Best Practices for PCB Layouts in Medical Device Design
Introduction The world of medical device design relies heavily on the precision of...

Achieving excellence in medical device design requires a multifaceted approach that includes:
This article explores best practices that can significantly enhance stackup efficiency, ensuring that devices are not only functional but also compliant and innovative. Given the rapid evolution of technology and regulatory landscapes, manufacturers must navigate these complexities effectively to deliver safe and effective medical solutions.
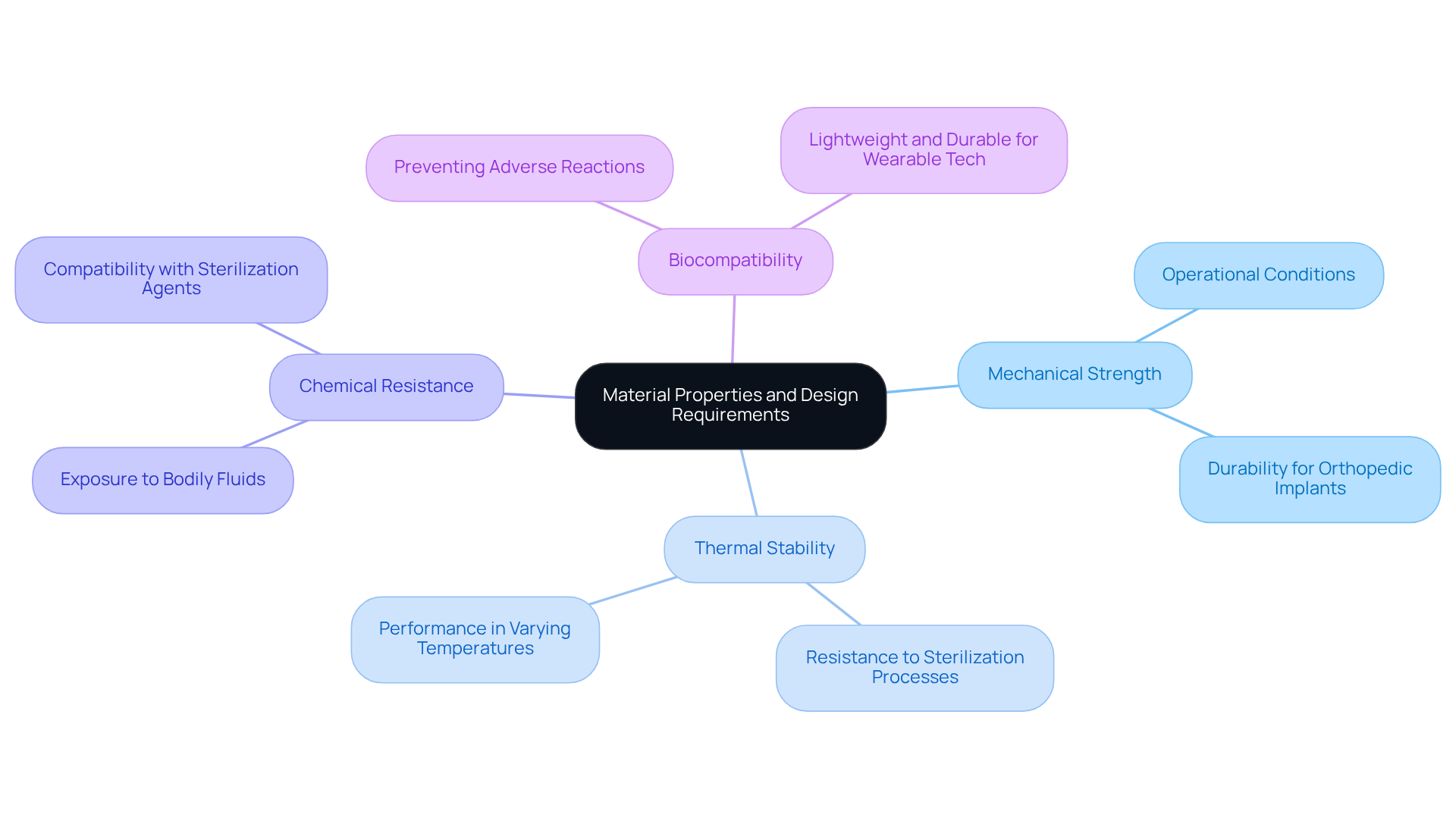
Achieving optimal stackup in the development of medical apparatus necessitates a thorough understanding of material properties. Key factors include:
For example, materials must endure the operational conditions of equipment, which frequently involve exposure to bodily fluids and rigorous sterilization processes. Furthermore, design requirements such as size constraints and electrical performance are critical. In the realm of wearable technology, materials must be both lightweight and durable, ensuring effective signal transmission.
A meticulous material selection process, which includes rigorous testing and validation, is essential to guarantee that the chosen materials perform reliably in real-world applications. Data indicates that mechanical strength and durability are vital for orthopedic implants, which must withstand daily stresses, while biocompatibility is crucial to prevent adverse biological reactions. By prioritizing these properties, manufacturers can enhance performance and ensure compliance with regulatory standards.
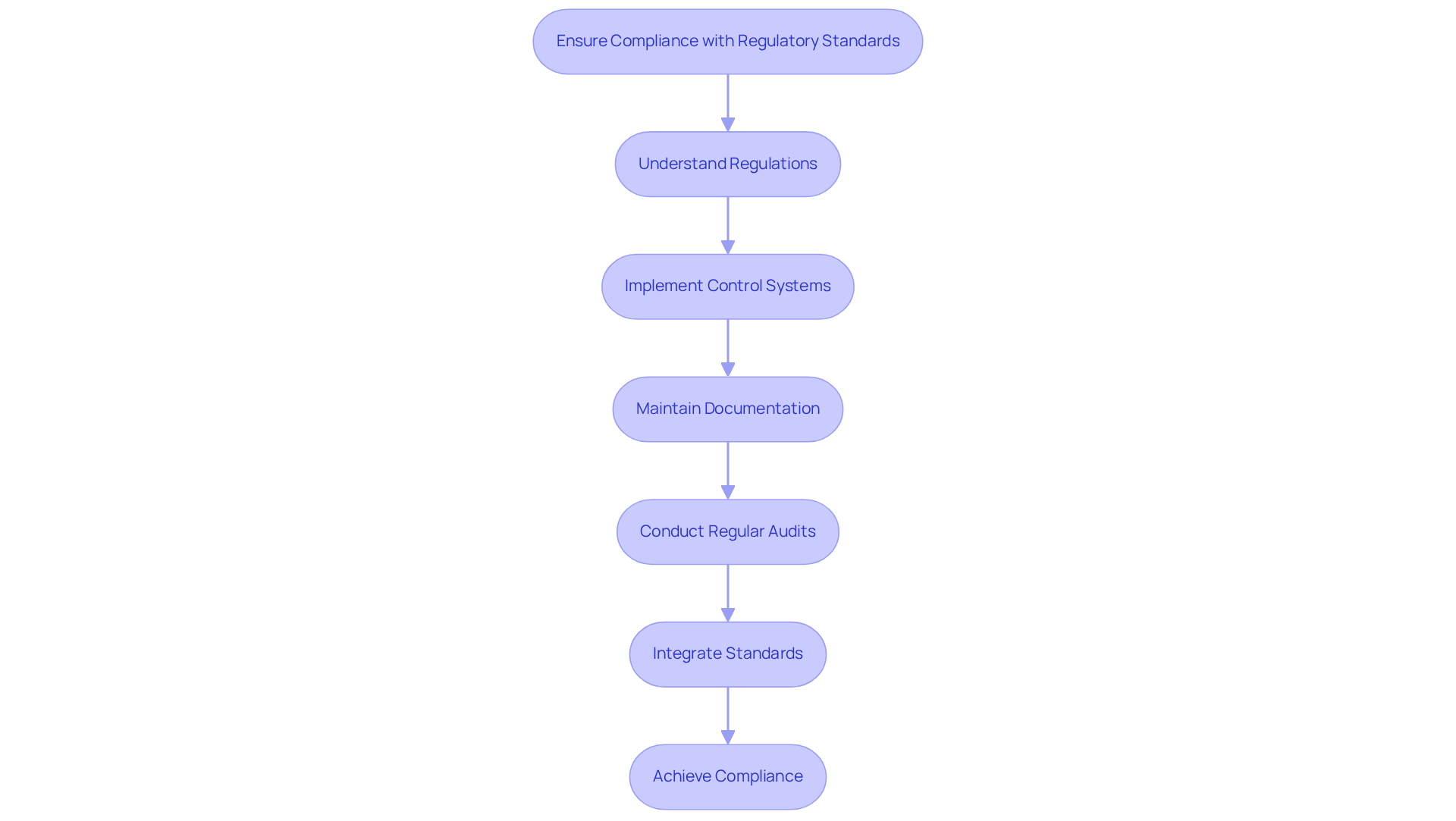
Adhering to regulatory standards is crucial in the development of medical devices. Producers must be well-versed in regulations such as the FDA's 21 CFR 820 and ISO 13485, which set forth criteria for quality management systems and control measures. A robust control system should include thorough documentation of inputs, outputs, verification, and validation activities. Regular audits and reviews are essential to ensure ongoing compliance.
For example, maintaining a comprehensive Design History File (DHF) that includes all design-related documentation can facilitate regulatory submissions and inspections. By prioritizing compliance from the outset, companies can significantly mitigate risks associated with non-compliance, such as costly recalls and legal repercussions. Recent trends show that manufacturers adhering to ISO 13485 are achieving higher compliance rates, highlighting the importance of integrating these standards into the development process. Successful implementation of ISO 13485 has been observed in various healthcare product firms, demonstrating its effectiveness in enhancing product quality and regulatory preparedness.
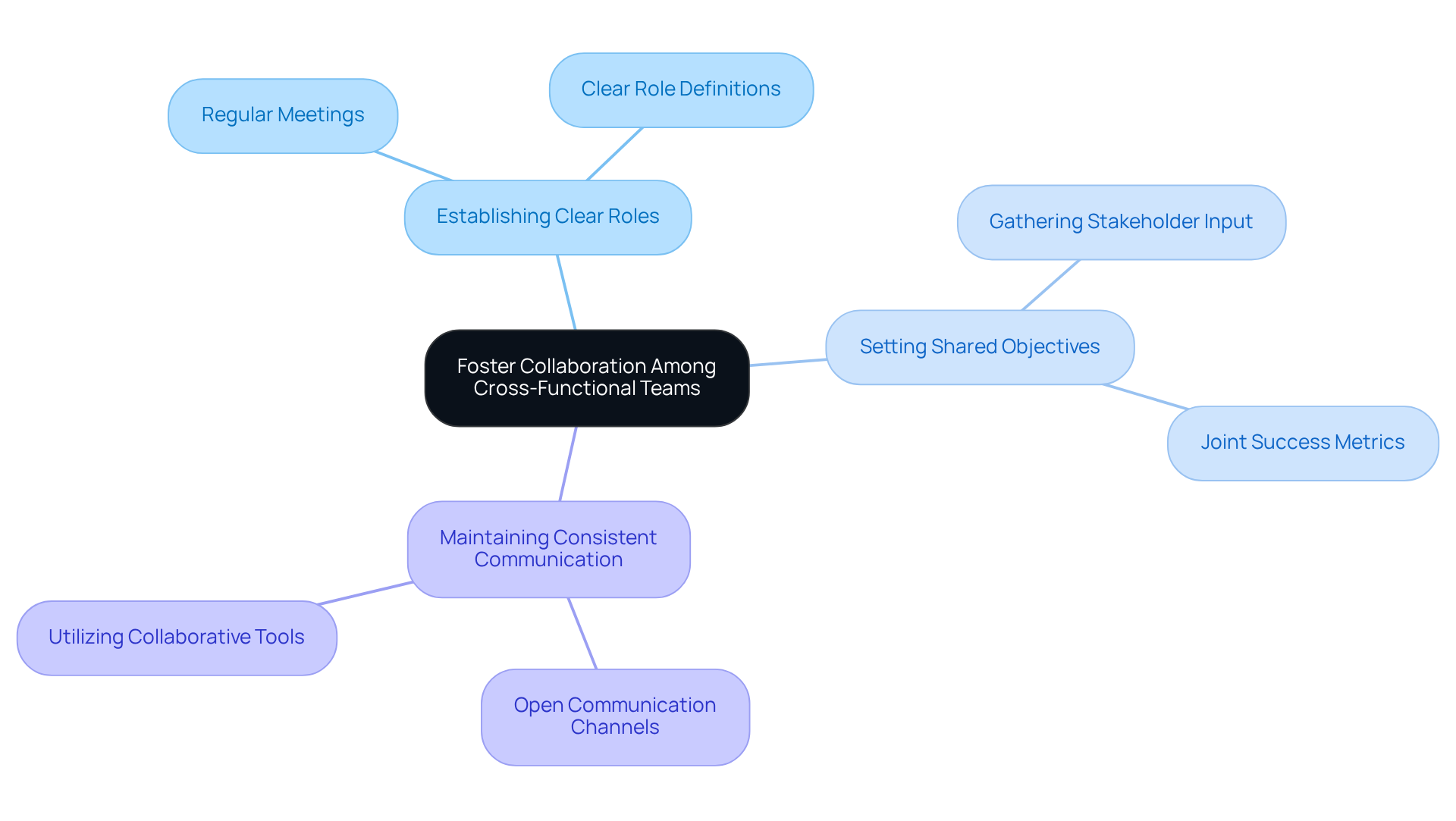
Effective collaboration among cross-functional teams is crucial for the success of medical device development. By integrating experts from engineering, regulatory affairs, quality assurance, and marketing, organizations can ensure that diverse perspectives are considered during the development phase. Regular meetings and open communication channels facilitate the exchange of ideas and feedback, fostering innovative solutions. Engaging regulatory specialists early in the planning phase allows teams to proactively identify potential compliance issues, thereby avoiding costly delays later on. Additionally, utilizing collaborative tools and platforms enhances coordination and streamlines workflows, resulting in a more efficient creative process.
Successful strategies for team collaboration include:
These practices collectively contribute to higher project success rates. Notably, healthcare organizations employing cross-functional teams for technology adoption have reported 42% stronger talent pipelines. Furthermore, the upcoming FDA's Quality Management System Regulation (QMSR), set to be implemented on February 2, 2026, underscores the importance of regulatory adherence in the development process.
Voler Systems exemplifies this approach through their innovative electronic device design projects, particularly in health-related, wearable, and IoT solutions. Their expertise in navigating compliance challenges supports startups in health technology, ensuring that interdisciplinary teamwork leads to successful outcomes. For instance, the partnership between Philips and Edwards on 'DeviceGuide AI' and Occam Design's collaboration with Duke University on the Porous Brain Infusion Catheter (PBIC) illustrate the effectiveness of interdisciplinary teamwork in achieving innovative healthcare solutions.
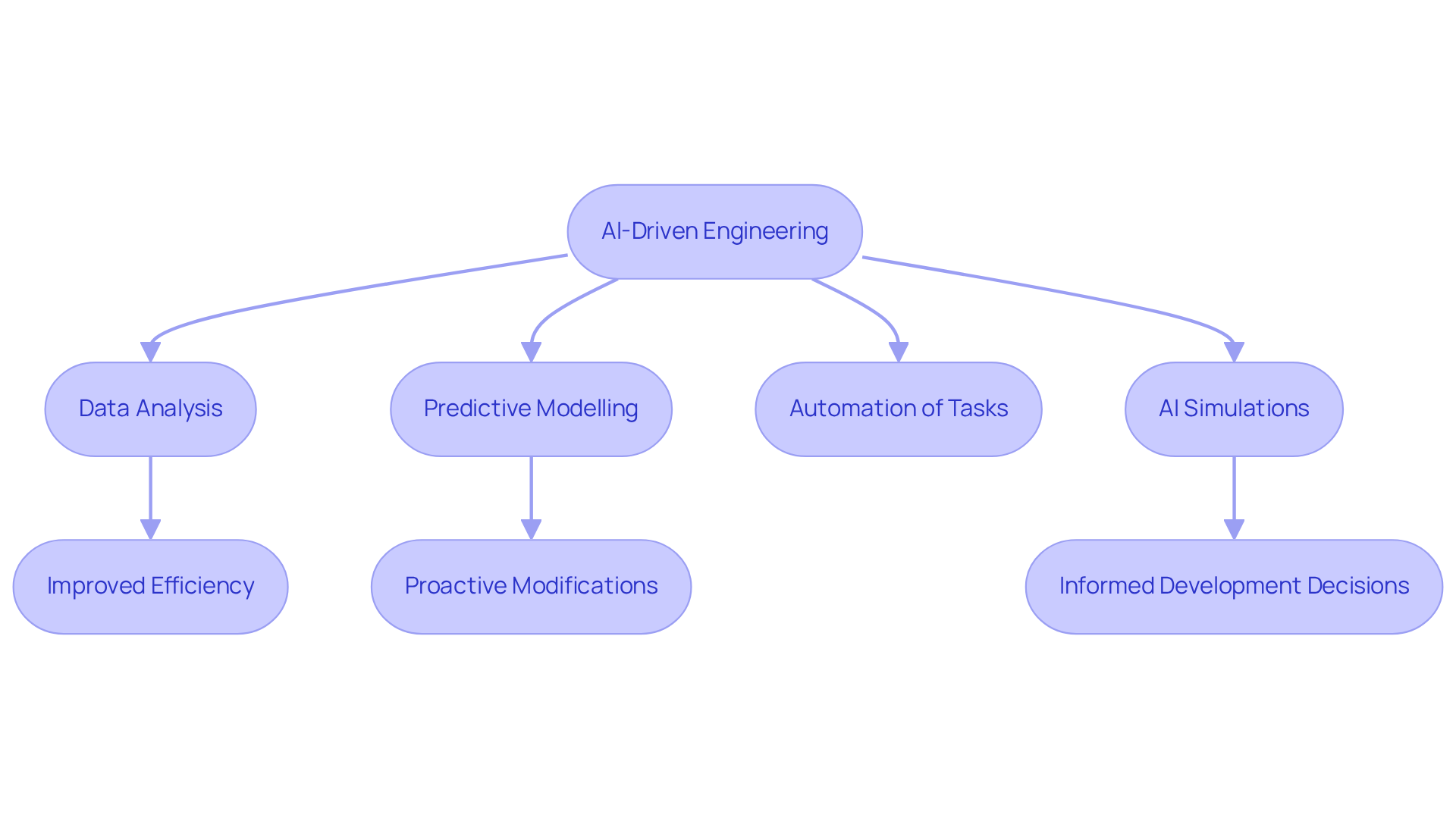
AI-driven engineering is revolutionizing the landscape of medical equipment creation by significantly enhancing both efficiency and precision. By leveraging Edge AI, creators at Voler Systems can employ machine learning algorithms to analyze extensive datasets, revealing patterns that improve key parameters. For instance, AI can predict potential failure points in equipment, allowing for proactive modifications prior to production. Additionally, AI tools automate repetitive tasks, enabling engineers to concentrate on more complex challenges associated with creation.
The implementation of AI-driven simulations offers valuable insights into device performance under varying conditions, leading to more informed development decisions. Companies that adopt AI technologies can anticipate shorter development timelines and improved product outcomes, aligning with contemporary trends in human-centered design and regulatory-ready development.
For further insights on electronic creation trends and success stories, please refer to our resources section. As highlighted by Naveen Shukla, integrating engineering and usability considerations early in the process can significantly enhance the likelihood of successful commercialization. Ultimately, the adoption of AI in medical device design positions companies for success in a rapidly evolving market.
Achieving effective stackup in medical device design is a complex endeavor that necessitates a thorough understanding of material properties, regulatory compliance, collaborative teamwork, and the integration of advanced technologies. The central theme underscores that a meticulous approach to these elements not only enhances the performance and safety of medical devices but also ensures compliance with the industry's stringent standards.
Key insights from the article emphasize the significance of selecting appropriate materials that provide mechanical strength, thermal stability, and biocompatibility. Furthermore, adhering to regulatory standards and promoting collaboration among cross-functional teams are essential strategies for navigating the complexities inherent in medical device development. The incorporation of AI-driven engineering further highlights the potential for increased efficiency and precision, empowering teams to innovate while ensuring compliance.
In conclusion, the successful development of medical devices relies on a holistic approach that prioritizes material selection, regulatory adherence, collaborative efforts, and the adoption of cutting-edge technologies. Embracing these best practices positions organizations to create safer and more effective medical solutions while fostering a culture of innovation that can lead to groundbreaking advancements in healthcare. By committing to these principles, stakeholders can significantly enhance the trajectory of medical device design, ultimately benefiting both patients and healthcare providers.
What are the key material properties important for medical apparatus development?
The key material properties include mechanical strength, thermal stability, chemical resistance, and biocompatibility.
Why is mechanical strength important in medical devices?
Mechanical strength is vital because medical devices, such as orthopedic implants, must withstand daily stresses and operational conditions, including exposure to bodily fluids and sterilization processes.
How does thermal stability affect medical apparatus?
Thermal stability ensures that materials can maintain their properties and performance under varying temperature conditions encountered during use and sterilization.
What role does chemical resistance play in material selection for medical devices?
Chemical resistance is crucial as materials must endure exposure to various chemicals and bodily fluids without degrading or losing functionality.
Why is biocompatibility significant in the development of medical devices?
Biocompatibility is essential to prevent adverse biological reactions when materials come into contact with the human body, ensuring patient safety.
What are the design requirements that need to be considered in medical apparatus?
Design requirements include size constraints and electrical performance, particularly for wearable technology, which must be lightweight and durable for effective signal transmission.
What is the importance of a meticulous material selection process?
A thorough material selection process, including rigorous testing and validation, is essential to ensure that the chosen materials perform reliably in real-world applications.
How can prioritizing material properties enhance medical device performance?
By prioritizing properties like mechanical strength, durability, and biocompatibility, manufacturers can enhance performance and ensure compliance with regulatory standards.
