Introduction
The development of electronic products for medical devices presents significant challenges that demand precision and adherence to regulatory standards. This article outlines a systematic approach that not only guides innovators through the intricate phases of product development but also emphasizes the importance of prototyping and rigorous testing to ensure safety and efficacy. To address these challenges, a structured approach is essential for ensuring compliance while fostering innovation.
Understand the Electronic Product Development Process
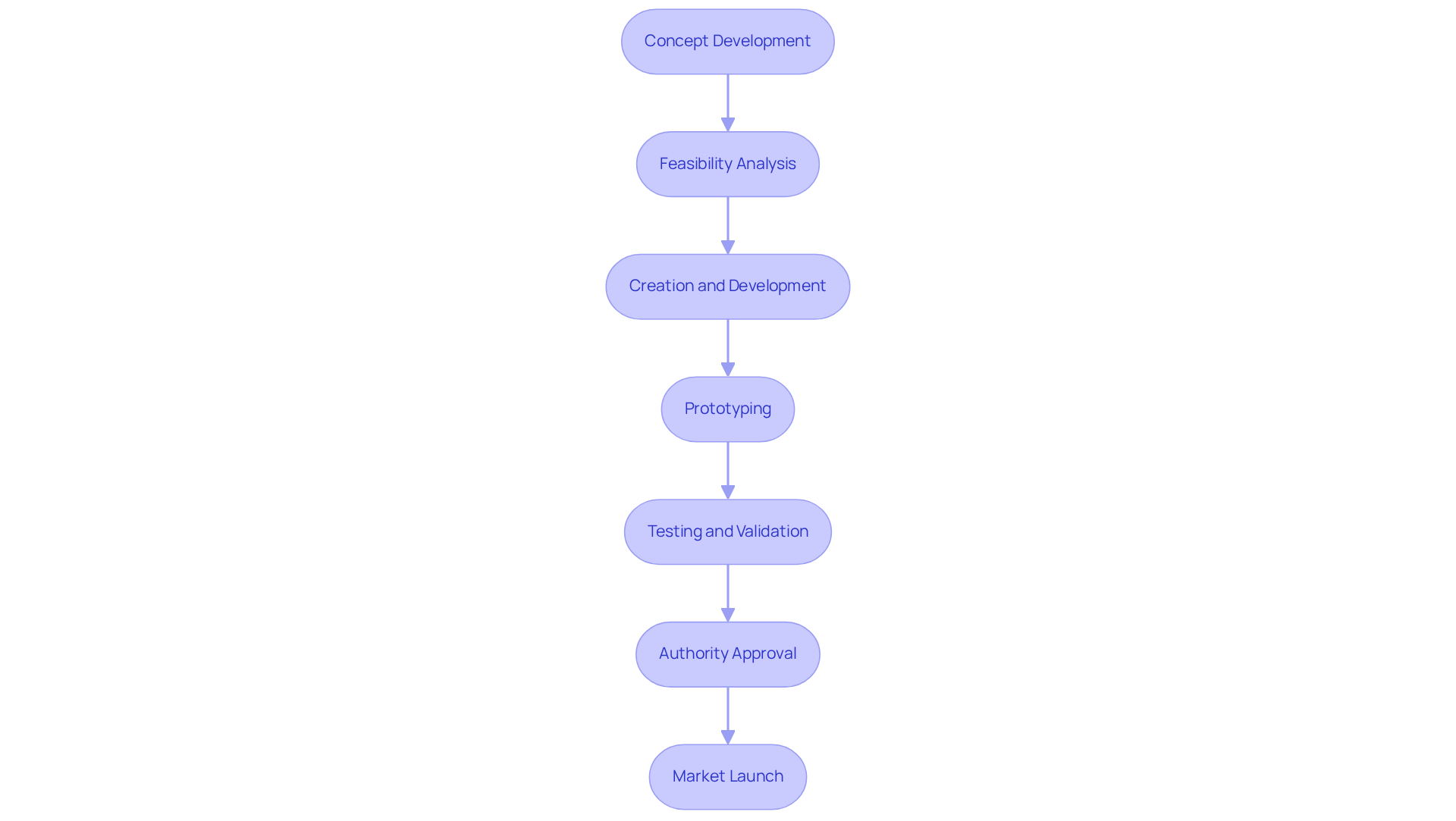
The process to develop electronic products for medical devices is a multifaceted journey that demands meticulous attention to detail at every stage.
- Concept Development: This initial phase involves identifying the clinical need your product will address. Carrying out comprehensive market research is crucial to comprehend current solutions and pinpoint gaps that your product can address.
- Feasibility Analysis: Here, you assess both the technical and economic viability of your concept. This encompasses initial planning tasks and cost assessment, which are essential for deciding if the project can move forward.
- Creation and Development: In this phase, detailed specifications and plans for your device are created, covering both hardware and software components. This step is vital for ensuring that we develop electronic products that align with user needs and regulatory requirements.
- Prototyping: Prototyping involves creating models that facilitate the evaluation and refinement of the initial concept, allowing for early identification and correction of design flaws. Rapid prototyping techniques, such as 3D printing, enable quick iterations, which are essential for addressing potential issues early in the process.
- Testing and Validation: Rigorous testing is conducted to ensure that the device meets safety and efficacy standards. Ensuring compliance with frameworks such as ISO 13485 and FDA regulations is paramount during this phase, as failure to do so can lead to significant setbacks.
- Authority Approval: Preparing comprehensive documentation for oversight review is essential to demonstrate compliance with industry standards. This step often involves navigating complex approval pathways, such as the FDA's 510(k) or De Novo processes.
- Market Launch: Finally, planning and executing the product launch involves developing marketing strategies and establishing distribution channels to ensure successful market entry.
Navigating these phases effectively can significantly enhance a company's ability to develop electronic products while delivering compliant and innovative medical solutions.
Utilize Prototyping to Validate Concepts and Reduce Risks
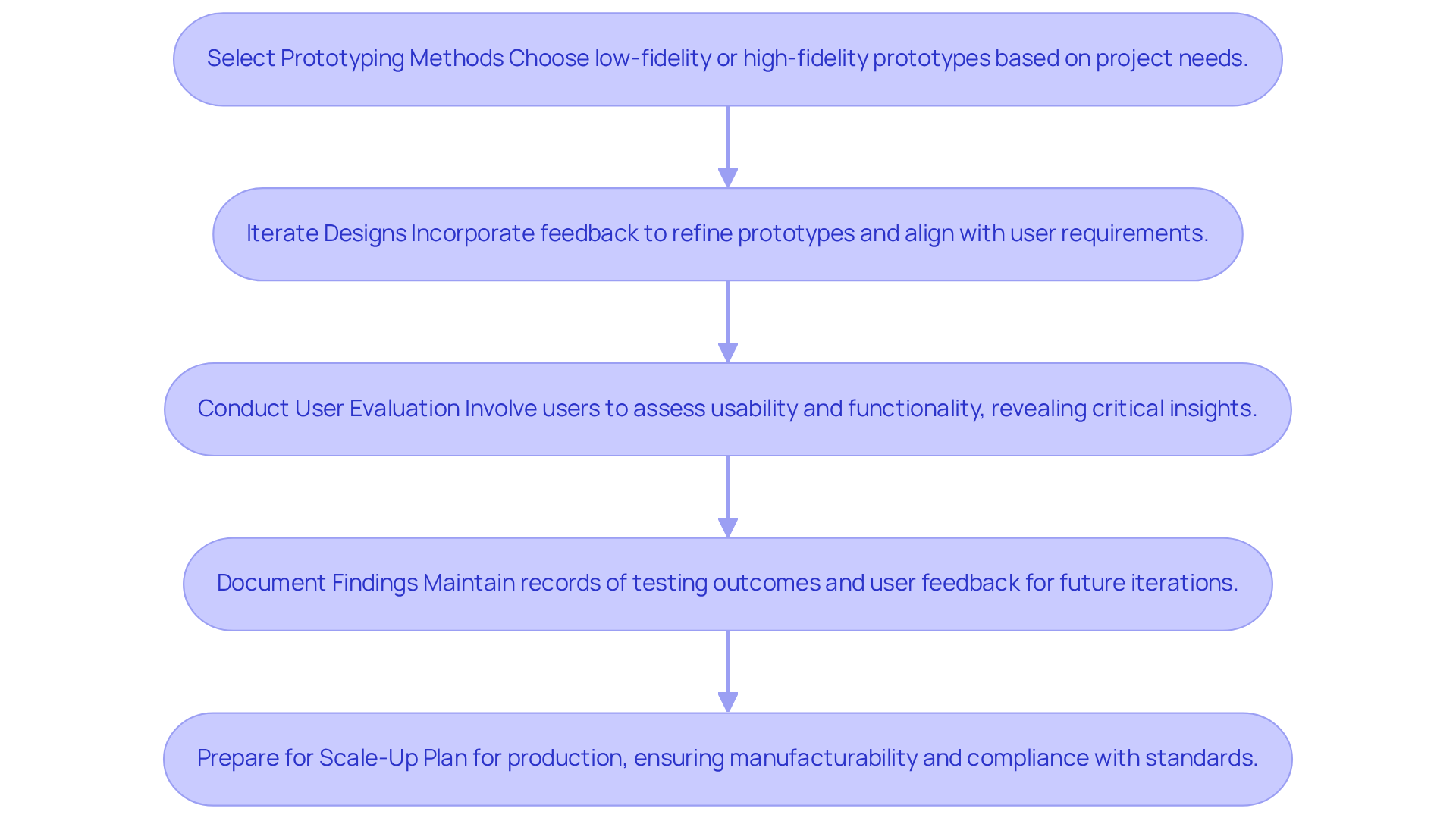
Prototyping serves as a pivotal phase in medical device development, enabling teams to validate concepts and effectively mitigate associated risks. Here’s how to optimize your prototyping process with the expertise of Voler Systems:
- Select Prototyping Methods: Choose between low-fidelity (such as paper models) and high-fidelity prototypes (functional models) based on your project requirements. Low-fidelity prototypes are ideal for initial concept validation, while high-fidelity prototypes are essential for assessing functionality and user interaction.
- Iterate Designs: Incorporate feedback from stakeholders and potential users to refine your prototypes. This iterative method enables early detection of problems, ensuring that the layout aligns with user requirements and anticipations. Voler Systems highlights the significance of modifying concepts for production, ensuring that prototypes are not only innovative but also feasible for manufacturing.
- Conduct User Evaluation: Involve actual users in assessing prototypes to gather insights on usability and functionality. Effective user evaluation can reveal critical information about user interactions and preferences, which is invaluable for making informed design decisions. For instance, functional prototypes can be utilized for usability studies and limited clinical evaluations, depending on your regulatory strategy. Evaluation methods such as drop assessments, leakage current evaluations, and temperature measurements are crucial for ensuring device safety and compliance.
- Document Findings: Maintain detailed records of testing outcomes and user feedback. This documentation is crucial for informing future development iterations and supporting compliance submissions, as it demonstrates adherence to industry standards. Understanding the materials used for prototyping, such as medical-grade plastics, silicone, metals, and biocompatible polymers, is also vital for effective prototyping. Voler Systems offers documentation compliance assistance, helping startups navigate compliance challenges effectively.
- Prepare for Scale-Up: Once the prototype is validated, initiate planning for scaling up production. Ensure that the design is manufacturable and meets all compliance standards, which is vital for a successful market launch. With the anticipated Medical Device User Fee Amendments (MDUFA VI) expected to go to Congress for authorization in 2026, staying informed about regulatory changes is crucial for compliance.
Without a structured prototyping process, teams may face unforeseen challenges that jeopardize product viability. This structured approach not only mitigates risks but also enhances the potential for successful product launches in the competitive medical device market. Ultimately, a well-executed prototyping strategy is essential for navigating the complexities involved in developing electronic products and ensuring compliance in a competitive landscape.
Design and Develop Hardware: Schematic and PCB Layout
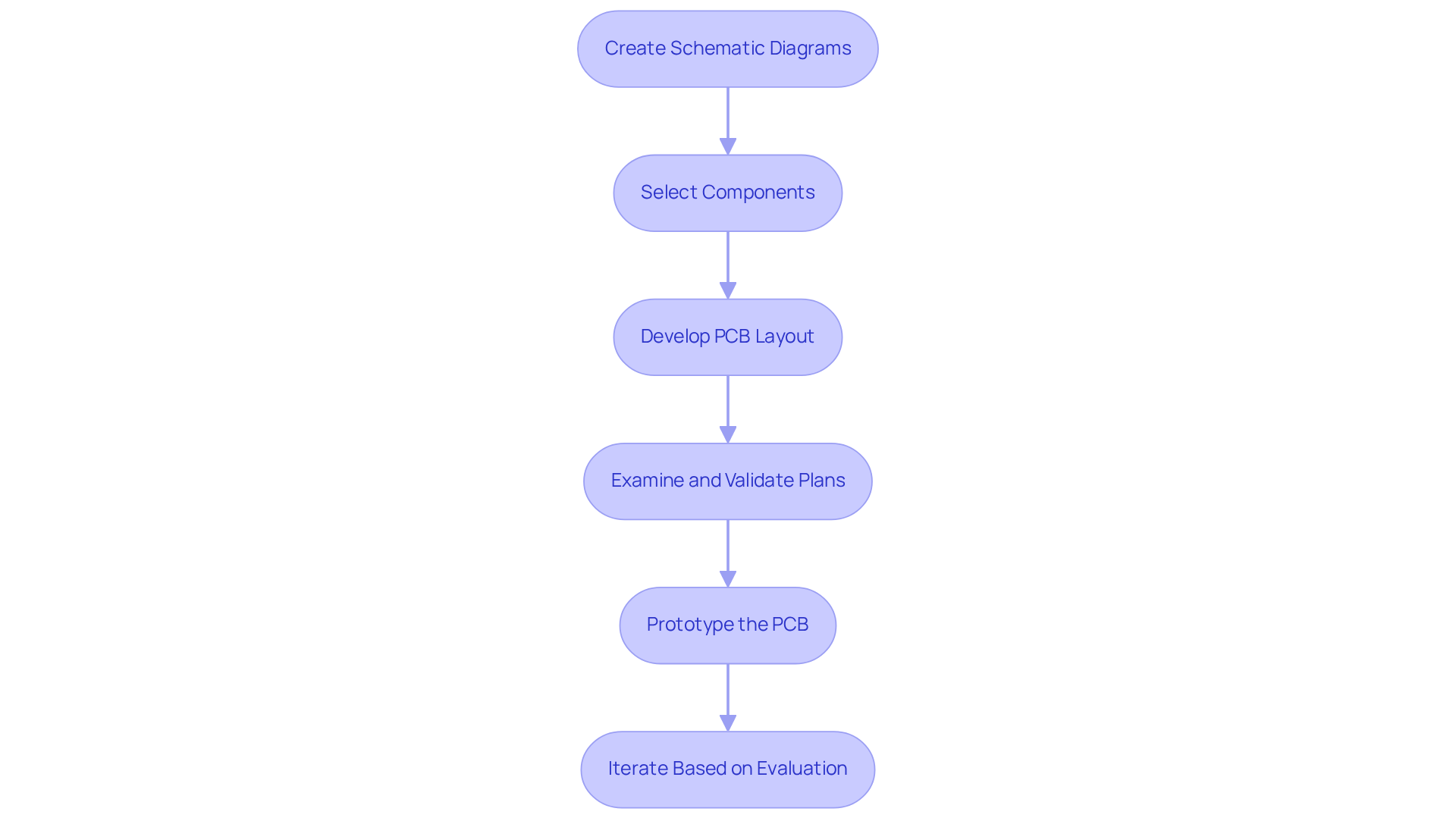
Designing and developing hardware for medical devices requires a systematic approach to ensure compliance and functionality:
- Create Schematic Diagrams: Start by drafting schematic diagrams that outline the electrical connections and components of your apparatus. Utilize advanced software tools such as Altium Designer or Eagle for precise representations.
- Select Components: Choose components that match your equipment's specifications, considering factors like power consumption, size, and regulatory standards. The trend towards smaller, lighter components is expected to enhance usability and compliance for patients with limited dexterity or cognitive challenges by 2026. Voler Systems enhances battery longevity in wireless medical equipment by implementing ultra-low power strategies, ensuring your products satisfy these changing requirements.
- Develop PCB Layout: After completing the schematic, create the PCB layout with careful attention to component placement, trace routing, and grounding techniques to minimize electromagnetic interference (EMI), essential for reliability. Innovative embedded systems development with FPGA can offer cost-efficient and flexible solutions for IoT applications, enhancing your device's functionality.
- Examine and Validate Plans: Conduct thorough evaluations with your engineering team to confirm that both the schematic and PCB layout meet all requirements. This diligence ensures compliance and accelerates the approval process, validating against official standards, including FDA controls and ISO 13485. Voler Systems offers documentation compliance support for medical device startups, effectively navigating these regulatory challenges.
- Prototype the PCB: Fabricate a prototype of the PCB to evaluate its functionality. This step is essential for identifying flaws early in the design process, preventing costly errors during manufacturing and enabling real-world evaluation of usability and performance.
- Iterate Based on Evaluation: Utilize feedback from prototype evaluation to refine the schematic and PCB layout, ensuring optimal performance and compliance with industry standards. Maintaining proper documentation and risk management files from the start is essential for streamlining verification and validation processes. Iterative prototyping is crucial for reducing costly redesigns and expediting the development timeline.
By following these steps and leveraging expertise in electronic design and development, you can create robust hardware designs that are ready for the next phases to develop electronic products for medical equipment.
Conduct Testing and Ensure Compliance for Market Readiness
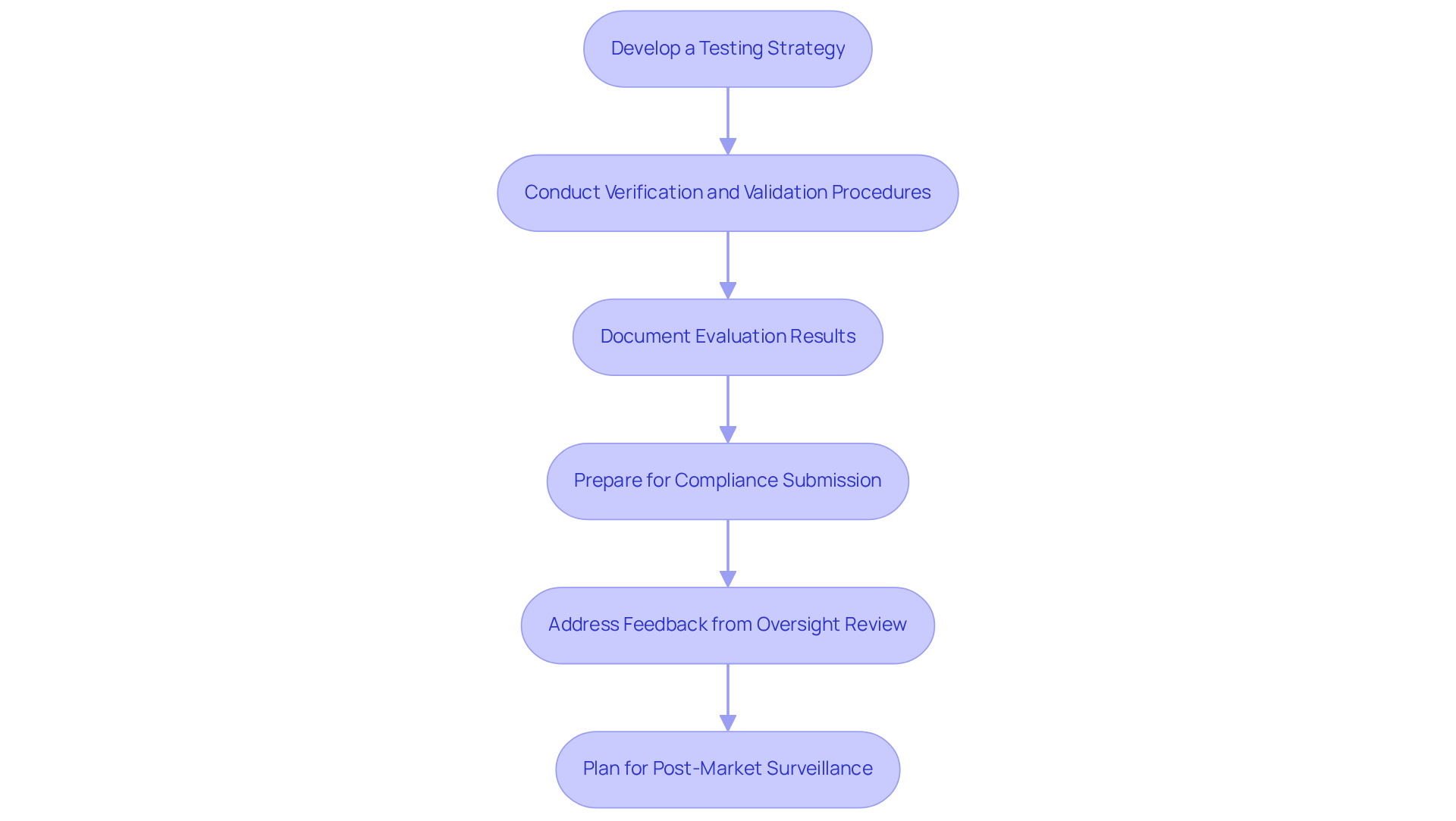
To ensure your medical device meets market standards, it is crucial to follow a structured approach to testing and compliance:
- Develop a Testing Strategy: A comprehensive assessment protocol must align with regulatory standards to ensure compliance and help prevent typical manufacturing assessment errors, including limited evaluation scope and overlooked safety reviews. This strategy should encompass functional evaluations, usability assessments, and safety evaluations.
- Conduct Verification and Validation Procedures: Begin with verification evaluations to confirm that the apparatus complies with design specifications. Follow this with validation assessments to ensure the device meets user needs and intended applications. Common oversights at this stage include the lack of end-user engagement in validation and inadequate documentation of the evaluation process.
- Document Evaluation Results: Maintain meticulous records of all assessment procedures and outcomes. This documentation is essential for compliance submissions and quality assurance processes. Ensure that all results are clearly recorded to avoid errors related to miscommunication or incomplete data.
- Prepare for Compliance Submission: Gather all essential documentation, including examination results, design specifications, and risk evaluations, for submission to oversight organizations such as the FDA. Submitting incomplete documentation can lead to significant delays in the approval process, so it is vital to address all compliance requirements thoroughly.
- Address Feedback from Oversight Review: Be prepared to respond to any comments or inquiries for additional information from oversight agencies, which may require further testing or modifications to the apparatus. Failure to engage proactively may result in prolonged review times and potential rejections.
- Plan for Post-Market Surveillance: After the product is launched, implement a post-market surveillance strategy to monitor its performance and safety in real-world conditions. This ongoing assessment is vital for maintaining compliance and ensuring long-term product safety, helping to identify any issues that may arise post-launch.
By adhering to these rigorous testing and compliance steps, you not only enhance your device's market readiness but also safeguard patient safety and product integrity.
Conclusion
The development of electronic products for medical devices is a multifaceted process that demands meticulous planning and execution at every stage. A structured approach that includes:
- Concept development
- Feasibility analysis
- Prototyping
- Testing
- Compliance
enables companies to create innovative solutions. These solutions address real clinical needs while meeting stringent regulatory standards.
Key steps such as conducting thorough market research, validating concepts through prototyping, and ensuring compliance through rigorous testing are essential to navigate the complexities of medical device development. Each phase is critical in shaping a product. From schematic design and PCB layout to market launch, these stages ensure the product is functional, safe, and effective for users.
Navigating the path from idea to market-ready product is complex, but it offers significant opportunities for innovation in healthcare. Embracing best practices in electronic product development not only enhances the potential for successful launches but also contributes to better patient outcomes. Stakeholders must prioritize staying informed and proactive to effectively navigate the evolving landscape of medical device regulations and market demands.
Frequently Asked Questions
What is the first phase of the electronic product development process for medical devices?
The first phase is Concept Development, which involves identifying the clinical need the product will address and conducting comprehensive market research to understand current solutions and identify gaps.
What is assessed during the Feasibility Analysis phase?
The Feasibility Analysis phase assesses both the technical and economic viability of the concept, including initial planning tasks and cost assessment to determine if the project can proceed.
What is created during the Creation and Development phase?
In the Creation and Development phase, detailed specifications and plans for the device are created, covering both hardware and software components to ensure alignment with user needs and regulatory requirements.
What is the purpose of prototyping in the electronic product development process?
Prototyping involves creating models to evaluate and refine the initial concept, allowing for early identification and correction of design flaws. Techniques like 3D printing enable quick iterations to address potential issues early.
What is the focus of the Testing and Validation phase?
The Testing and Validation phase focuses on rigorous testing to ensure the device meets safety and efficacy standards, ensuring compliance with frameworks such as ISO 13485 and FDA regulations.
What is required during the Authority Approval phase?
During the Authority Approval phase, comprehensive documentation must be prepared for oversight review to demonstrate compliance with industry standards, often involving complex approval pathways like the FDA's 510(k) or De Novo processes.
What does the Market Launch phase involve?
The Market Launch phase involves planning and executing the product launch, which includes developing marketing strategies and establishing distribution channels for successful market entry.
List of Sources
- Understand the Electronic Product Development Process
- Top medtech trends to watch in 2026 (https://medtechdive.com/news/medtech-trends-outlook-2026/810902)
- Top MedTech Trends to Watch in 2026 (https://ptc.com/en/blogs/medtech/top-medtech-industry-trends?srsltid=AfmBOort8w94EJzMLMIB7cmQv5kkYAqEoyqwtuSgQeuzksvrUOQuiFUu)
- Medical Product Development Trends - 2026 | IDP (https://innovativedesignproducts.com/medical-product-development-trends-2026)
- Top 15 Medical Device Trends in 2026 To Watch (https://pharmadocx.com/top-15-medical-device-trends-in-2026)
- MedTech Product Development Trends in 2026 (https://enlil.com/blog/medtech-product-development-trends-in-2026)
- Utilize Prototyping to Validate Concepts and Reduce Risks
- Medical Device Prototyping In Accelerating Time To Market (https://europlaz.co.uk/medical-device-prototyping-in-accelerating-time-to-market)
- Medical Device Testing Prevents Costly Redesigns (https://designnews.com/prototyping/early-medical-device-testing-prevents-costly-redesigns-ul-solutions-engineer-advises-at-mdm-west)
- Top medtech trends to watch in 2026 (https://medtechdive.com/news/medtech-trends-outlook-2026/810902)
- Accelerating Medical Device Innovation: A Guide to Rapid Prototyping (https://nicerapid.com/blog/accelerating-medical-device-innovation-a-guide-to-rapid-prototyping)
- Design and Develop Hardware: Schematic and PCB Layout
- Innovative Trends Shaping Medical Device Design in 2026 (https://ritareviews.net/innovative-trends-shaping-medical-device-design-in-2026)
- 2026 PCB Outlook: Design, Mfg & Supply Chain Trends (https://pcbinq.com/2026-pcb-outlook-design-mfg-supply-chain-trends)
- ISO 13485 PCB Assembly 2026: Medical Device Manufacturing (https://pcbaassembly.com/blogs/news/iso-13485-pcb-assembly-in-2026-meeting-the-highest-standards-for-medical-device-manufacturing)
- Medical Device Design Trends 2026 | Startups & OEMs | IDP (https://innovativedesignproducts.com/medical-device-design-trends-2026)
- Top medtech trends to watch in 2026 (https://medtechdive.com/news/medtech-trends-outlook-2026/810902)
- Conduct Testing and Ensure Compliance for Market Readiness
- Top Medical Device Regulations to Watch in 2026 (https://xtalks.com/top-medical-device-regulations-to-watch-in-2026-4549)
- For 2026, FDA signals shifts in digital health framework | Nixon Peabody LLP (https://nixonpeabody.com/insights/alerts/2026/01/27/for-2026-fda-signals-shifts-in-digital-health-framework)
- Navigating New FDA Expectations Under QMSR (https://healthcarepackaging.com/industry-news/regulatory/article/22963491/navigating-new-fda-expectations-under-qmsr)
- FDA Implements Quality Management System Regulation for Medical Devices (https://aabb.org/news-resources/news/article/2026/02/04/fda-implements-quality-management-system-regulation-for-medical-devices)
- FDA Tightens Its Medical Device Cybersecurity Guidance (https://fedtechmagazine.com/article/2026/03/fda-tightens-its-medical-device-cybersecurity-guidance-perfcon)





