Introduction
Embedded software is fundamental to the functionality of modern medical devices, significantly impacting patient care through its essential roles in monitoring and data processing. As healthcare technology evolves, understanding best practices in embedded system software development becomes essential for manufacturers aiming to enhance device functionality while ensuring compliance with stringent regulatory standards.
Developers face significant hurdles in ensuring compliance and security, which can impede innovation. Addressing these challenges is crucial for developers to balance innovation with the rigorous demands of the healthcare landscape.
Define Embedded Software and Its Importance
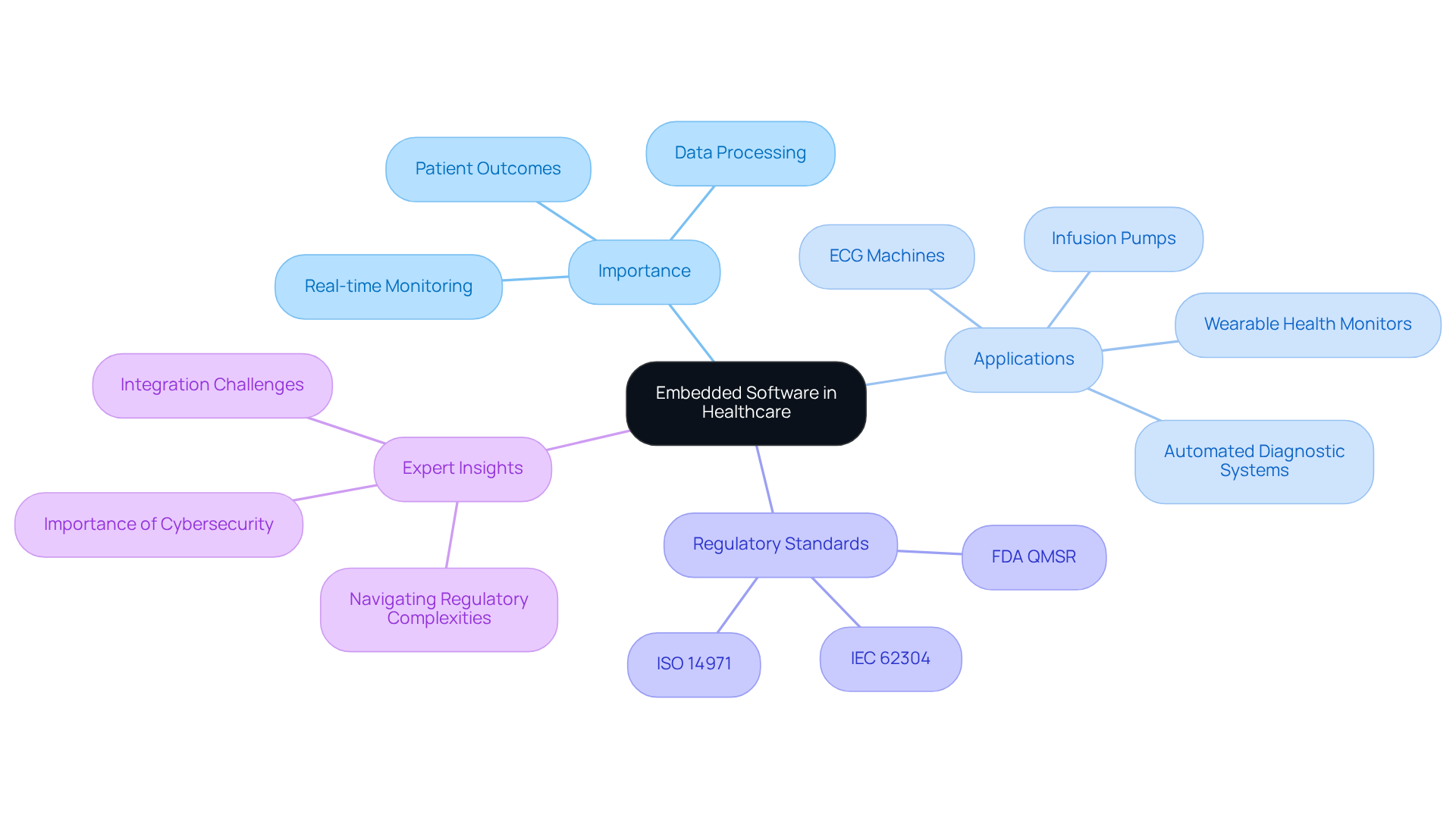
Embedded programming serves as a critical component within healthcare hardware, enabling essential functions that impact patient care. In medical instruments such as infusion pumps, ECG machines, heart pumps, liquid biopsy platforms, and wearable health monitors, this programming is vital for managing operations, processing data, and ensuring compliance with stringent safety standards. The reliability of embedded programming is vital, directly impacting patient outcomes through real-time monitoring and precise data processing.
For example, automated diagnostic systems significantly reduce patient data processing time compared to manual methods, thereby enhancing the speed and accuracy of clinical decisions. Compliance with regulatory standards, including IEC 62304 and ISO 14971, highlights the critical role of embedded system software development in ensuring product safety and efficacy.
As healthcare technology advances, the integration of robust embedded system software development is increasingly recognized as a cornerstone for enhancing functionality and ensuring high-quality patient care. However, navigating the regulatory landscape can be complex and varies significantly across regions and device types. This understanding is essential for manufacturers to ensure their products meet safety and efficacy requirements.
Industry experts emphasize that understanding FDA's QMSR and Cybersecurity Mandates for Medical Devices is crucial for manufacturers to effectively navigate these complexities.
Explore Types of Embedded Software Development
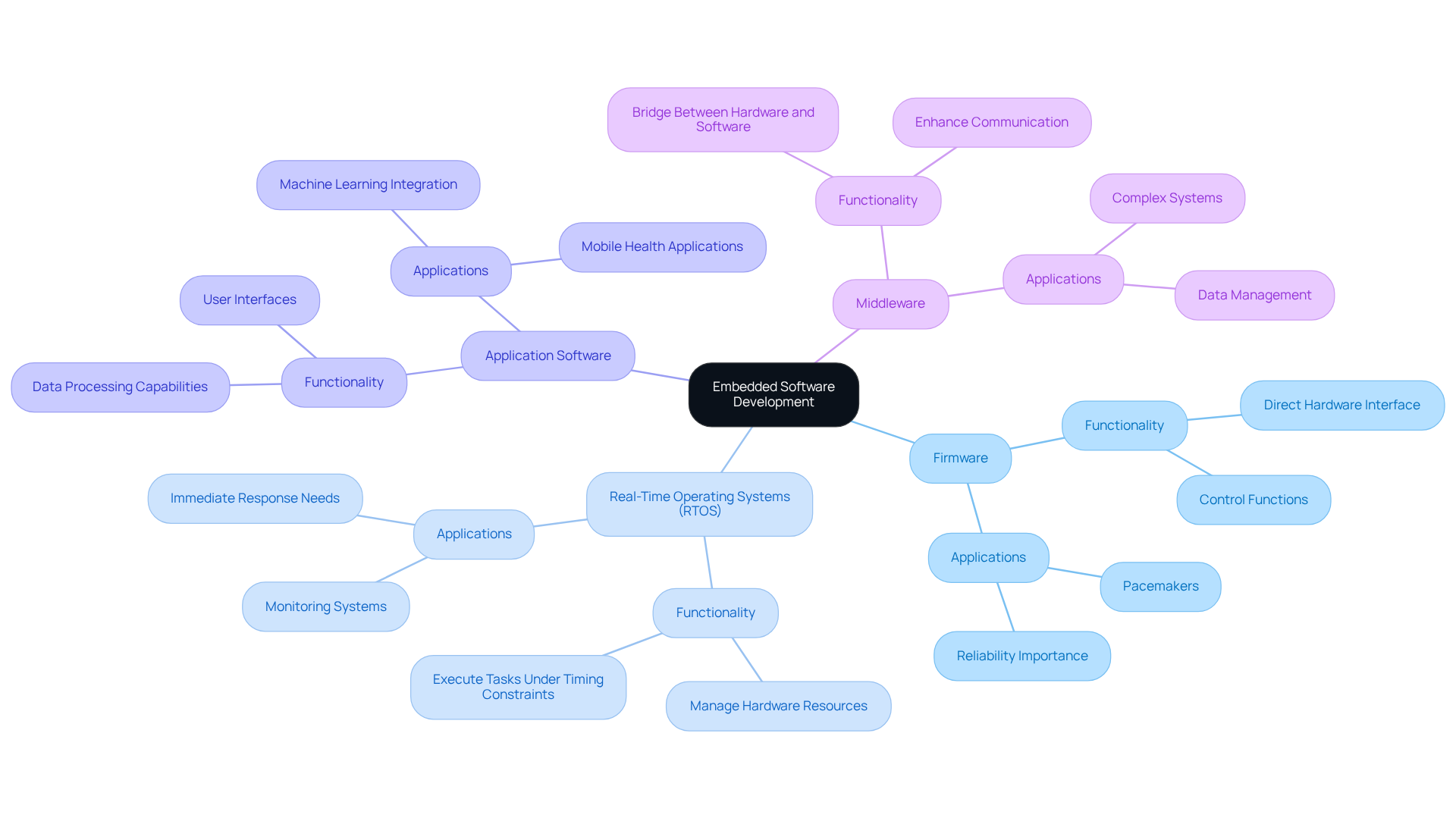
Embedded software development plays a critical role in the functionality of medical devices, with various types tailored to meet specific needs:
- Firmware: This low-level program directly interfaces with hardware components, delivering essential control functions. It is crucial for instruments that require precise timing and control, such as pacemakers, where reliability is paramount.
- Real-Time Operating Systems (RTOS): RTOS are engineered to manage hardware resources and execute tasks under strict timing constraints. They are indispensable in applications requiring immediate responses, such as monitoring systems that must react swiftly to changes in patient conditions.
- Application Software: This software layer provides higher-level functionality, often incorporating user interfaces and data processing capabilities. It is essential for tools that require user interaction, such as mobile health applications that monitor patient data and enable communication. By integrating machine learning inference engines, these platforms can recognize patterns and interpret physiological data more effectively, increasing their value in modern healthcare. This innovation reflects Voler Systems' commitment to leveraging artificial intelligence for improved healthcare equipment functionality.
- Middleware: Serving as a bridge between hardware and application software, middleware enhances communication and data management. It is particularly beneficial in complex systems where multiple components must interact seamlessly, ensuring efficient operation.
Grasping these categories allows developers to choose the most appropriate method for embedded system software development based on the particular needs of the healthcare tool being created, ultimately improving functionality and adherence to regulatory standards. Navigating the complex regulatory landscape can be daunting for startups in the healthcare sector. Voler Systems also offers documentation compliance assistance, which is crucial for startups maneuvering through these challenges.
Identify Challenges in Embedded Software Development for Medical Devices
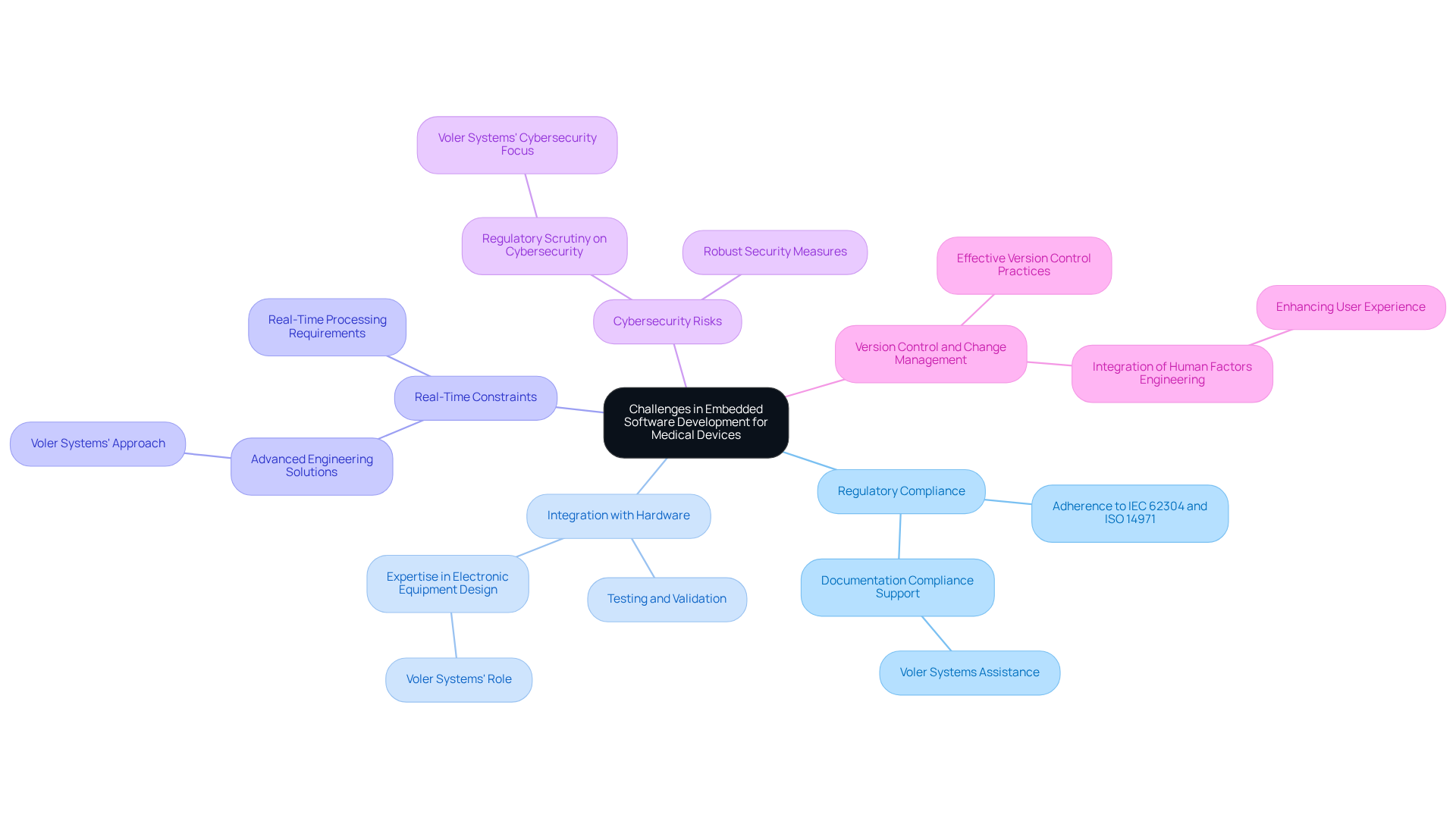
Embedded software development for medical devices presents a series of complex challenges that require careful navigation:
- Regulatory Compliance: Adhering to stringent regulations such as IEC 62304 and ISO 14971 is essential. Developers must ensure their applications meet safety and efficacy standards throughout their lifecycle. This process can be both time-consuming and resource-intensive. Voler Systems offers detailed documentation compliance support, assisting startups in navigating these intricate regulatory requirements effectively.
- Integration with Hardware: The close coupling of programs and hardware often leads to integration issues. Testing and validating that the application functions smoothly with hardware components is essential for ensuring reliability and performance. Voler Systems' expertise in electronic equipment design ensures seamless integration, enhancing overall reliability.
- Real-Time Constraints: Numerous healthcare instruments require real-time processing abilities, making design and testing more challenging. Developers must ensure that the software can respond to inputs within strict time limits to maintain functionality and patient safety. Not meeting these constraints can jeopardize both patient safety and the functionality of the device. Voler Systems leverages advanced engineering solutions to meet these real-time demands effectively.
- Cybersecurity Risks: With the growing interconnectivity of medical equipment, the danger of cyber threats intensifies. Implementing robust security measures is essential to safeguard patient information and uphold equipment integrity, particularly as regulatory scrutiny on cybersecurity increases. At Voler Systems, we prioritize cybersecurity in our development processes, ensuring that devices are resilient against emerging threats.
- Version Control and Change Management: Effective version control is critical for managing software updates and changes, particularly in regulated environments. Maintaining thorough documentation and traceability is essential to comply with regulatory requirements and ensure product quality. Additionally, integrating human factors engineering (HFE) practices into the development process is crucial for safety validation and regulatory strength. Voler Systems incorporates HFE principles to enhance user experience and adherence.
Addressing these challenges necessitates a comprehensive understanding of both the technical and regulatory landscapes related to embedded system software development, alongside a steadfast commitment to quality and safety. With the right expertise, manufacturers can not only meet these challenges but also enhance the safety and efficacy of their medical devices.
Implement Best Practices for Quality and Compliance
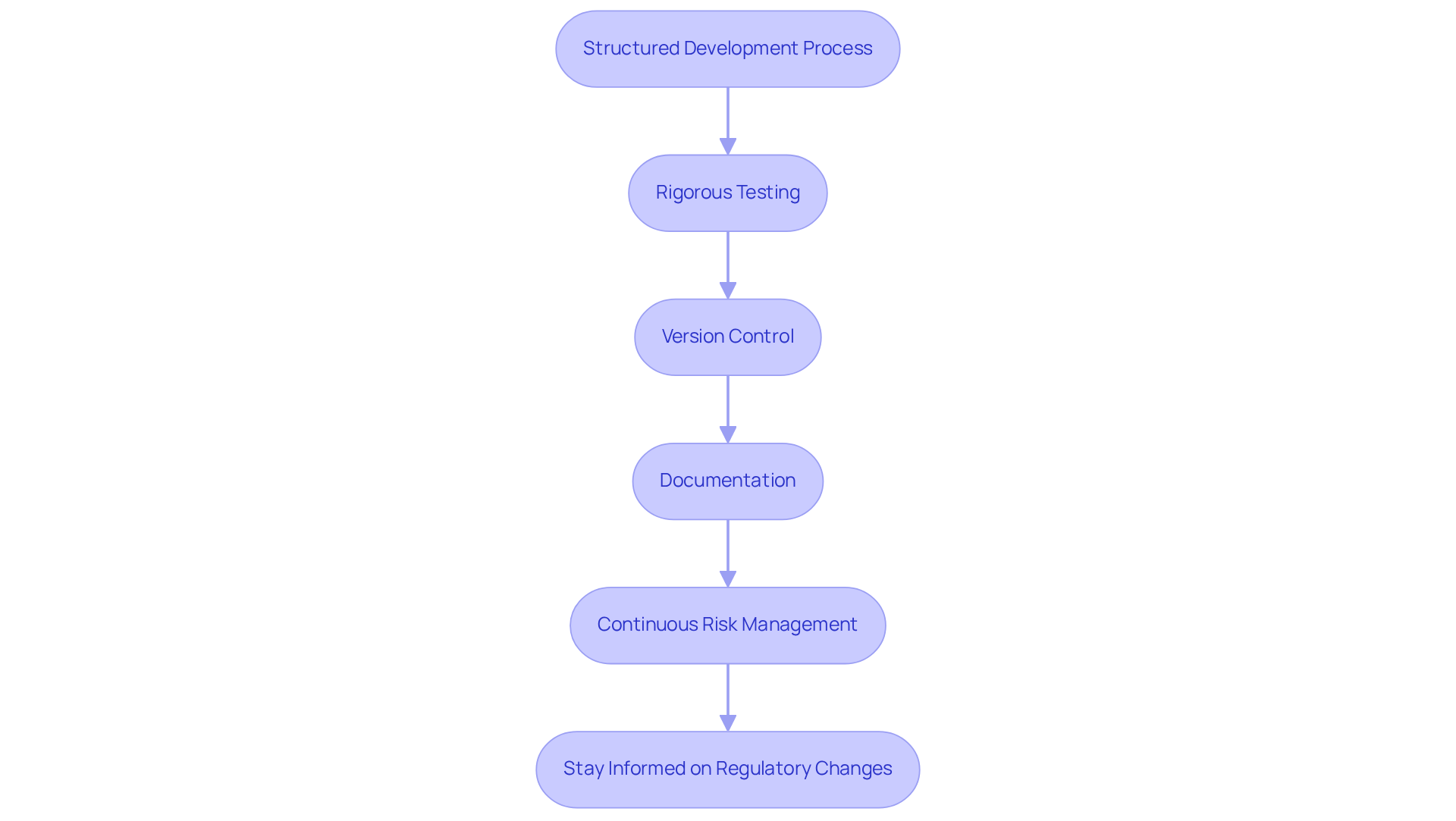
To ensure quality and compliance in embedded software development for medical devices, developers must adopt a structured approach:
- Adopt a Structured Development Process: Implement a well-defined development lifecycle (SDLC) that encompasses requirements analysis, design, implementation, testing, and maintenance. This organized method ensures comprehensive coverage of all aspects related to embedded system software development. It facilitates adherence to standards such as IEC 62304, which emphasizes safety and risk management throughout the system lifecycle.
- Conduct Rigorous Testing: Employ comprehensive testing strategies, including unit testing, integration testing, and system testing. Automated testing tools enhance efficiency and precision. They ensure that the program meets all functional and safety requirements. Regular security checks and vulnerability scans are essential to prevent data breaches and uphold system integrity.
- Implement Version Control: Utilize version control systems to manage changes in applications effectively. This practice guarantees that all changes are recorded and traceable, which is essential for regulatory adherence and preserving a clear history of system development.
- Focus on Documentation: Maintain thorough documentation throughout the development process, including design specifications, testing protocols, and regulatory records. Proper documentation is vital for demonstrating adherence to regulatory standards and ensuring audit readiness, particularly in light of the FDA's updated cybersecurity guidance.
- Engage in Continuous Risk Management: Regularly evaluate and handle risks linked to the application. This involves identifying potential hazards, evaluating their impact, and implementing mitigation strategies to minimize risks. Ongoing observation of application performance after launch is essential for guaranteeing reliability and adherence to changing regulations.
- Stay Informed on Regulatory Changes: Keep abreast of updates to relevant regulations and standards. This proactive strategy ensures that the program remains compliant with the latest requirements, particularly as cybersecurity considerations increasingly influence procurement decisions in healthcare.
By neglecting these best practices, developers risk compromising the safety and effectiveness of medical devices.
Conclusion
Embedded software is crucial in medical devices, underpinning both functionality and safety in healthcare technology. This article highlights the critical role of embedded software development in enhancing patient care and ensuring compliance with rigorous regulatory standards. As healthcare continues to evolve, the integration of robust embedded systems becomes increasingly essential to ensure both efficacy and safety of medical devices.
Key insights have been discussed, including the various types of embedded software - such as firmware, real-time operating systems, application software, and middleware - each tailored to meet specific healthcare needs. Developers often grapple with stringent regulatory requirements and the need for seamless hardware integration. The challenges faced in this field, including real-time processing demands, cybersecurity risks, and effective version control, highlight the complexities that developers must navigate. Adopting best practices, including structured development processes, rigorous testing, and continuous risk management, is vital for maintaining high-quality standards and ensuring patient safety.
The advancement of embedded software in medical devices presents a significant opportunity to enhance healthcare outcomes. Developers and manufacturers are encouraged to remain vigilant in their adherence to best practices and regulatory guidelines, ensuring that they not only meet current standards but also proactively address emerging challenges. By prioritizing quality and compliance, the healthcare industry can drive innovation, ultimately enhancing patient care and safety.
Frequently Asked Questions
What is embedded software?
Embedded software is a type of programming that is integrated into healthcare hardware to manage operations, process data, and ensure compliance with safety standards.
Why is embedded software important in healthcare?
Embedded software is vital in healthcare as it impacts patient care by enabling essential functions in medical instruments, ensuring reliable operation, real-time monitoring, and precise data processing.
Can you provide examples of medical devices that use embedded software?
Examples of medical devices that utilize embedded software include infusion pumps, ECG machines, heart pumps, liquid biopsy platforms, and wearable health monitors.
How does embedded software improve patient care?
Embedded software improves patient care by automating processes, which significantly reduces data processing time and enhances the speed and accuracy of clinical decisions.
What regulatory standards are relevant to embedded software in healthcare?
Relevant regulatory standards include IEC 62304 and ISO 14971, which ensure that embedded system software development meets safety and efficacy requirements.
What challenges do manufacturers face regarding embedded software in healthcare?
Manufacturers face challenges in navigating a complex regulatory landscape that varies across regions and device types, which is essential for ensuring product safety and efficacy.
What should manufacturers understand about FDA regulations related to embedded software?
Manufacturers should understand the FDA's Quality Management System Regulations (QMSR) and Cybersecurity Mandates for Medical Devices to effectively navigate regulatory complexities.
List of Sources
- Define Embedded Software and Its Importance
- Software recent news | MDDI (https://mddionline.com/digital-health/software)
- Why Embedded Systems Are Crucial in the Medical Device Industry (https://volersystems.com/blog/why-embedded-systems-are-crucial-in-the-medical-device-industry)
- How Embedded Software is transforming Healthcare devices (https://exaud.com/blog/embedded-software-solutions-in-healthcare-devices)
- 4 Takeaways on the Impact of Embedded Systems in Medical Devices (https://blog.boston-engineering.com/embedded-systems-med-devices)
- Explore Types of Embedded Software Development
- MedTech in 2026: what’s changing device innovation (https://testresults.io/blog/medtech-in-2026-the-trends-redefining-medical-device-innovation)
- RunSafe Security Releases 2026 Medical Device Cybersecurity Index as Attacks Impacting Medical Devices Rise and Procurement Standards Tighten (https://businesswire.com/news/home/20260429787733/en/RunSafe-Security-Releases-2026-Medical-Device-Cybersecurity-Index-as-Attacks-Impacting-Medical-Devices-Rise-and-Procurement-Standards-Tighten?feedref=JjAwJuNHiystnCoBq_hl-cFcUWvK4NSyDWO6DSHmLQqqcp-o_pnudlUwsb5apQ1S4gUE65BTfjH3-pSuqdv0gW3cb3F4oTIgUqCPafFkgu6AeRucmCP4ZHknI_zC0f97)
- Embedded Systems for Smart Medical Wearables in 2026 | Design and Connectivity Strategies (https://promwad.com/news/embedded-systems-smart-medical-wearables-2026-design-and-connectivity-strategies)
- The Future of Remote Firmware Updates in Medical Devices - Simbex (https://simbex.com/the-future-of-remote-firmware-updates-in-medical-devices)
- Medical Device Software Development (2026 Guide) - CLEIO (https://cleio.com/insights/blog/medical-device-software-development)
- Identify Challenges in Embedded Software Development for Medical Devices
- FDA Tightens Its Medical Device Cybersecurity Guidance (https://fedtechmagazine.com/article/2026/03/fda-tightens-its-medical-device-cybersecurity-guidance-perfcon)
- What Medtech Companies Can Expect in 2026: Regulation, Risk, and Resilience (https://morganlewis.com/pubs/2026/03/what-medtech-companies-can-expect-in-2026-regulation-risk-and-resilience)
- MedTech Product Development Trends in 2026 (https://enlil.com/blog/medtech-product-development-trends-in-2026)
- The 2026 challenges facing the MedTech industry | Medical Economics (https://medicaleconomics.com/view/the-2026-challenges-facing-the-medtech-industry)
- Medical Device Software Development (2026 Guide) - CLEIO (https://cleio.com/insights/blog/medical-device-software-development)
- Implement Best Practices for Quality and Compliance
- FDA Tightens Its Medical Device Cybersecurity Guidance (https://fedtechmagazine.com/article/2026/03/fda-tightens-its-medical-device-cybersecurity-guidance-perfcon)
- Medical Device Software Development (2026 Guide) - CLEIO (https://cleio.com/insights/blog/medical-device-software-development)
- Understanding Software Life Cycle in Medical Devices (https://dashtechinc.com/blog/software-life-cycle-management-for-medical-devices)
- FDA’s new guidance: Computer Software Assurance (CSA) for production and quality system software (https://aurevia.com/news/fda-new-guidance-transforms-software-validation-for-production-and-quality-systems)





