Introduction
Embedded technology is fundamentally altering the landscape of healthcare by enhancing the capabilities of medical devices for real-time monitoring and patient care. As the demand for innovative healthcare solutions grows, organizations must effectively harness embedded technology to meet stringent safety requirements while delivering innovative medical solutions.
However, organizations face considerable hurdles in aligning design processes with regulatory compliance and maintenance demands. Addressing these challenges is crucial for organizations aiming to innovate while ensuring compliance and safety in medical device development.
Understand Embedded Technology Fundamentals
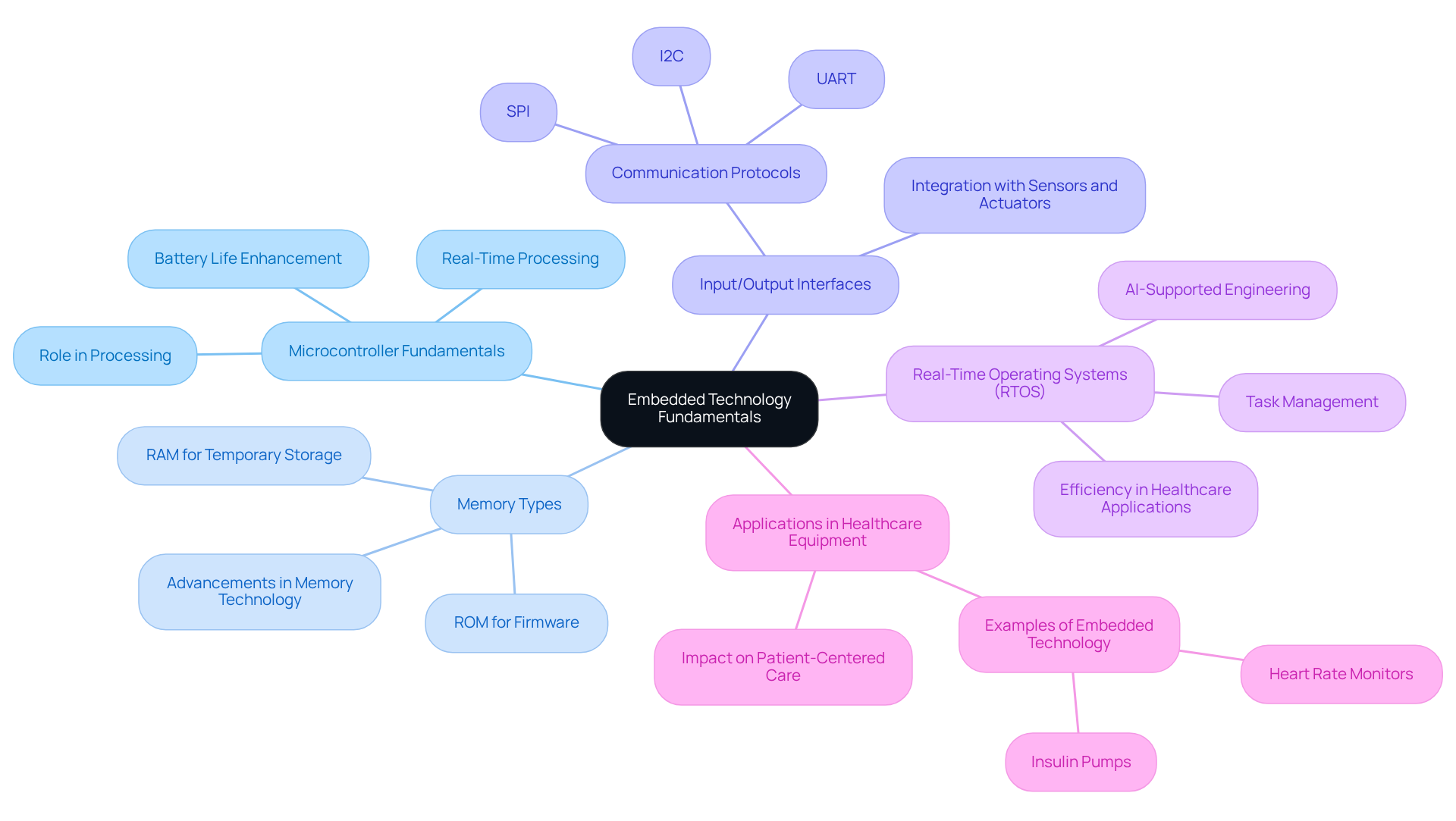
To excel in the field of embedded technology in medical equipment, it is essential to understand the core elements of embedded architectures. These setups typically comprise a microcontroller or microprocessor, memory (both RAM and ROM), and input/output interfaces.
- Microcontroller Fundamentals: Microcontrollers act as the brain of embedded systems, processing information and managing equipment. They carry out programmed instructions to perform specific tasks, making them essential for functionality in healthcare applications. Their real-time processing abilities allow prompt reactions according to patient requirements, which enhances the reliability and safety of the equipment through real-time processing. Voler Systems employs advanced microcontroller designs. This approach enhances battery life in wireless medical equipment, ensuring longevity and efficiency.
- Memory Types: Different types of memory are utilized in embedded systems. RAM is used for temporary data storage, while ROM stores firmware essential for device operation. Limited memory capacity can hinder the development of complex software applications, but advancements are tackling these constraints. These advancements allow for the development of more sophisticated healthcare applications. Voler Systems' expertise in software development ensures that memory constraints are effectively managed, facilitating compliance from prototype to production.
- Input/Output Interfaces: Embedded systems interact with the external environment through sensors and actuators. Familiarity with protocols like I2C, SPI, and UART is crucial for effective communication. These interfaces enable the integration of various communication protocols in healthcare equipment, utilizing embedded technology to support functionalities such as real-time health monitoring and data transmission. Voler Systems' design strategies enhance the reliability of these interfaces, contributing to the overall quality of healthcare devices.
- Real-Time Operating Systems (RTOS): RTOS play a critical role in managing hardware resources and ensuring timely execution of tasks, which is particularly important in healthcare applications where timing can be crucial. Automated processes can conduct tests that formerly required weeks in merely hours, improving efficiency and adherence to regulatory standards. Voler Systems employs AI-supported engineering to optimize RTOS integration. This guarantees that health-related equipment meets rigorous quality and reliability standards.
- Applications in Healthcare Equipment: Examining examples of embedded technology in healthcare equipment, such as heart rate monitors and insulin pumps, demonstrates how these components collaborate to ensure functionality and reliability. For example, the recall of Tandem Diabetes Care's Mobi insulin pumps because of software issues emphasizes the significance of software dependability and stringent quality assurance in healthcare products. Moreover, as Stephanie Pinteric from the 8-bit Microcontroller Business Unit mentions, microcontrollers featuring Core Independent Peripherals (CIPs) improve the performance and reliability of intelligent healthcare tools. The incorporation of microcontrollers in wearable technology enables continuous health monitoring, contributing to a shift towards patient-centered care that extends beyond traditional hospital environments. This commitment ensures that manufacturers can bring innovative healthcare solutions to market efficiently and effectively.
Identify Regulatory and Safety Requirements
Navigating the compliance landscape is essential for the successful design and implementation of embedded technology within medical devices. Following compliance standards ensures safety and paves the way for market entry. Here are the critical steps to follow:
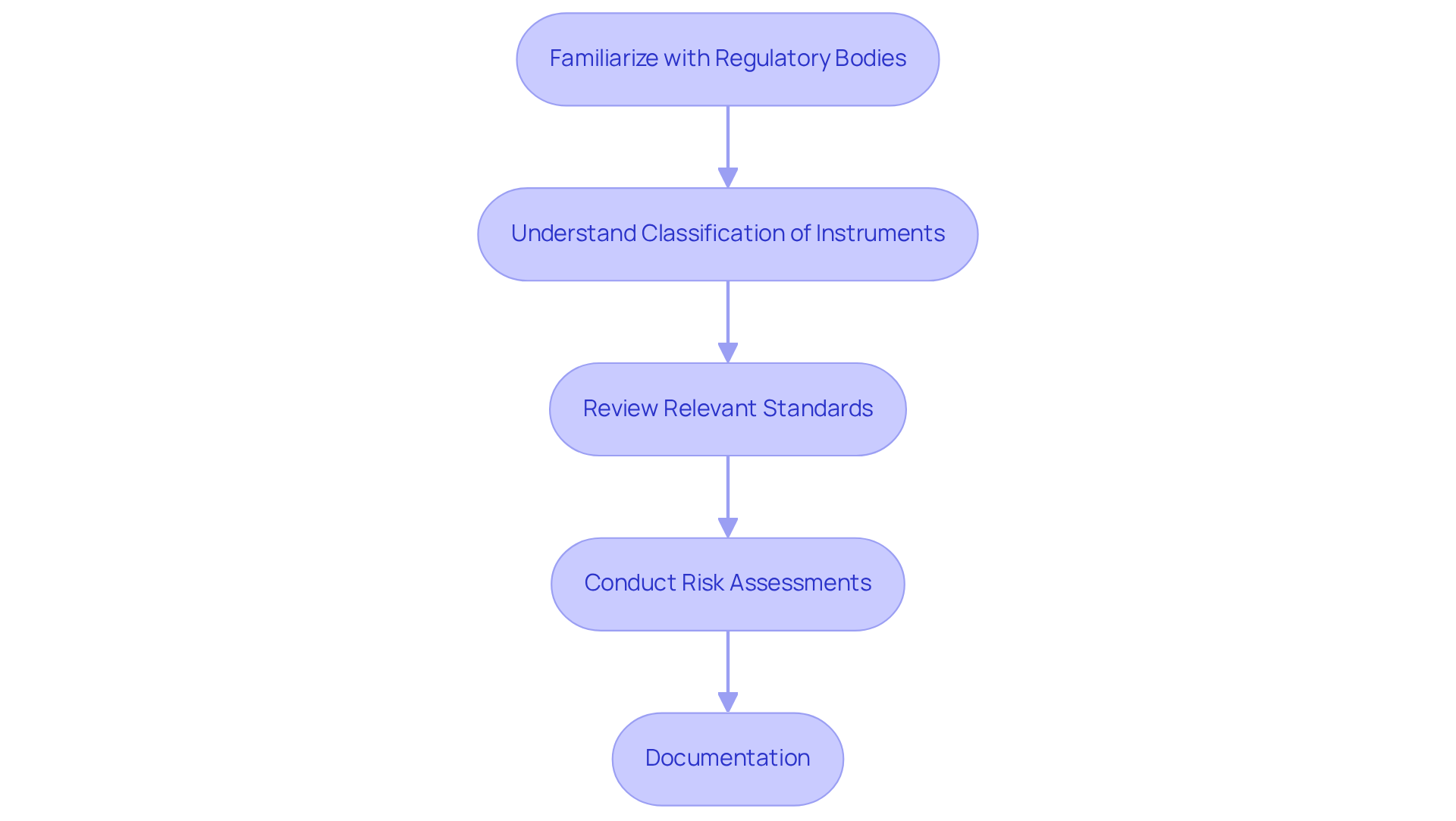
- Familiarize with Regulatory Bodies: Recognize the roles of key regulatory entities such as the FDA (U.S. Food and Drug Administration) and ISO (International Organization for Standardization). These organizations manage the approval and monitoring processes for medical instruments, ensuring they meet safety and efficacy standards.
- Understand Classification of Instruments: Medical instruments are classified into three categories: Class I, II, and III. Each class has distinct regulatory requirements, with higher-risk items (Class II and III) necessitating more rigorous testing and comprehensive documentation to demonstrate safety and effectiveness.
- Review Relevant Standards: Familiarity with standards such as ISO 13485, which outlines quality management systems, and IEC 60601, which addresses the safety and performance of medical electrical equipment, is crucial. These standards provide frameworks for ensuring product quality and compliance.
- Conduct Risk Assessments: Implement risk management processes as specified in ISO 14971. This involves identifying potential hazards associated with your device and developing strategies to mitigate these risks effectively, ensuring patient safety and device reliability.
- Documentation: Prepare comprehensive compliance submissions, including design history files, risk management files, and clinical evaluation reports. Ensuring that all documentation meets compliance expectations is vital for successful approval and market access.
Ultimately, neglecting compliance can jeopardize not only market access but also patient safety.
Design and Implement Embedded Systems
To successfully design and implement embedded technology in medical equipment, a structured approach that aligns with technological advancements and regulatory requirements is imperative, including documentation compliance support from Voler Systems. Here are the key steps:
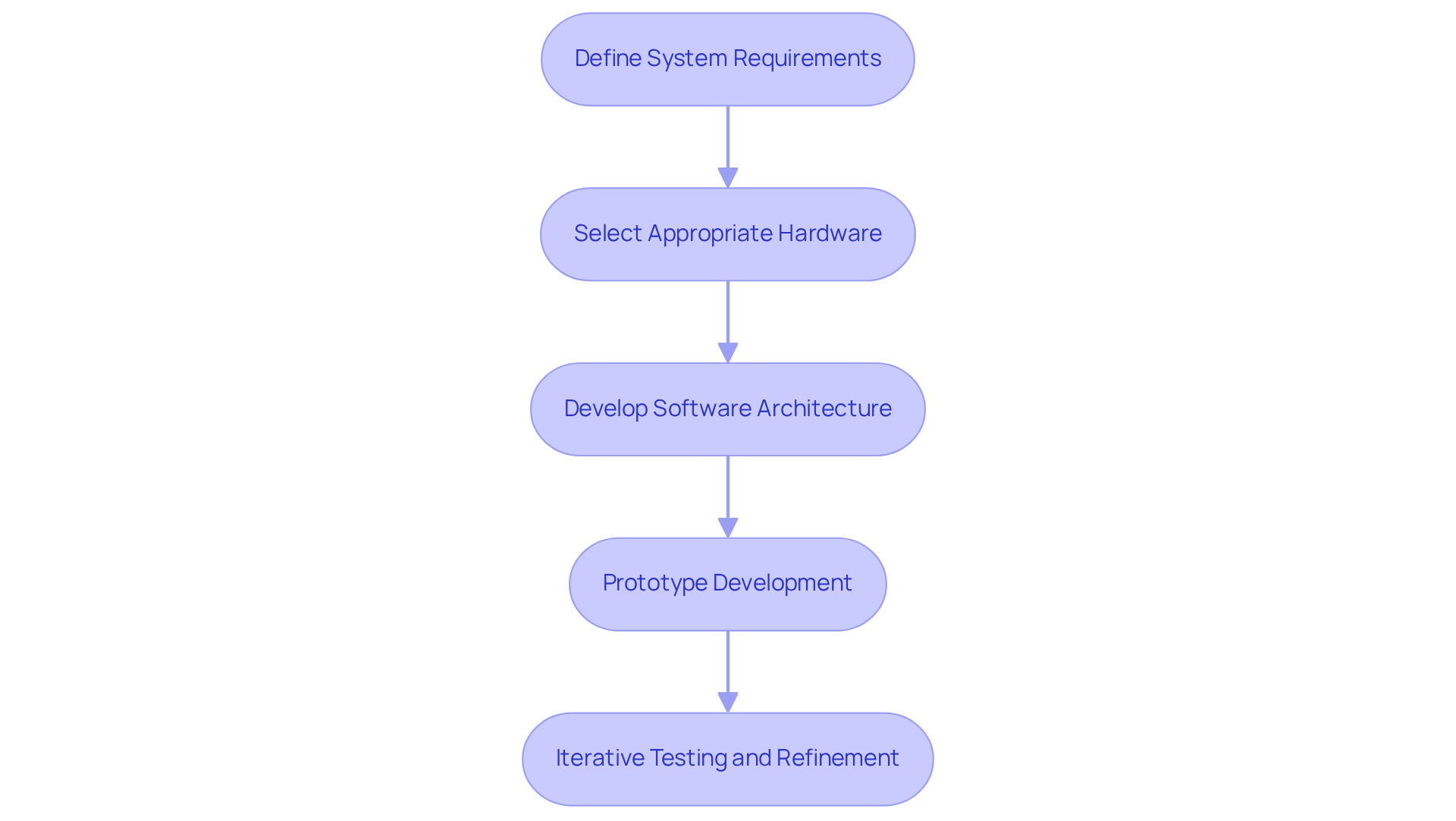
- Define System Requirements: Begin by clearly outlining the functional and non-functional requirements of your product. This should include performance metrics, user interface design, and power consumption specifications. These elements ensure the device meets the needs of both healthcare providers and patients.
- Select Appropriate Hardware: Choosing the right microcontroller or processor is critical. Factors to consider include processing power, memory capacity, peripheral support, and compliance with industry standards such as ISO 13485. The latest trends indicate a shift towards microcontrollers that utilize embedded technology for enhanced connectivity and security features, which are vital for modern medical applications.
- Develop Software Architecture: Establish a robust software architecture that defines the interactions between different components of your framework. This includes selecting an appropriate programming language and development environment that facilitate compliance with regulatory requirements and support future scalability.
- Prototype Development: Create a prototype of your embedded device using development boards and simulation tools. This phase enables early testing of design concepts and identification of potential issues prior to full-scale production. Utilizing Voler Systems' expertise in prototype development can enhance hardware design efficiency and facilitate necessary adjustments.
- Iterative Testing and Refinement: Implement iterative testing to uncover and resolve issues throughout the development process. Gathering feedback from testing phases is crucial for refining both hardware and software components. This method not only improves user satisfaction but also guarantees adherence to regulatory standards, ultimately resulting in a more dependable and efficient healthcare instrument.
By following these structured steps, manufacturers can mitigate risks associated with non-compliance and enhance the reliability of their medical devices with embedded technology.
Test and Validate Embedded Technology
Ensuring the safety and effectiveness of medical devices hinges on a rigorous testing and validation process. Follow these steps:
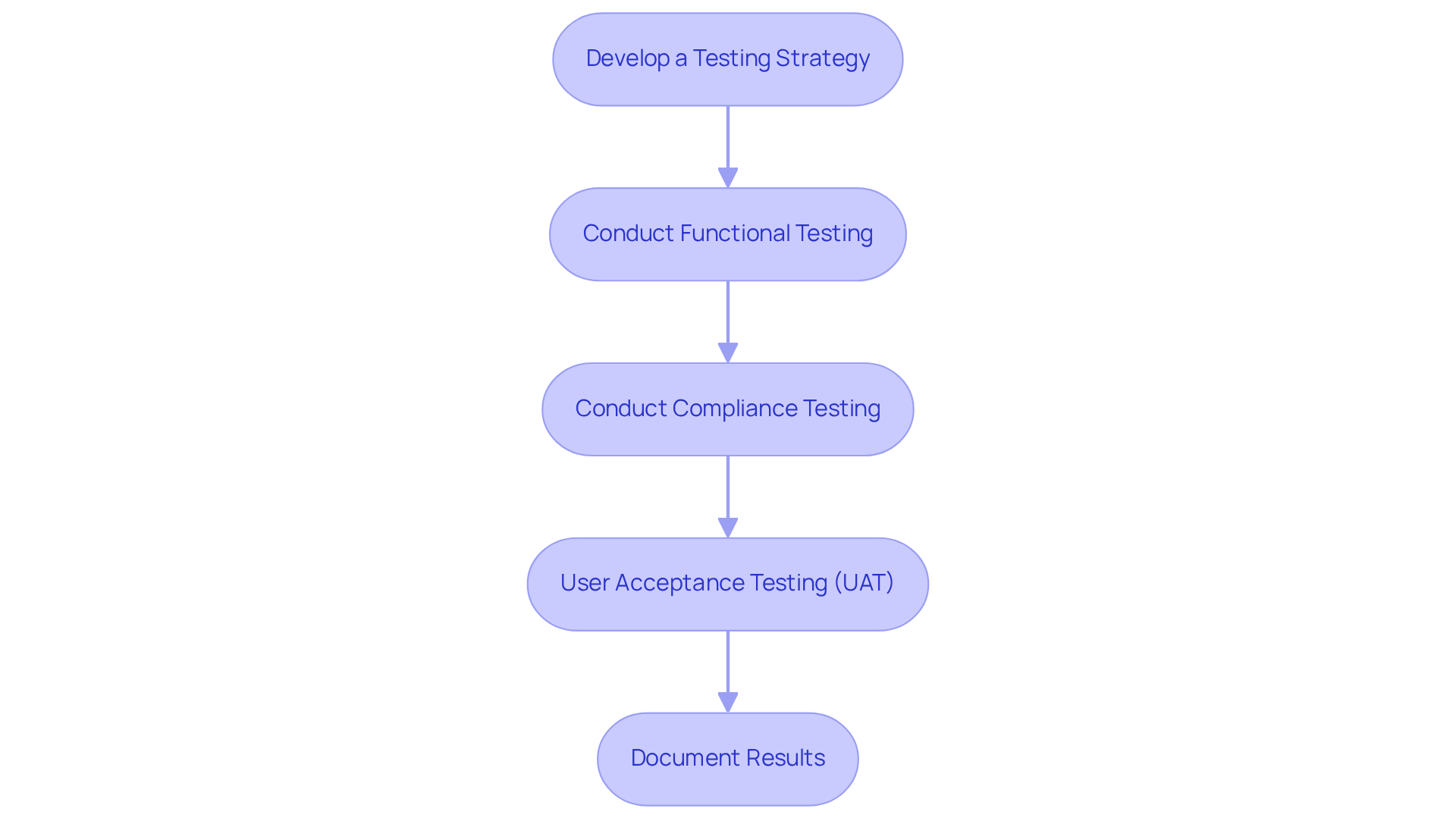
- Develop a Testing Strategy: Create a comprehensive testing strategy that encompasses unit testing, integration testing, and overall testing. Clearly define the criteria for success for each test to ensure thorough evaluation.
- Conduct Functional Testing: Verify that the system performs all intended functions correctly. This includes testing all inputs and outputs under various conditions to ensure reliability and performance.
- Conduct Compliance Testing: Verify that your equipment meets all applicable requirements by performing compliance testing against relevant standards, such as IEC 60601. This is crucial for demonstrating adherence to safety and performance benchmarks.
- User Acceptance Testing (UAT): Involve end-users in testing to gather feedback on usability and functionality. UAT is essential for verifying that the equipment meets user needs and expectations, ultimately enhancing patient safety and satisfaction.
- Document Results: Maintain detailed records of all testing activities, results, and any corrective actions taken. This documentation is essential for compliance submissions and serves as a reference for future development efforts.
Ultimately, thorough testing can be the difference between a successful product launch and potential safety issues.
Maintain and Update Embedded Systems
Ensuring the ongoing performance and compliance of embedded technology in medical devices is a critical challenge that requires systematic management. Follow these steps to effectively manage your systems:
- Establish a Maintenance Schedule: Develop a routine maintenance schedule that encompasses software updates, hardware inspections, and performance assessments to ensure optimal functionality. Integrating AI-driven engineering enhances the reliability of evaluations by providing data-driven insights.
- Monitor System Performance: Utilize advanced monitoring tools to track the real-time performance of your embedded technology systems. This proactive approach allows for early detection of potential issues, preventing critical failures. Voler Systems offers expertise in optimizing these processes, ensuring they remain adaptable to the latest technological advancements.
- Update Software and Firmware: Regularly implement software and firmware updates to mitigate security vulnerabilities, enhance functionality, and comply with the latest legal standards. Keeping software up to date is essential for preserving confidence in healthcare technology and ensuring patient safety, particularly in wireless medical equipment.
- Conduct Periodic Compliance Reviews: Regularly evaluate adherence to regulatory requirements to confirm that your product meets necessary safety and performance standards. Navigating the complexities of regulatory compliance can be daunting for many organizations. This is essential as regulations are constantly changing, and Voler Systems' expertise in creating compliant electronic products can offer valuable insights during these evaluations.
- Gather User Feedback: Continuously request input from users to pinpoint enhancement opportunities and ensure that the tool adapts to evolving requirements. Soliciting user feedback is essential for identifying improvement opportunities that enhance user experience and device performance. Voler Systems recommends utilizing specific tools and methodologies to streamline this feedback process.
Failure to implement these strategies could jeopardize patient safety and undermine the integrity of healthcare technology.
Conclusion
Mastering embedded technology in medical devices is essential for improving healthcare delivery and patient outcomes. This article has outlined essential steps that encompass:
- Understanding the fundamentals
- Navigating the intricate landscape of regulatory requirements
- Designing and implementing systems
- Rigorous testing
- Ongoing maintenance
Each phase is vital for creating reliable and efficient medical devices that meet safety standards and user needs.
Key insights include the importance of microcontrollers, memory management, and input/output interfaces in creating robust embedded systems. Understanding regulatory bodies and compliance standards ensures that devices not only meet legal requirements but also prioritize patient safety. Furthermore, a structured approach to design, testing, and continuous maintenance is vital for the longevity and effectiveness of medical technology.
As the healthcare landscape evolves, the integration of advanced embedded technology will play a significant role in shaping the future of medical devices. By adopting these strategies, the medical device industry can ensure that innovations not only advance technology but also prioritize patient safety and care quality.
Frequently Asked Questions
What are the core elements of embedded architectures in medical equipment?
The core elements of embedded architectures typically comprise a microcontroller or microprocessor, memory (both RAM and ROM), and input/output interfaces.
What role do microcontrollers play in embedded systems?
Microcontrollers act as the brain of embedded systems, processing information and managing equipment. They execute programmed instructions to perform specific tasks, enhancing reliability and safety through real-time processing.
How does Voler Systems enhance microcontroller designs?
Voler Systems employs advanced microcontroller designs that improve battery life in wireless medical equipment, ensuring longevity and efficiency.
What types of memory are used in embedded systems and what are their functions?
RAM is used for temporary data storage, while ROM stores firmware essential for device operation. Limited memory capacity can hinder the development of complex software applications, but advancements are being made to address these constraints.
Why are input/output interfaces important in embedded systems?
Input/output interfaces allow embedded systems to interact with the external environment through sensors and actuators. Familiarity with communication protocols like I2C, SPI, and UART is crucial for effective integration in healthcare equipment.
What is the significance of Real-Time Operating Systems (RTOS) in healthcare applications?
RTOS manage hardware resources and ensure timely execution of tasks, which is critical in healthcare applications where timing is essential. They improve efficiency and adherence to regulatory standards.
How does Voler Systems optimize RTOS integration?
Voler Systems employs AI-supported engineering to optimize RTOS integration, ensuring that health-related equipment meets rigorous quality and reliability standards.
Can you provide examples of embedded technology applications in healthcare equipment?
Examples include heart rate monitors and insulin pumps, which demonstrate how embedded components collaborate to ensure functionality and reliability in healthcare devices.
What are the regulatory bodies involved in the compliance of medical devices?
Key regulatory entities include the FDA (U.S. Food and Drug Administration) and ISO (International Organization for Standardization), which oversee the approval and monitoring processes for medical instruments.
How are medical instruments classified and what does this imply for regulatory requirements?
Medical instruments are classified into three categories: Class I, II, and III. Higher-risk items (Class II and III) have more rigorous testing and comprehensive documentation requirements to demonstrate safety and effectiveness.
What standards should be reviewed for compliance in medical devices?
Important standards include ISO 13485, which outlines quality management systems, and IEC 60601, which addresses the safety and performance of medical electrical equipment.
What is the importance of conducting risk assessments for medical devices?
Conducting risk assessments, as specified in ISO 14971, helps identify potential hazards associated with devices and develop strategies to mitigate risks, ensuring patient safety and device reliability.
What documentation is necessary for compliance in medical device approval?
Comprehensive compliance submissions should include design history files, risk management files, and clinical evaluation reports to meet compliance expectations for successful approval and market access.
List of Sources
- Understand Embedded Technology Fundamentals
- Top medtech trends to watch in 2026 (https://medtechdive.com/news/medtech-trends-outlook-2026/810902)
- Medical applications for the PIC18-Q40 microcontroller (https://todaysmedicaldevelopments.com/news/microchip-mcus-smart-medical-device-microcontroller)
- Medical technology is unthinkable without embedded systems | embedded world (https://embedded-world.de/en/knowledge/2026/03/medical-technology-is-unthinkable-without-embedded-systems)
- MedTech in 2026: what’s changing device innovation (https://testresults.io/blog/medtech-in-2026-the-trends-redefining-medical-device-innovation)
- Microcontroller Applications in Medical Devices (https://vemeko.com/blog/microcontroller-applications-in-medical-devices.html)
- Identify Regulatory and Safety Requirements
- FDA Implements Quality Management System Regulation for Medical Devices (https://aabb.org/news-resources/news/article/2026/02/04/fda-implements-quality-management-system-regulation-for-medical-devices)
- How Will FDA's New Rules Impact Your Health Gadgets? (https://spectrum.ieee.org/fda-medical-device-rules)
- FDA’s 2026 Guidance on General Wellness Devices: Policy for Low-Risk Devices— Key Compliance and Regulatory Insights for Digital Health Companies - Kendall PC (https://kendallpc.com/fdas-2026-guidance-on-general-wellness-devices-policy-for-low-risk-devices-key-compliance-and-regulatory-insights-for-digital-health-companies)
- A QMSR State of Mind: FDA Adopts New Inspection Approach for Medical Devices as Quality Management System Regulation Takes Effect | Insights | Ropes & Gray LLP (https://ropesgray.com/en/insights/alerts/2026/02/a-qmsr-state-of-mind-fda-adopts-new-inspection-approach-for-medical-devices)
- Design and Implement Embedded Systems
- Engineering for the Future: The Trends Transforming Medtech in 2026 (https://medicaldesignbriefs.com/component/content/article/54404-engineering-for-the-future-0126)
- MedTech in 2026: what’s changing device innovation (https://testresults.io/blog/medtech-in-2026-the-trends-redefining-medical-device-innovation)
- Best Practices for Embedded System Services in Medical Device Development (https://volersystems.com/blog/best-practices-for-embedded-system-services-in-medical-device-development)
- MedTech Product Development Trends in 2026 (https://enlil.com/blog/medtech-product-development-trends-in-2026)
- Embedded Systems for Smart Medical Wearables in 2026 | Design and Connectivity Strategies (https://promwad.com/news/embedded-systems-smart-medical-wearables-2026-design-and-connectivity-strategies)
- Test and Validate Embedded Technology
- Embedded Software Testing for Medical Devices - Parasoft (https://parasoft.com/blog/medical-devices-embedded-software-testing)
- UAT in healthcare: A practical guide to user acceptance testing for medical apps - DeviQA (https://deviqa.com/blog/user-acceptance-testing-and-healthcare-what-is-the-purpose-of-uat-in-medical-apps)
- Usability Testing For Medical Devices: All You Should Know (https://info.idrmedical.com/blog/usability-testing-for-medical-devices)
- The Importance of Human Factors Validation and Usability Testing (https://nectarpd.com/the-importance-of-human-factors-validation-and-usability-testing)
- The Pivotal Importance of Usability Testing for Medical Devices (https://medicaltechoutlook.com/news/the-pivotal-importance-of-usability-testing-for-medical-devices-nwid-2708.html)
- Maintain and Update Embedded Systems
- Why your medical software should be updated? (https://blog.peekmed.com/medical-software-update)
- Importance of Software Updates and HIPAA regulations (https://usservicecenter.com/educational/importance-of-software-updates-and-hipaa-regulations)
- Why many existing medical devices fall short of the FDA's new cybersecurity standards (https://todaysmedicaldevelopments.com/news/why-many-existing-medical-devices-fall-short-fda-new-cybersecurity-standards)
- Theodore Cohen: The Importance of Software Security Updates for Medical Devices | AAMI Blog (https://array.aami.org/content/blog-post/theodore-cohen-importance-software-security-updates-medical-devices)
- Biomedical Engineering: Advanced Maintenance for Life-Critical Systems (https://oxmaint.com/blog/post/biomedical-engineering-advanced-maintenance-for-lifecritical-systems)






