10 Essential Elements of Product Design for Medical Devices
Introduction The complex realm of medical device design integrates innovation, precision,...

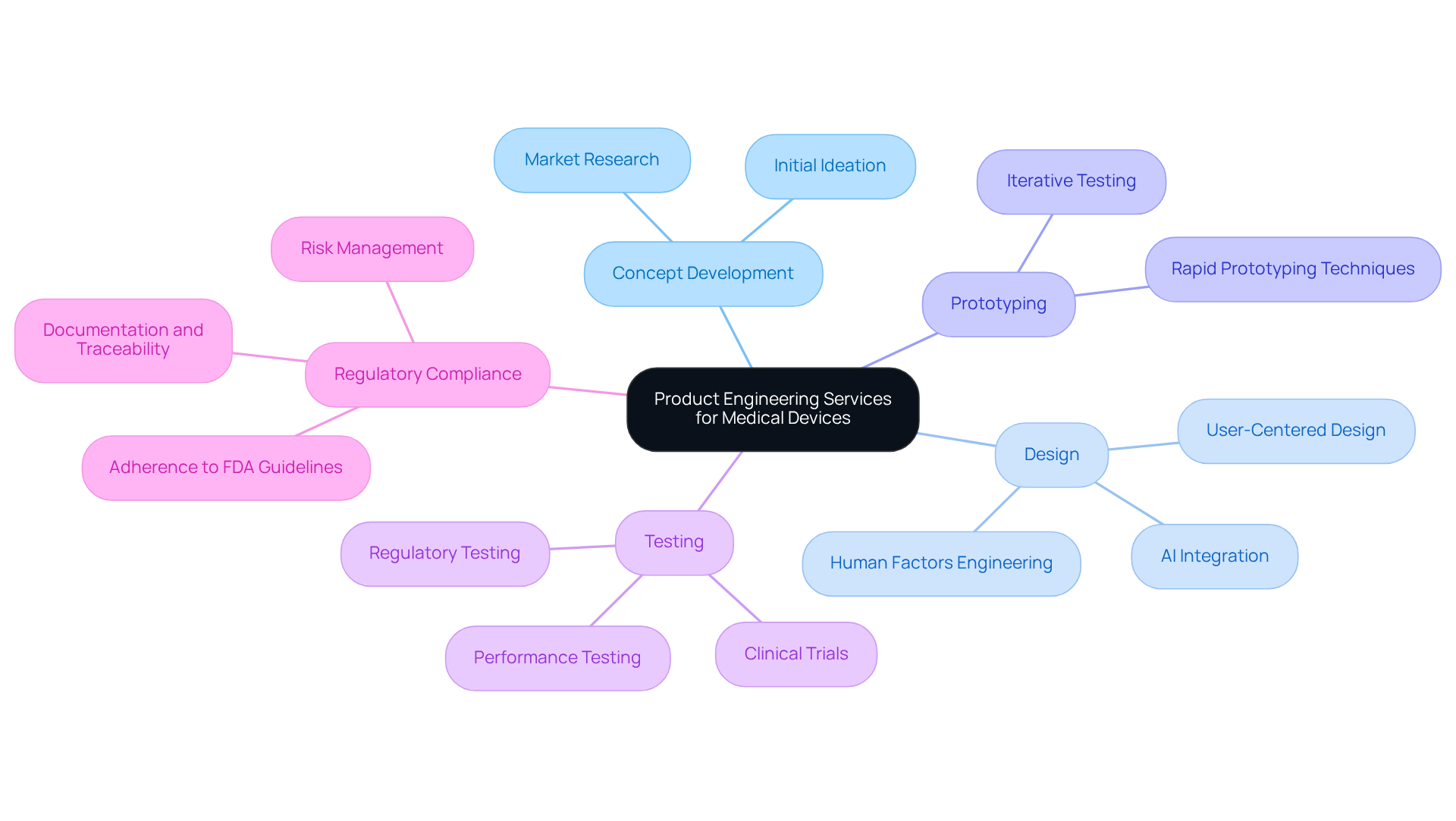
As the healthcare landscape evolves, manufacturers are confronted with the challenge of meeting increasing demands for innovative and compliant medical devices, driven by complex regulations and technological advancements. Product engineering services play a pivotal role in this context, guiding the development process from concept to market. Manufacturers must carefully evaluate which engineering partner aligns with their specific requirements. This article provides a comparative analysis of leading product engineering services in the USA, emphasizing their strengths and weaknesses to assist medical device manufacturers in making informed decisions.
In the rapidly evolving landscape of healthcare, product engineering services USA are essential for ensuring compliance and innovation. These services encompass a comprehensive array of activities, including:
They are crucial for guaranteeing that healthcare instruments meet strict safety and efficacy standards. The industry's increasing regulatory complexity and the need for thorough clinical trials are driving demand for innovative engineering solutions. By 2026, the integration of advanced technologies such as AI and IoT will be vital, prompting companies to seek partners adept at navigating these complexities.
Key players in this field often provide integrated solutions that span the entire product lifecycle, from initial ideation to market launch. For instance, the FDA's evolving regulatory framework underscores the importance of adherence, particularly as equipment becomes more software-intensive. Case studies illustrate that implementing Model-Based Systems Engineering (MBSE) enhances product lifecycle management through structured requirement mapping and early risk identification. This approach not only streamlines regulatory submissions but also ensures that each modification aligns with compliance requirements.
Statistics show an increasing demand for healthcare equipment product engineering services USA, fueled by the necessity for dependable and efficient instruments in healthcare environments. As companies work to fulfill these requirements, regulatory adherence is critical in healthcare product engineering, as it directly impacts market readiness and product safety. Therefore, it is essential for producers to partner with knowledgeable associates who can provide high-quality, compliant solutions. Without strategic partnerships in engineering, companies risk falling behind in a competitive market.
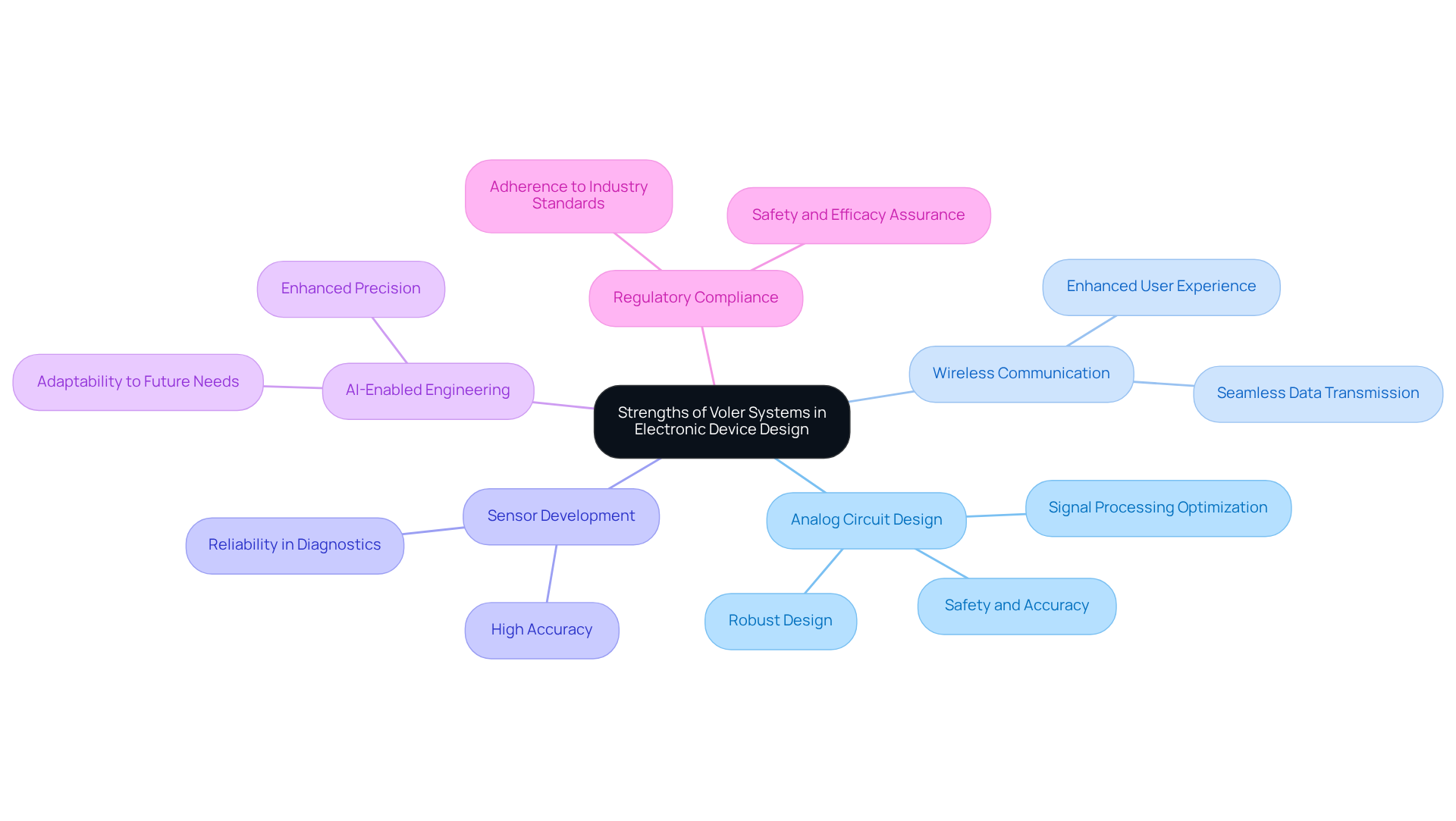
With over 44 years of experience in the electronic equipment sector, the company has established itself as a leader through its technical expertise and commitment to quality. The company's key strengths are as follows:
These strengths not only enhance product quality but also significantly contribute to customer satisfaction. By prioritizing quality and regulatory compliance, the company positions itself as an indispensable partner for healthcare product manufacturers navigating the complexities of the industry.
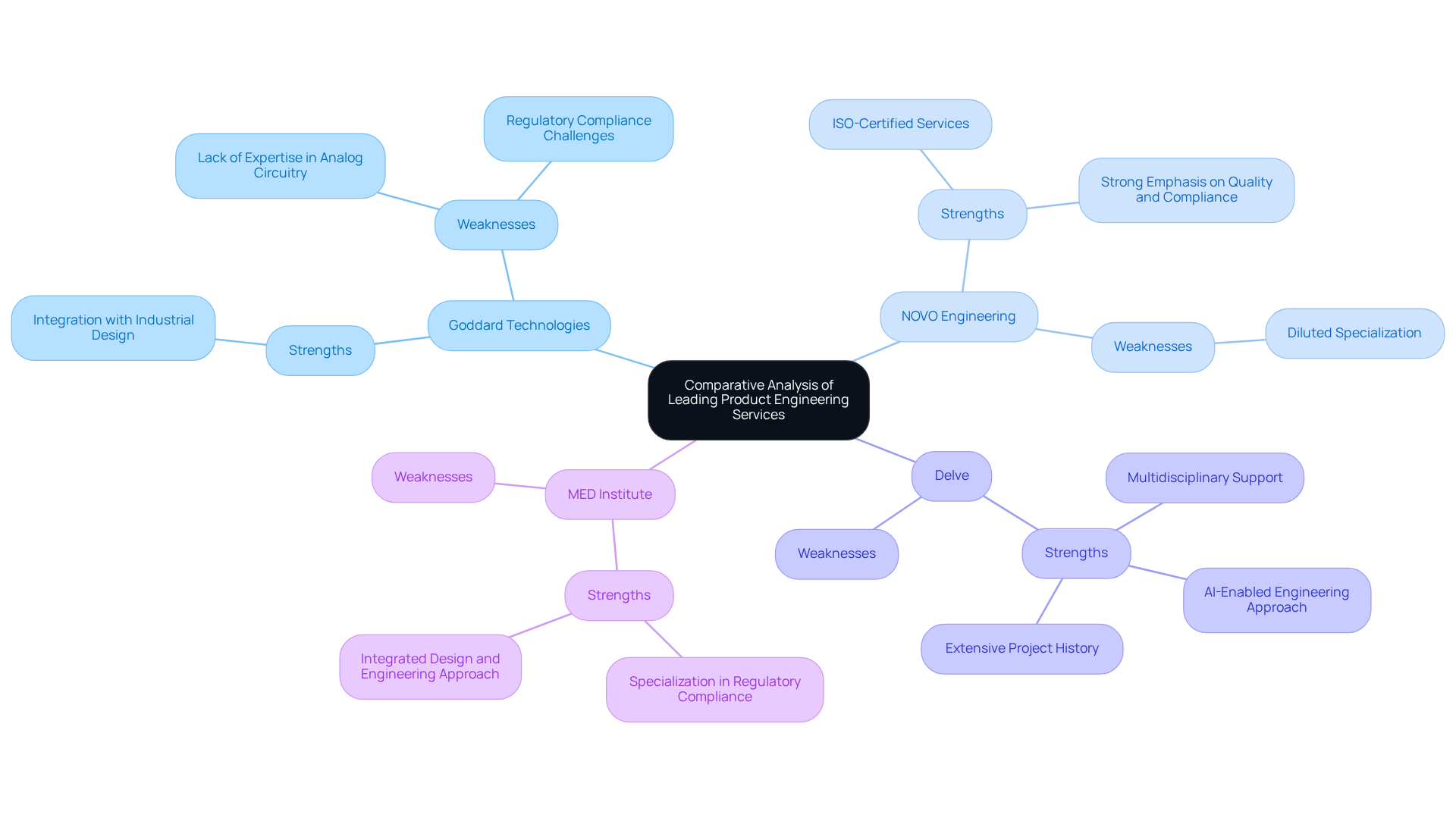
In the competitive landscape of healthcare technology engineering, a comparative analysis reveals the strengths and weaknesses of various firms, including Voler Systems:
This analysis underscores the importance of specialized expertise in navigating the complexities of the medical device engineering sector.
Manufacturers in the medical device industry face critical decisions when selecting engineering service providers, balancing quality, compliance, and cost-effectiveness.
Goddard Technologies:
NOVO Engineering:
Delve:
MED Institute:
This analysis equips manufacturers with a clear framework to evaluate engineering service options based on their specific project needs, budget constraints, and desired outcomes. Ultimately, the choice of an engineering partner can significantly influence the success of medical device projects, making careful evaluation essential.
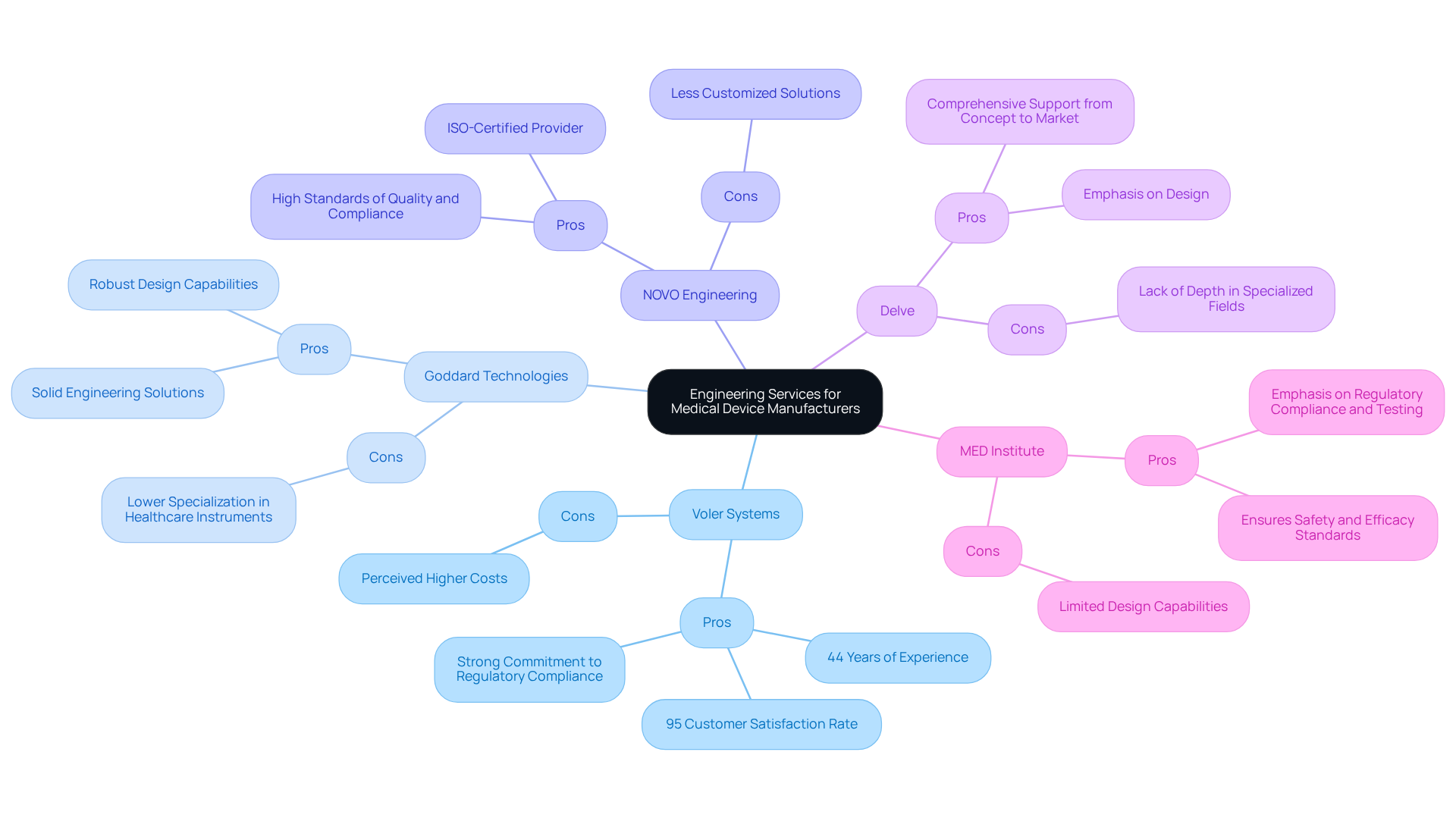
In the intricate realm of medical device manufacturing, the role of product engineering services is critical to ensuring innovation and compliance with safety standards. Manufacturers face a critical need for specialized expertise in concept development, design, prototyping, and regulatory compliance. Partnering with proficient engineering firms like Voler Systems can significantly enhance a manufacturer's ability to bring compliant and effective medical devices to market.
The article delves into the strengths and weaknesses of various engineering service providers, emphasizing the unique advantages that specialized firms offer. Voler Systems, with its extensive experience and focus on regulatory compliance, stands out as a reliable partner for medical device manufacturers. Other firms may excel in specific areas but often lack the comprehensive expertise essential for the highly regulated healthcare sector. This comparative analysis illustrates that choosing the right partner involves more than cost; it requires alignment with a firm that comprehends the unique challenges of the medical device industry.
Ultimately, the choice of a product engineering service provider can have far-reaching implications for the success of medical device projects. Manufacturers are encouraged to evaluate their options carefully, considering factors such as compliance, specialization, and proven track records. By making informed decisions and fostering strategic partnerships, companies can not only meet regulatory demands but also enhance their competitiveness in a rapidly evolving market. Ultimately, the right engineering partner can be the difference between a product's success and its failure in a competitive landscape.
What are product engineering services for medical devices?
Product engineering services for medical devices encompass a range of activities including concept development, design, prototyping, testing, and regulatory compliance to ensure that healthcare instruments meet safety and efficacy standards.
Why are product engineering services important in the healthcare industry?
They are crucial for ensuring compliance with strict safety and efficacy standards, addressing the increasing regulatory complexity, and meeting the demand for innovative engineering solutions in healthcare.
What technologies are expected to impact product engineering services by 2026?
The integration of advanced technologies such as artificial intelligence (AI) and the Internet of Things (IoT) is expected to be vital for product engineering services by 2026.
What is Model-Based Systems Engineering (MBSE) and how does it benefit product lifecycle management?
MBSE is an approach that enhances product lifecycle management through structured requirement mapping and early risk identification, streamlining regulatory submissions and ensuring compliance with each modification.
What role does regulatory compliance play in healthcare product engineering?
Regulatory compliance is critical as it directly impacts market readiness and product safety, making it essential for producers to partner with knowledgeable associates for high-quality, compliant solutions.
What are the consequences of not having strategic partnerships in product engineering?
Without strategic partnerships in engineering, companies risk falling behind in a competitive market, as they may struggle to meet regulatory requirements and innovate effectively.
What is driving the demand for healthcare equipment product engineering services?
The demand is driven by the necessity for dependable and efficient instruments in healthcare environments, alongside the increasing complexity of regulatory requirements.
