Master PCB Design Layout: Best Practices for Medical Devices
Introduction Mastering PCB design for medical devices is not merely a technical...

Crafting a Product Requirements Document (PRD) for medical devices is a crucial step that can significantly influence the success of a project within a highly regulated industry. This guide outlines the essential steps for writing an effective PRD, providing insights into how to clearly define objectives, gather comprehensive requirements, and structure the document for optimal clarity and usability. The challenge, however, lies in ensuring that the perspectives of all stakeholders are considered while adhering to regulatory compliance.
How can teams effectively balance these demands to produce a document that not only meets requirements but also fosters innovation?
To initiate the writing of a (PRD), it is crucial to clearly define its purpose. A PRD functions as a comprehensive guide that delineates the , functionalities, and constraints.
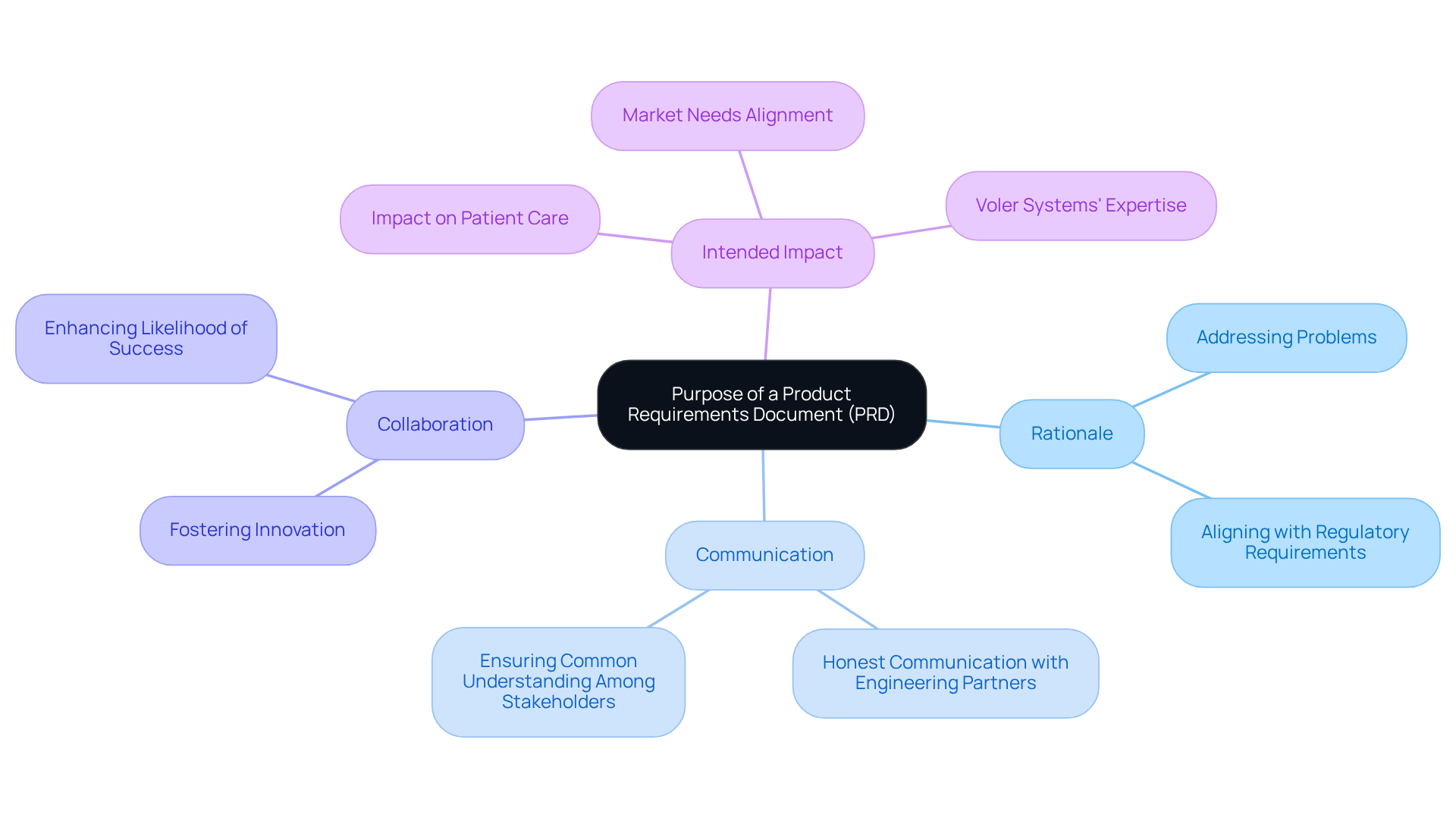
After establishing the purpose, the next critical step is to define specific goals and objectives for how to write a . Employing the - ensures that each goal is well-structured. For instance, a measurable objective could be to create a that accurately monitors heart rate with a precision of 95% within a six-month timeframe.
Moreover, integrating insights from successful engineering design initiatives, such as a reported 25% rise in , can further enhance clarity and focus. It is also essential to consider emerging trends in for s, which can lead to innovative solutions and improved efficiency.
Clearly is essential for understanding how to write a , as it serves as a and facilitates effective tracking of progress throughout the project lifecycle.
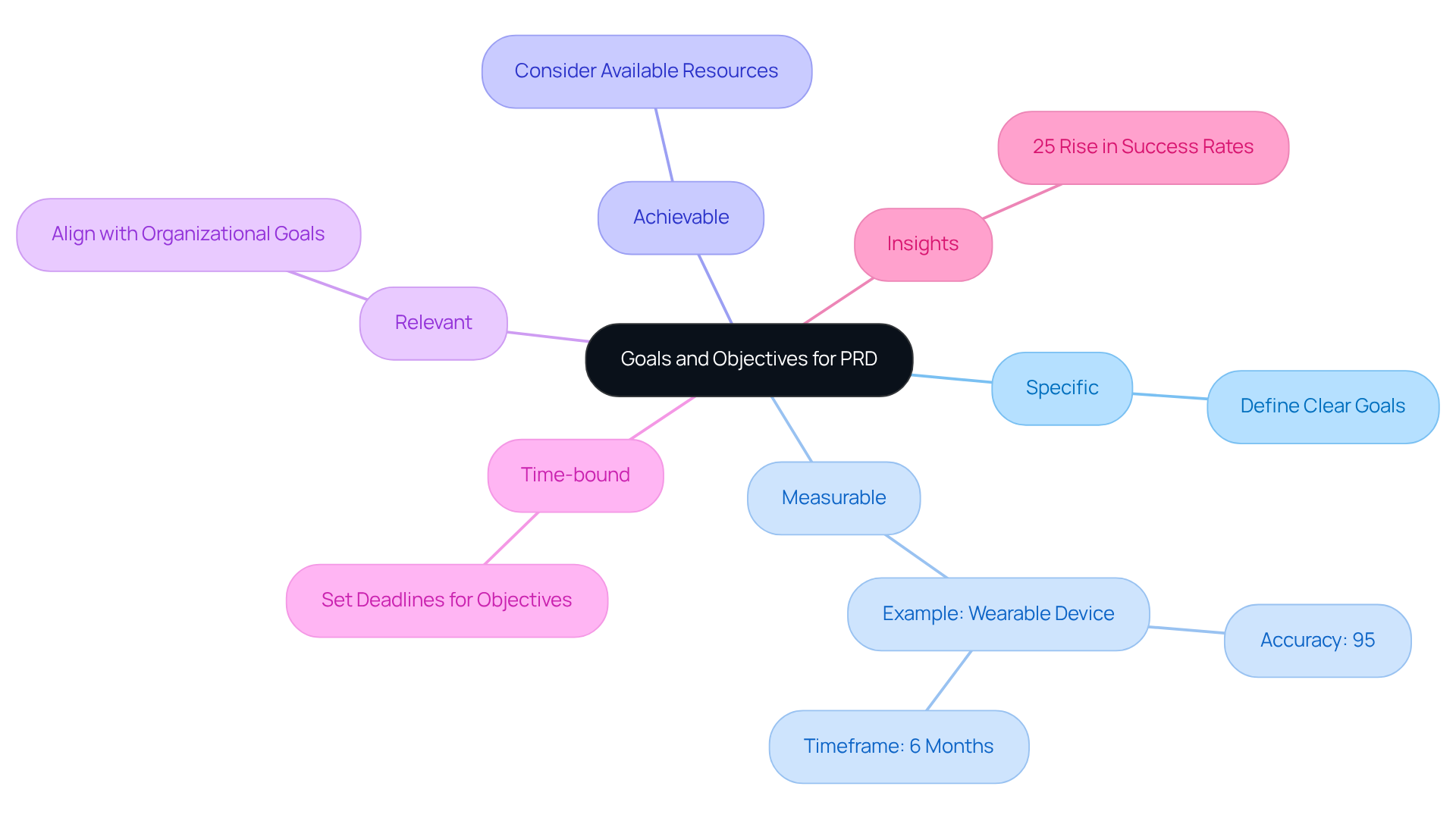
To effectively , - including engineers, designers, and end-users - is essential. Techniques such as interviews, surveys, and workshops should be employed to gather comprehensive input. This collaborative approach not only enhances the specifications but also fosters a sense of ownership among stakeholders.
It is important to document both , such as the capability of a to , and non-functional criteria, which may include . Clarity and precision in recording these specifications are vital, as they facilitate effective communication and execution throughout the development process.
Current trends indicate that can significantly enhance the efficiency of , ensuring that all voices are heard and considered.
To enhance the usability of your PRD, it is essential to adopt a . Begin with a title page that includes the project name, date, and authors. Following this, include a to facilitate easy navigation. Organize the document into such as:
Utilize to break down into manageable parts. Additionally, consider incorporating , such as diagrams or flowcharts, to illustrate processes or connections between needs. This will promote better understanding and collaboration among team members.
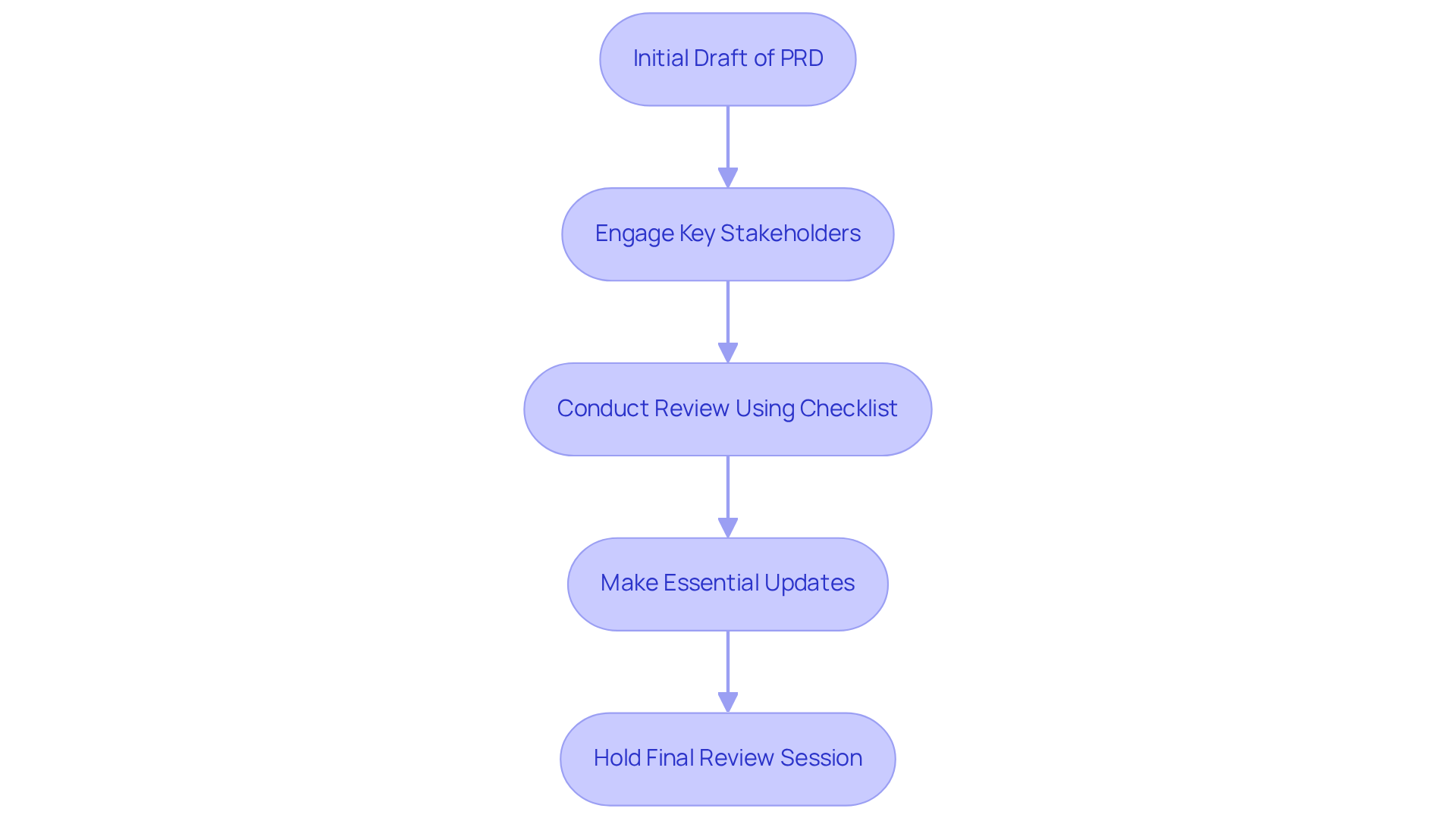
After the initial drafting of how to write a , a meticulous review is essential to ensure its accuracy and completeness. in this process is crucial, as their diverse perspectives can uncover inconsistencies, unclear language, or missing information that may otherwise go unnoticed. Specifications must be without ambiguity.
Utilizing a and objectives facilitates a , ensuring that all critical aspects are addressed. After making essential updates, it is recommended to hold a final review session with involved parties to ensure that the document meets their expectations and project objectives.
This collaborative approach enhances the quality and reliability of the PRD while fostering a culture of , which is vital in the fast-evolving landscape. Current trends highlight the incorporation of input from interested parties throughout the revision process, ensuring that the PRD remains relevant and compliant with regulatory standards.
By prioritizing , organizations can significantly , ultimately leading to more successful .
Crafting a Product Requirements Document (PRD) for medical devices is a crucial process that establishes the groundwork for successful product development. By clearly defining the purpose, goals, and requirements, stakeholders can ensure that the product not only complies with regulatory standards but also addresses genuine market needs. This comprehensive approach promotes collaboration and innovation, ultimately leading to enhanced patient care and outcomes.
The article delineates five essential steps for creating an effective PRD:
Each step underscores the importance of collaboration among engineers, designers, and end-users, reinforcing that a well-constructed PRD acts as a roadmap for the entire development team. Employing techniques such as SMART criteria and engaging stakeholders throughout the process improves the quality and relevance of the document.
In the rapidly evolving field of medical device development, the importance of a thorough and well-structured PRD cannot be overstated. It not only mitigates risks associated with miscommunication and oversight but also aligns the team toward a common goal. By prioritizing clear documentation and stakeholder involvement, organizations can pave the way for innovative solutions that meet the demands of the healthcare market, ultimately contributing to improved patient outcomes and advancing the field of medical technology.
What is the purpose of a Product Requirements Document (PRD)?
A PRD serves as a comprehensive guide that outlines the product's objectives, functionalities, and constraints, while also addressing the problems it aims to solve and how it aligns with regulatory requirements.
Why is communication important when creating a PRD?
Honest communication with engineering partners is vital to ensure that all stakeholders, including engineers, designers, and regulatory bodies, share a common understanding of the objectives, which fosters collaboration and enhances the likelihood of developing effective solutions.
What should be included in a PRD to highlight its significance?
A PRD should include a succinct overview of the product's intended impact on patient care or market needs, illustrating how the expertise in AI-driven engineering and ultra-low power solutions can contribute to successful outcomes.
How can goals and objectives be defined for a PRD?
Goals and objectives can be defined using the SMART criteria-Specific, Measurable, Achievable, Relevant, and Time-bound-to ensure they are well-structured and clear.
Can you provide an example of a measurable objective for a PRD?
An example of a measurable objective could be to create a wearable device that accurately monitors heart rate with a precision of 95% within a six-month timeframe.
What is the benefit of applying SMART goals in engineering design initiatives?
Applying SMART goals can enhance clarity and focus, with reports indicating a 25% rise in success rates when these goals are utilized.
Why is it important to document objectives in the PRD?
Clearly documenting objectives in the PRD is essential as it serves as a roadmap for the development team and facilitates effective tracking of progress throughout the project lifecycle.
