From AI strategy to execution | ZS (https://zs.com/insights/medtech-trends-2026-industry-strategy-and-execution)
- Incorporate Regulatory Compliance and Quality Assurance into Your Plan
- FDA Aligns U.S. Medical Device Rules with Global Standards (https://mddionline.com/regulatory-quality/medical-device-companies-must-navigate-historic-fda-rule-change)
- FDA’s 2026 Guidance Expands Pathway for Low-Risk Digital Health Products—But Caution Remains Essential | Berkley Lifesciences (https://berkleyls.com/blog/fdas-2026-guidance-expands-pathway-low-risk-digital-health-products-caution-remains-essential)
- FDA Implements Quality Management System Regulation for Medical Devices (https://aabb.org/news-resources/news/article/2026/02/04/fda-implements-quality-management-system-regulation-for-medical-devices)
- Navigating New FDA Expectations Under QMSR (https://healthcarepackaging.com/industry-news/regulatory/article/22963491/navigating-new-fda-expectations-under-qmsr)
- Foster Collaboration Among Cross-Functional Teams
- Beyond the Operating Room: How MedTech Firms Are Building Multidisciplinary Teams for 2026 (https://mrinetwork.com/hiring-talent-strategy/medtech-multidisciplinary-teams-2026)
- Annual Benchmark Report Highlights the Need for Cross-Functional Team Collaboration, Technology & AI Investments – MedLearn Publishing (https://racmonitor.medlearn.com/annual-benchmark-report-highlights-the-need-for-cross-functional-team-collaboration-technology-ai-investments)
- Want to Drive Innovation? You Need Cross-Functional Collaboration (https://americanhealthcareleader.com/2025/drive-innovation-with-cross-functional-collaboration)
- The Winning Formula: Cross-Functional Collaboratio... (https://medicalaffairsspecialist.org/blog/the-winning-formula-cross-functional-collaboration-as-a-catalyst)
- Utilize Tools and Resources to Streamline Development
- 19 Best Healthcare Project Management Software in 2026 (https://thedigitalprojectmanager.com/tools/best-healthcare-project-management-software)
- 10 best healthcare project management software solutions in 2026 | The Jotform Blog (https://jotform.com/blog/healthcare-project-management-software)
- Top medtech trends to watch in 2026 (https://medtechdive.com/news/medtech-trends-outlook-2026/810902)
- Best medical device electronic Quality Management Systems (eQMS) 2026 (https://cognidox.com/blog/best-eqms-software-medical-device-2026)
- Best Healthcare Project Management Software in 2026 (https://celoxis.com/article/healthcare-project-management-software)






