Introduction
In the rapidly evolving landscape of healthcare technology, the development of medical devices offers significant opportunities alongside complex challenges. A well-crafted product strategy is essential, as it not only addresses critical healthcare needs but also ensures adherence to stringent regulatory standards. The industry is currently confronted with pressing issues, such as the alarming number of individuals without access to safe surgical care. This raises an important question: how can medical device manufacturers effectively integrate user-centered design, compliance, and technological feasibility into their product strategies to develop innovative solutions that resonate with both healthcare providers and patients?
Establish a Comprehensive Product Strategy Framework
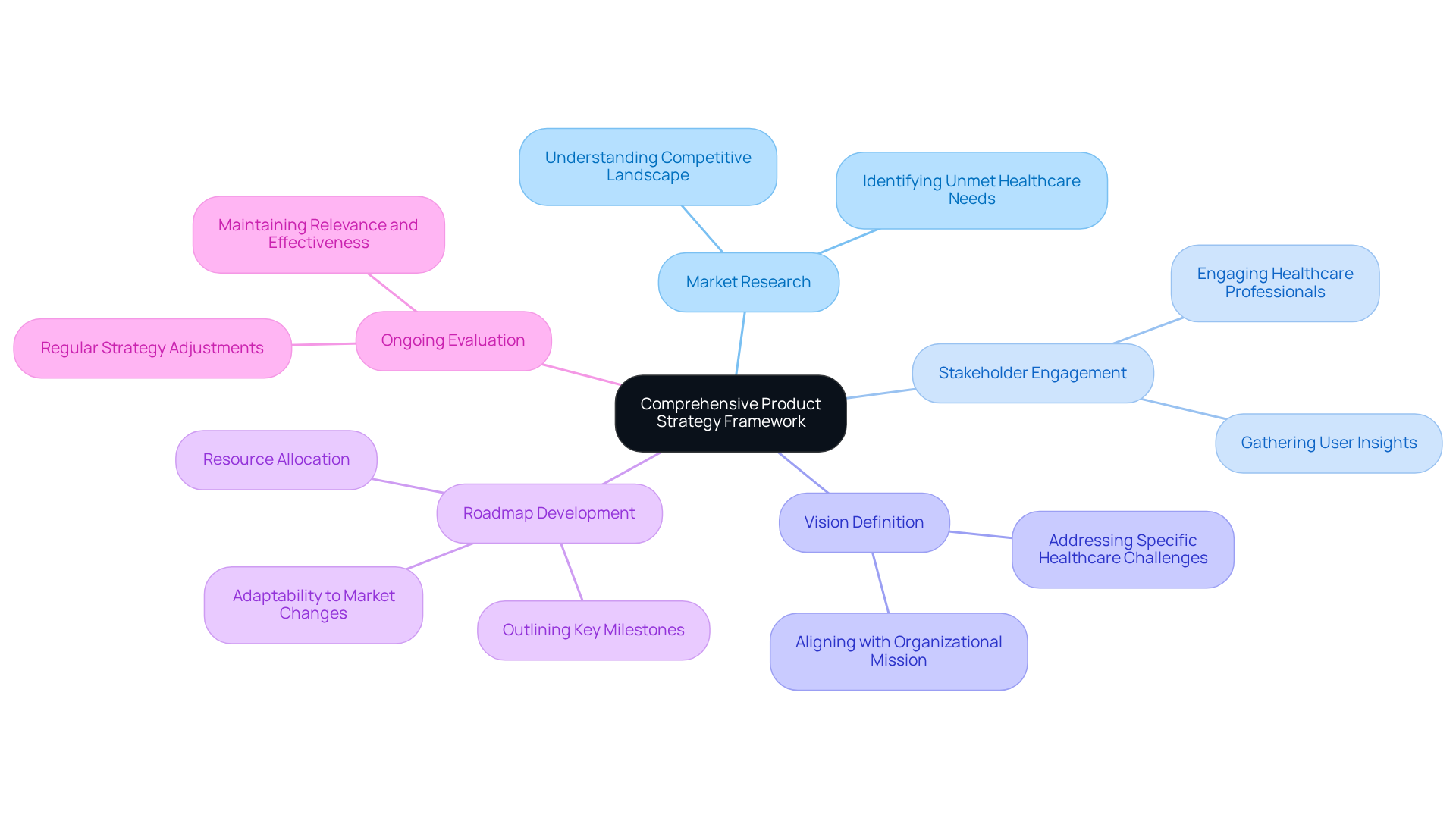
To establish a robust strategy framework, begin with comprehensive that focuses on understanding the competitive landscape and identifying unmet . Engaging with stakeholders, including healthcare professionals and potential users, is essential for gathering insights into their requirements and pain points. This engagement not only informs but also fosters connections that can enhance .
Next, define a clear vision for the offering that aligns with your organization's mission and addresses specific . This vision serves as a guiding principle for all subsequent decisions and actions throughout the development lifecycle. Industry leaders emphasize that a strong framework connects the vision for offerings, market research, user needs, and business objectives, ensuring that the strategy remains focused and effective.
Furthermore, develop a that outlines key milestones, timelines, and resource allocations. This roadmap should be adaptable to changes in market dynamics or regulatory requirements, reflecting the evolving landscape of healthcare technology. Regular evaluations and adjustments to the strategy are vital to maintain its relevance and effectiveness in achieving goals. Statistics reveal that nearly 5 billion people lack access to safe surgical care, underscoring the urgent need for to bridge these gaps. By prioritizing and leveraging insights from market research, can significantly enhance their product elements to better meet the needs of healthcare providers and patients alike.
Identify Key Components of Effective Product Strategies
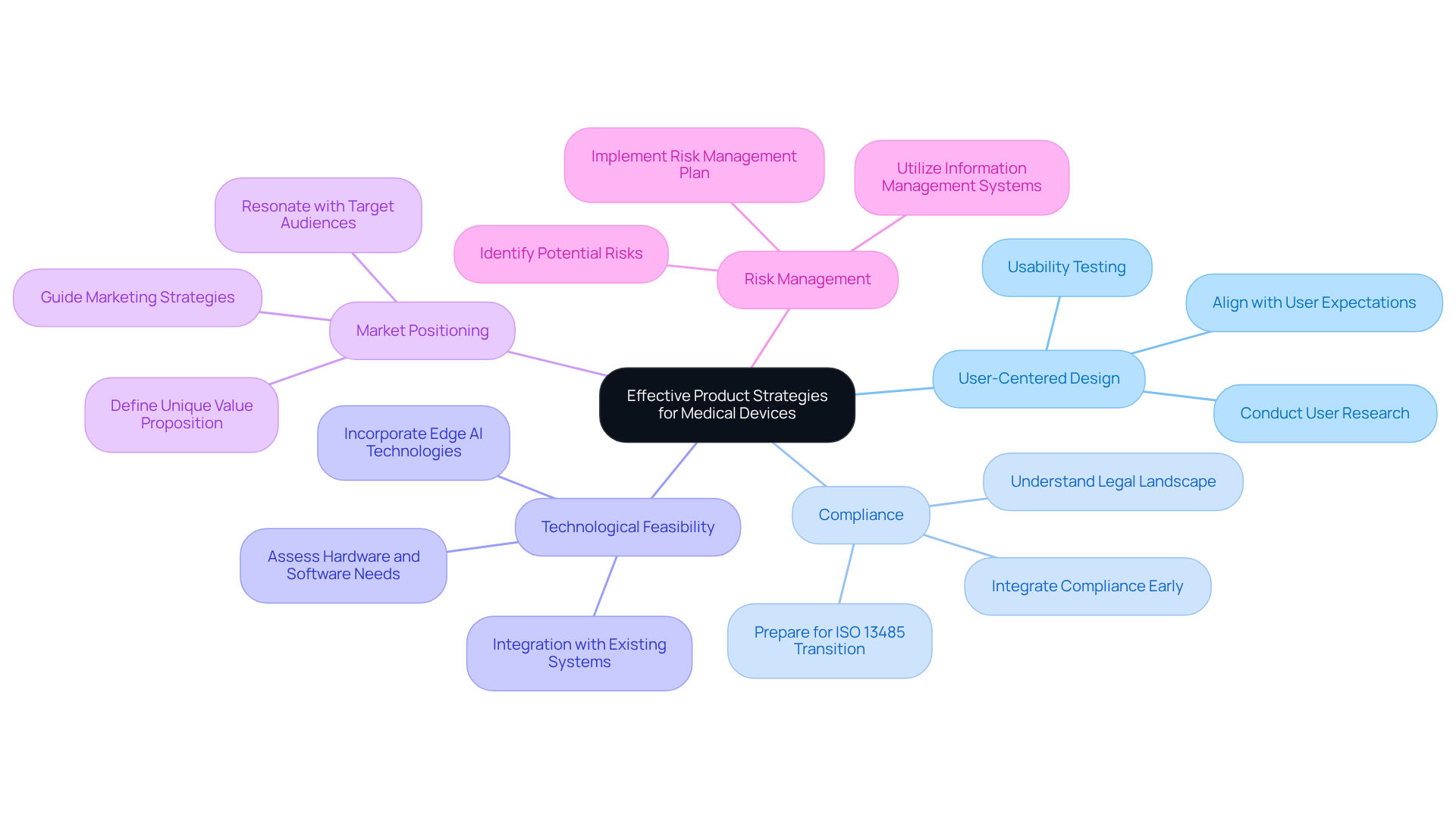
Effective product strategies for medical devices must encompass several critical components:
- : Prioritizing user needs is essential. Conducting thorough user research and usability testing ensures that the product aligns with the expectations and requirements of end-users, thereby enhancing adoption and satisfaction.
- Compliance: A comprehensive understanding of the legal landscape is vital. . Integrating compliance factors from the outset, including recognizing suitable oversight routes, assists in guaranteeing that the product fulfills all essential standards and promotes easier market entry. Manufacturers must ensure compliance with all applicable quality system requirements by this date. , offering documentation support that is essential for medical equipment startups.
- : Evaluating the technological capabilities needed for product creation is crucial. This includes assessing product elements such as hardware, software, and integration with existing systems, which informs design decisions and resource allocation, ultimately supporting successful implementation. ' prototype strategy streamlines hardware development and design adjustments, ensuring that aligns with user needs and regulatory requirements. Furthermore, incorporating Edge AI technologies can enhance functionality and user experience, indicating notable progress in contemporary healthcare.
- : Clearly defining the unique value proposition of the product is necessary for differentiation in a competitive market. This positioning guides marketing strategies and communication efforts, ensuring that the device resonates with target audiences.
- Risk Management: Implementing a robust is essential. This plan should identify potential risks throughout the development process and outline effective mitigation strategies, ensuring compliance and enhancing reliability. Ongoing monitoring and transparent reporting for connected and AI-driven products are critical to maintaining compliance and addressing risks effectively. Employing Information Management Systems (RIMS) can simplify the handling of compliance data, offering a central platform for oversight tracking and reporting.
can further enhance the effectiveness of these strategies, ensuring that medical device manufacturers are well-prepared to navigate the evolving regulatory landscape with the .
Translate Strategy into Actionable Development Plans
To effectively translate your product strategy into actionable development plans, consider the following steps:
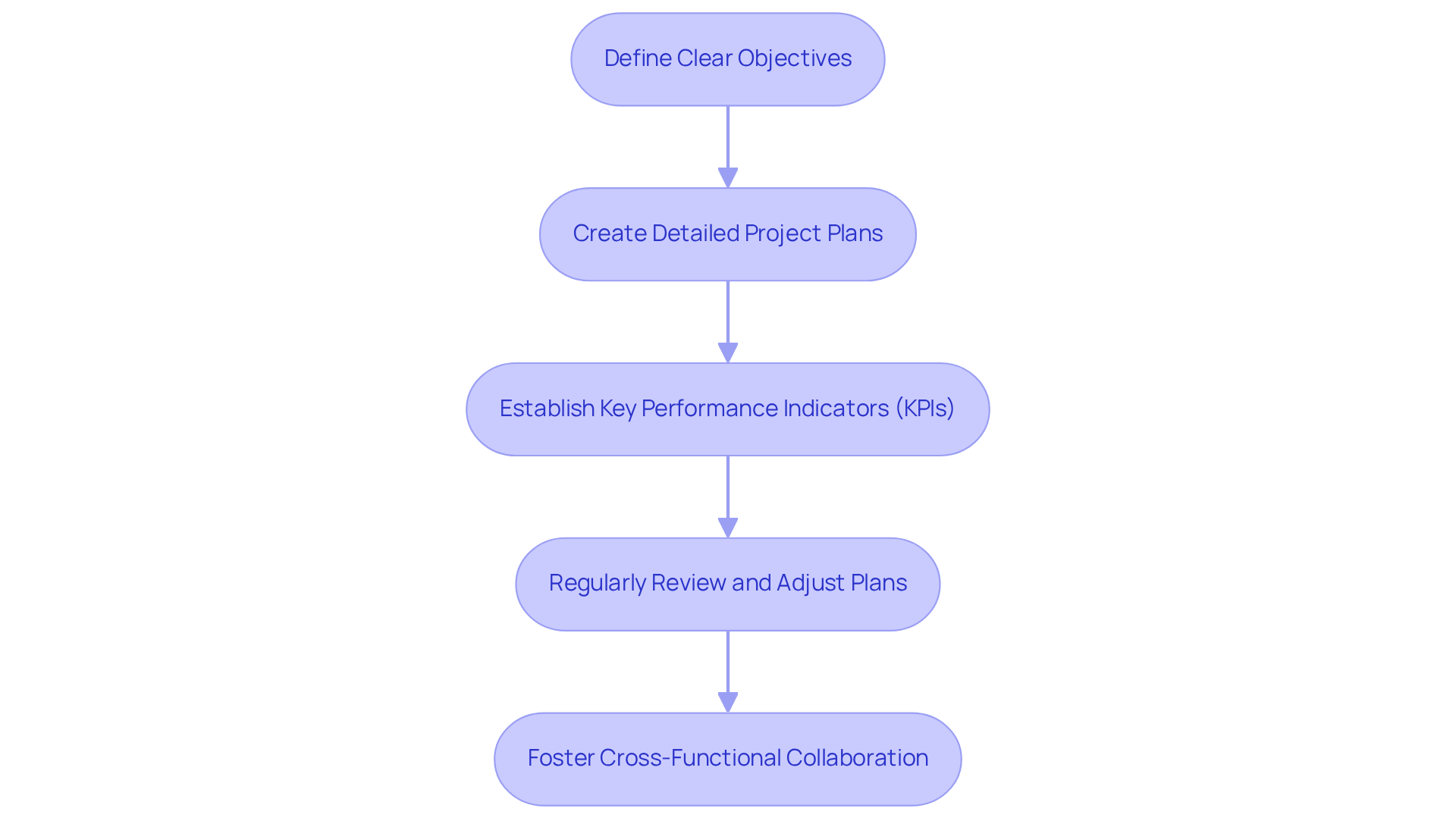
- : Break down the overarching product strategy into specific, measurable goals that can be monitored throughout the creation process. This clarity is essential for guiding teams and ensuring alignment with .
- : Develop comprehensive project plans that outline tasks, timelines, and responsibilities for each team member. Utilizing can enhance collaboration and communication, which are crucial in managing the complexities of .
- Establish : Identify KPIs that will measure progress and success. These indicators should align with strategic goals and provide insights into the efficiency of the development process, ensuring that teams remain focused on delivering .
- : Schedule consistent check-ins to assess progress against growth plans. Be prepared to adjust timelines and resources as necessary to address challenges or shifts in the market environment, which is vital for maintaining project momentum.
- Foster : Promote teamwork among various teams, including engineering, marketing, and compliance affairs. This integration ensures that all aspects of product creation are aligned, facilitating compliance with industry standards and enhancing overall project success.
Ensure Compliance with Regulatory Standards and Quality Control
To ensure compliance with and maintain quality control in , consider the following practices:
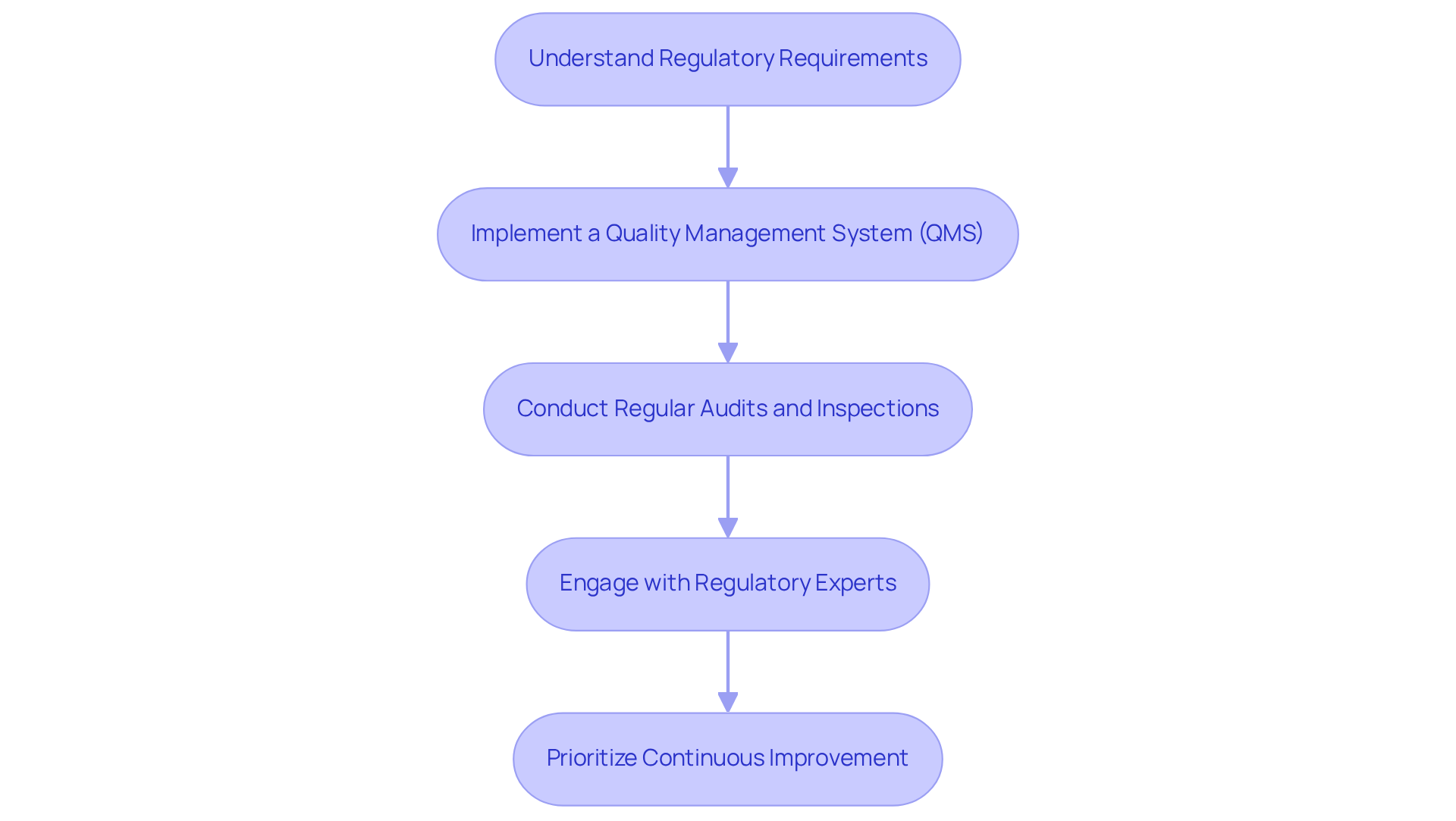
- Understand Regulatory Requirements: Familiarize yourself with relevant regulations, such as , that apply to your device. This foundational knowledge should inform all , ensuring they align with industry expectations.
- Implement a (QMS): Establish a robust QMS that outlines . Regular reviews and updates of this system are essential to ensure that product elements reflect changes in regulations and industry best practices, thereby enhancing operational efficiency.
- Conduct Regular Audits and Inspections: Arrange and assess the effectiveness of the QMS. Being prepared for external evaluations by governing entities is vital, as these assessments can significantly influence market entry and the credibility of product elements.
- Engage with Regulatory Experts: Collaborate with regulatory affairs professionals to navigate complex regulatory pathways. Their expertise ensures that all are accurate and complete, minimizing the risk of delays or non-compliance.
- Prioritize Continuous Improvement: Foster a culture of continuous improvement by regularly reviewing processes and outcomes. Utilize feedback from audits, user experiences, and market performance to make informed adjustments to the development process, ultimately .
Conclusion
Establishing an effective product strategy for medical devices is essential to address the pressing needs within the healthcare sector. A well-defined framework that incorporates thorough market research, user-centered design, compliance, and risk management enhances product development and ensures that devices meet the expectations of healthcare providers and patients alike. By aligning product strategies with organizational missions and healthcare challenges, firms can create solutions that significantly impact patient care.
Key components such as user-focused design, understanding regulatory requirements, technological feasibility, and market positioning are pivotal in shaping effective strategies. Each element contributes to a comprehensive approach that facilitates compliance, drives innovation, and enhances the overall quality of medical devices. Regular evaluations and adjustments to development plans further ensure that strategies remain relevant and effective in a rapidly evolving industry.
Ultimately, the significance of these strategies is profound. As the healthcare landscape continues to change, it is imperative for medical device manufacturers to prioritize the integration of user feedback, regulatory insights, and technological advancements into their product development processes. By doing so, they not only enhance their competitive edge but also contribute to improving access to safe and effective medical solutions for all. Embracing these best practices empowers organizations to navigate the complexities of medical device development and deliver impactful innovations that meet the urgent needs of the healthcare community.
Frequently Asked Questions
What is the first step in establishing a comprehensive product strategy framework?
The first step is to conduct comprehensive market research that focuses on understanding the competitive landscape and identifying unmet healthcare needs.
Why is stakeholder engagement important in the product strategy framework?
Engaging with stakeholders, including healthcare professionals and potential users, is essential for gathering insights into their requirements and pain points, which informs product development and fosters future collaboration.
How should the vision for the product offering be defined?
The vision for the offering should be clear and align with the organization's mission, addressing specific healthcare challenges. It serves as a guiding principle for all subsequent decisions and actions throughout the development lifecycle.
What role does a flexible roadmap play in the product strategy framework?
A flexible roadmap outlines key milestones, timelines, and resource allocations, and should be adaptable to changes in market dynamics or regulatory requirements, reflecting the evolving landscape of healthcare technology.
Why is it important to regularly evaluate and adjust the product strategy?
Regular evaluations and adjustments are vital to maintain the strategy's relevance and effectiveness in achieving goals, especially in a rapidly changing healthcare environment.
What statistic highlights the need for innovative solutions in healthcare?
Statistics reveal that nearly 5 billion people lack access to safe surgical care, underscoring the urgent need for innovative solutions to bridge these gaps.
How can medical equipment firms enhance their product elements?
By prioritizing user-focused design and leveraging insights from market research, medical equipment firms can significantly enhance their product elements to better meet the needs of healthcare providers and patients alike.
List of Sources
- Establish a Comprehensive Product Strategy Framework
- 6 Important Quotes from Medtech Leaders (https://mddionline.com/business/6-important-quotes-from-medtech-leaders)
- 37 Quotes on Health Care and Health Tech from 2016 (https://medium.com/@r_scott_munro/37-quotes-on-health-care-and-health-tech-from-2016-e34576edfc1f)
- glassbox.com (https://glassbox.com/blog/product-strategy-framework)
- 2026 Forecast: Medical manufacturing industry outlook (https://todaysmedicaldevelopments.com/article/2026-forecast-todays-medical-developments-industry-outlook)
- deliberatedirections.com (https://deliberatedirections.com/quotes-future-of-healthcare)
- Identify Key Components of Effective Product Strategies
- Navigating Medical Device Compliance in 2026 (https://regdesk.co/blog/guide-to-regulatory-compliance-for-medical-devices)
- Medical Device Compliance: 2025 Insights & 2026 Priorities (https://medenvoyglobal.com/blog/medical-device-compliance-2025-insights-2026-priorities)
- Regulatory Innovation as a Catalyst: How New Compliance Strategies Are Speeding Up Healthcare Technology Adoption - MedCity News (https://medcitynews.com/2026/02/regulatory-innovation-as-a-catalyst-how-new-compliance-strategies-are-speeding-up-healthcare-technology-adoption)
- Translate Strategy into Actionable Development Plans
- Medical Device Industry Facts, Trends and Statistics 2025 (https://arterexmedical.com/medical-device-industry-statistics)
- The 62 Most Inspiring Project Management Quotes (https://inloox.com/company/blog/articles/the-62-most-inspiring-project-management-quotes)
- 50 Project Management Quotes to Inspire You Before Your Next Project (https://toggl.com/blog/50-project-management-quotes)
- 34 of the Best Planning Quotes (https://projectmanager.com/blog/planning-quotes)
- The Top 200 Project Management & Planning Quotes (https://praxie.com/top-project-management-planning-quotes)
- Ensure Compliance with Regulatory Standards and Quality Control
- lumafield.com (https://lumafield.com/case-studies/case-study-maintaining-fda-compliance-with-validated-ct-technology)
- Experts' Views on FDA Regulatory Standards for Drug and High-Risk Medical Devices: Implications for Patient Care - PubMed (https://pubmed.ncbi.nlm.nih.gov/35138547)
- A guide to implementing and maintaining a medical device QMS (https://medicaldevicehq.com/articles/the-illustrated-guide-to-implementing-and-maintaining-a-medical-device-quality-management-system)
- The Ultimate QARA Guide to Medical Device Quality Management Systems | Scilife (https://scilife.io/blog/medical-device-quality-management-system-guide)
- A QMSR State of Mind: FDA Adopts New Inspection Approach for Medical Devices as Quality Management System Regulation Takes Effect | Insights | Ropes & Gray LLP (https://ropesgray.com/en/insights/alerts/2026/02/a-qmsr-state-of-mind-fda-adopts-new-inspection-approach-for-medical-devices)





