Introduction
In the rapidly advancing field of medical technology, effective communication and data transmission are paramount for device interoperability. The integration of IP QAM modulators is crucial for effective communication and data transmission among devices. These modulators enhance operational efficiency and are vital for delivering timely patient information that impacts care outcomes.
Healthcare providers aim to optimize device performance, yet engineers often struggle with the intricate specifications of IP QAM modulator integration. Identifying best practices and strategies is essential for success in this critical aspect of medical device development.
Understand IP QAM Modulators and Their Role in Medical Devices
In the realm of medical technology, the IP QAM modulator plays an increasingly pivotal role for high-speed data transmission. These modulators convert digital information into analog signals, enabling efficient communication between devices and systems. In healthcare, where timely and accurate information transfer is essential, IP QAM modulators enable the transmission of substantial volumes of diagnostic details, telemetry, and patient monitoring insights. Without effective modulation, healthcare providers may struggle to receive timely patient data, leading to potential delays in care.
The incorporation of the IP QAM modulator significantly boosts performance by enhancing transmission rates and reducing latency. For instance, in telemedicine applications, these modulators allow for timely medical responses, ensuring that patient information reaches healthcare providers in real-time. Engineers involved in designing and creating healthcare instruments must understand the operational principles of the IP QAM modulator. Understanding these principles is vital for engineers to select the right modulator for specific applications and to troubleshoot integration challenges effectively. As the healthcare landscape evolves in 2026, failure to grasp these principles may result in inefficient device integration and compromised patient care.
To ensure the success of these projects, engineers must develop essential skills such as:
- Planning and executing project strategies
- Assessing project viability
- Recognizing when a project is in trouble to intervene effectively
Voler Systems utilizes AI-driven engineering and vast expertise in developing health-related products, ensuring that projects are completed on schedule and within budget. By utilizing advanced FPGA design solutions, Voler Systems revolutionizes contemporary healthcare, offering intelligent, high-performance solutions that satisfy the requirements of today's health technology landscape. As healthcare technology advances, the mastery of the IP QAM modulator will be essential for engineers to ensure optimal patient outcomes.
Implement Best Practices for Integration and Compliance
To effectively integrate IP QAM modulators into medical devices, engineers should adhere to several best practices that align with current regulatory standards and address compliance challenges:
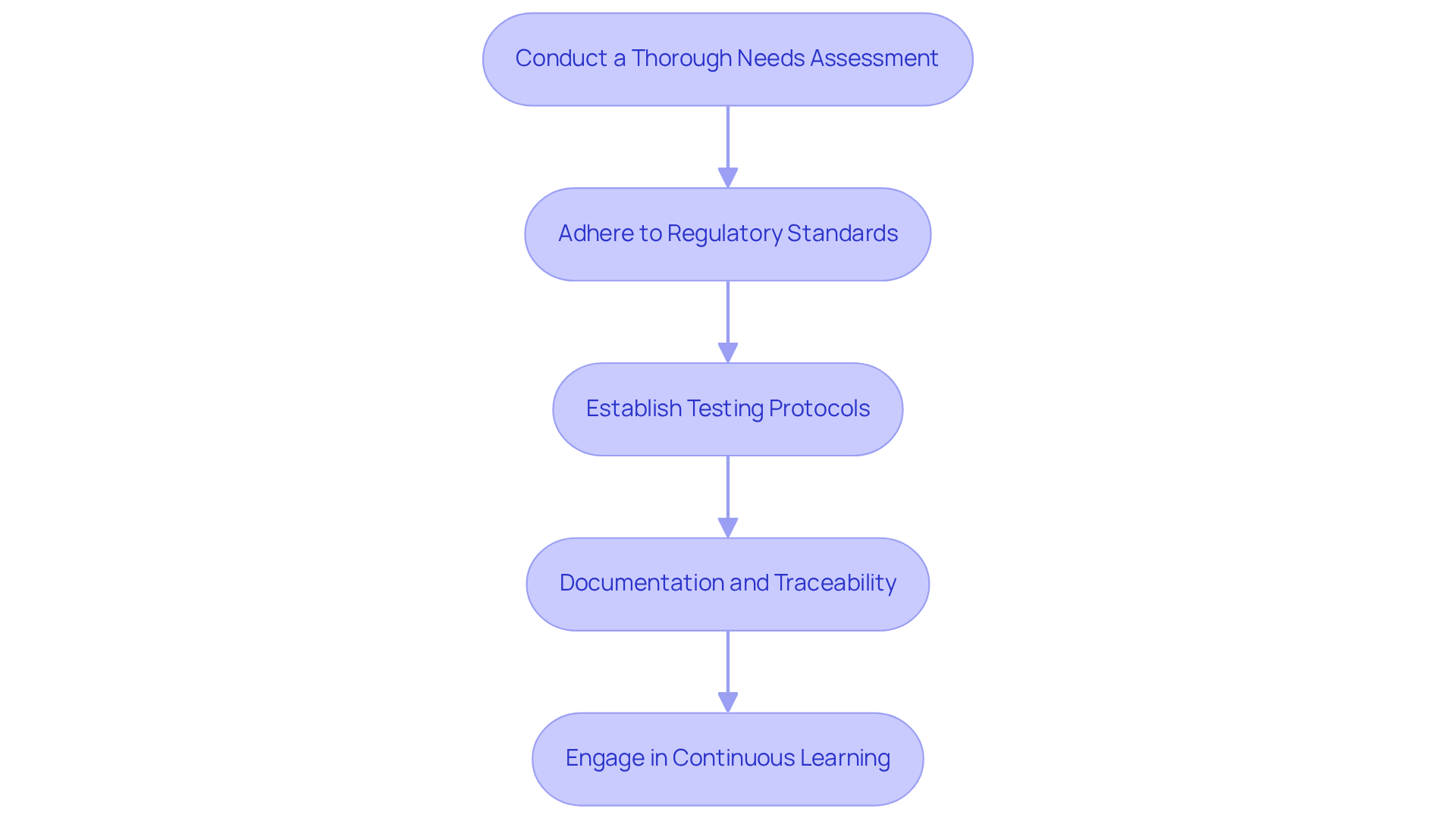
- Conduct a Thorough Needs Assessment: Begin by evaluating the specific requirements of the healthcare instrument, including data transmission rates, power consumption, and environmental conditions. This evaluation is essential for choosing the suitable IP QAM modulator that meets the operational requirements of the system.
- Adhere to Regulatory Standards: Familiarize yourself with essential regulatory standards such as ISO 13485 and IEC 62304. Compliance with these standards is essential. It ensures that design and integration processes lead to smoother approvals from regulatory bodies, particularly in light of the upcoming Quality Management System Regulation (QMSR) changes. The FDA's shift from QSR to QMSR signifies one of the most important structural regulatory changes in US medical quality oversight in decades, making compliance with these standards more vital than ever.
- Establishing comprehensive testing protocols is crucial for evaluating the performance of the IP QAM modulator within the system. This evaluation should focus on signal integrity, data throughput, and compliance with electromagnetic compatibility (EMC) requirements, which are crucial for meeting regulatory expectations. Be aware that compliance activities for healthcare product manufacturers can extend timelines significantly, impacting project schedules, so planning these protocols early is crucial.
- Documentation and Traceability: Maintain meticulous documentation throughout the integration process. Documentation must include design specifications, testing results, and compliance checks, all of which are essential for regulatory submissions and audits. The FDA's emphasis on documentation under QMSR highlights the importance of thorough record-keeping, as records created before February 2, 2026, will be reviewed under QMSR.
- Engage in Continuous Learning: Stay informed about advancements in modulation technologies and evolving regulatory requirements. Continuous education and training are key to enhancing the team's capability to integrate new technologies effectively while ensuring ongoing compliance with regulatory standards. Delays in compliance could jeopardize submission timelines or complicate FDA audits, so proactive learning is vital.
Integrating an IP QAM modulator into medical devices offers substantial benefits that enhance performance across various applications, particularly in innovative wearable medical devices and cardiac monitoring.
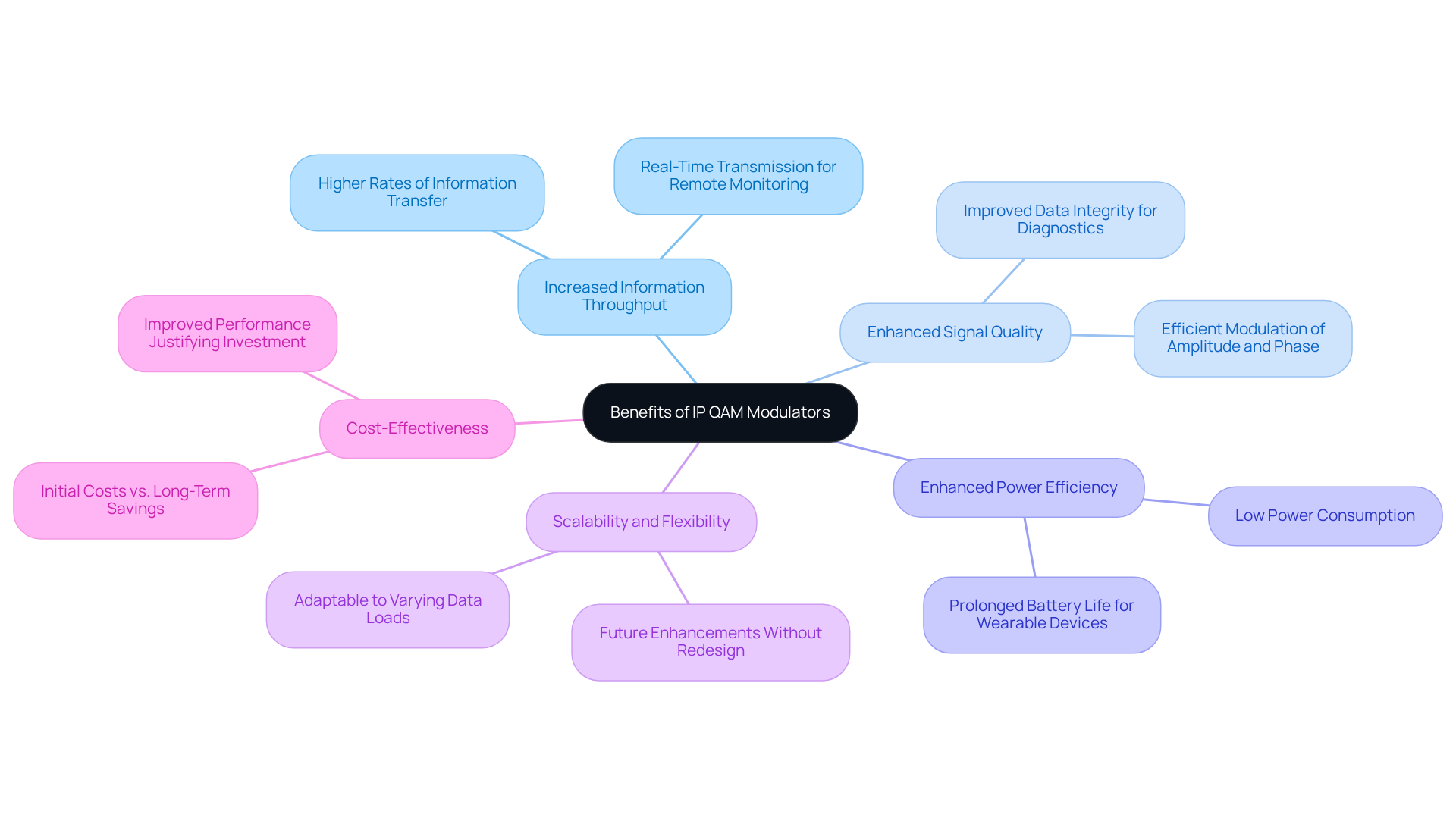
- The IP QAM modulator enables increased information throughput by allowing higher rates of information transfer compared to conventional modulation methods, which is particularly advantageous for applications needing real-time transmission, such as remote patient monitoring and telehealth services. Yousra Lamrani emphasizes that selecting the right modulation types significantly enhances receiver sensitivity, underscoring the importance of information throughput in healthcare settings.
- Enhanced Signal Quality: By modulating both amplitude and phase, the IP QAM modulator conveys information more efficiently, resulting in improved signal quality and lower error rates. This is vital in healthcare, where data integrity is key for accurate diagnostics and patient care. The examination of healthcare equipment receiver sensitivity performance utilizing various modulation methods emphasizes how these innovations can improve healthcare quality in wireless NB-IoT networks, especially in cardiac monitoring solutions.
- Enhanced power efficiency is a feature of many IP QAM modulators, which are specifically engineered for low power consumption, making them ideal for battery-operated healthcare devices. This efficiency not only prolongs battery life but also reduces the necessity for frequent recharging or replacement, which is crucial in continuous monitoring situations, such as those utilized in cutting-edge wearable technology.
- The IP QAM modulator offers scalability and flexibility, as it can be easily scaled to handle varying data loads, making it suitable for a diverse range of healthcare applications. This adaptability enables future enhancements without requiring substantial redesign efforts, ensuring longevity in utility, particularly in the changing landscape of healthcare innovation.
- Cost-effectiveness: The initial costs of the IP QAM modulator technology can be a barrier for some manufacturers. However, the long-term benefits-including reduced operational costs and improved performance of equipment-often justify the expense. This investment leads to significant operational savings over time, making IP QAM modulators a viable option for manufacturers like Voler Systems, aiming to improve their product offerings while ensuring compliance with stringent regulatory standards.
Ultimately, the strategic implementation of IP QAM modulators can redefine operational efficiency and patient outcomes in the medical device sector.
Troubleshoot Common Integration Challenges Effectively
Integrating the ip qam modulator into medical equipment presents various technical challenges that require careful consideration. Here are common issues and effective troubleshooting strategies:
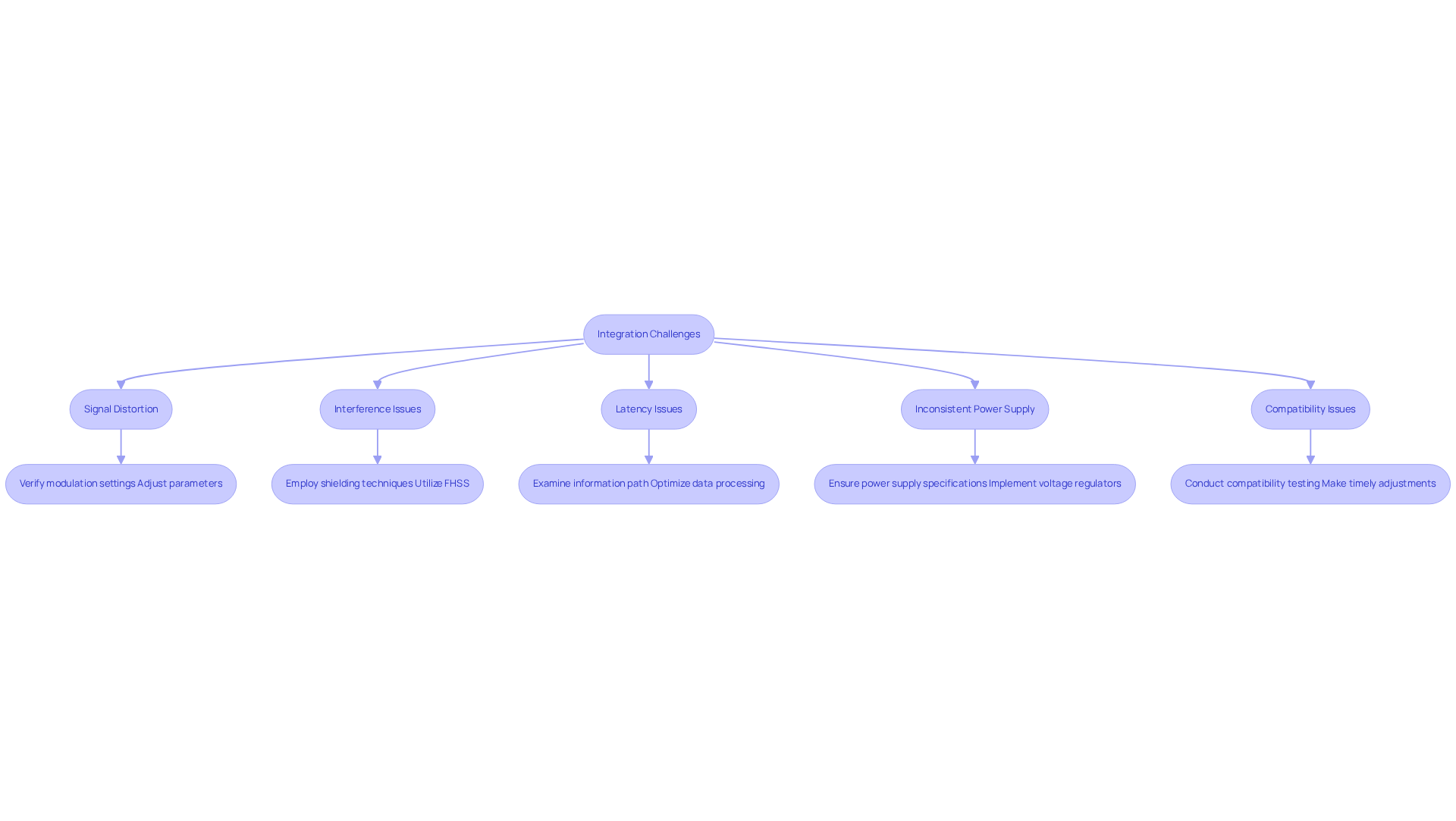
- Signal Distortion: Distorted transmitted signals can arise from improper modulation settings. Verify that the modulator is configured correctly for the specific application, and adjust modulation parameters as needed to resolve distortion issues.
- Interference Issues: Medical instruments often function in settings with considerable electromagnetic interference, which can disturb signal integrity. Employing shielding techniques and utilizing frequency-hopping spread spectrum (FHSS) can effectively mitigate these effects, enhancing signal reliability.
- Latency Issues: Increased latency can hinder the timely transmission of information, negatively impacting device performance. To troubleshoot, examine the complete information path from the sensor to the modulator, identifying potential bottlenecks. Optimizing data processing algorithms and ensuring efficient data handling can significantly reduce latency.
- Inconsistent power supply can disrupt the functionality of the ip qam modulator, leading to performance degradation. Ensure that the power supply meets the required specifications and consider implementing voltage regulators to stabilize the power input, ensuring consistent performance.
- Compatibility issues with the ip qam modulator and other components in the medical apparatus are crucial. Conduct thorough compatibility testing during the design phase to identify potential issues early, allowing for timely adjustments and ensuring seamless integration.
Common Pitfalls: Manufacturers often overlook the importance of environmental assessments and fail to account for potential interference sources during the design phase. Identifying and mitigating these common pitfalls can significantly reduce the risk of delays and enhance integration efficiency.
Expected Impact: By resolving these integration challenges, manufacturers can significantly enhance device reliability and ensure better patient outcomes, reinforcing the importance of rigorous testing and validation in the development process. Leveraging Voler Systems' expertise in embedded systems design can further support manufacturers in overcoming these challenges.
Conclusion
Integrating IP QAM modulators into medical devices is essential for advancing healthcare delivery and improving patient outcomes. These modulators are crucial for ensuring high-speed data transmission, which is vital for real-time patient monitoring and timely medical responses. As healthcare technology evolves, it's essential for engineers and manufacturers to grasp the operational principles and best practices of IP QAM modulators to enhance patient outcomes.
Key insights from this article highlight the importance of thorough needs assessments, adherence to regulatory standards, and the establishment of robust testing protocols. Engaging in continuous learning and meticulous documentation are also critical to navigating the complexities of compliance and integration. By leveraging the benefits of IP QAM modulators, such as enhanced signal quality, increased information throughput, and improved power efficiency, manufacturers can significantly elevate the performance of medical devices.
In closing, mastering these integration techniques is essential for ensuring that medical devices not only meet current demands but also adapt to future challenges in patient care. As engineers and manufacturers embrace these advanced technologies and adhere to best practices, they play a crucial role in driving innovation and ensuring that medical devices meet the ever-increasing demands of patient care.
Frequently Asked Questions
What is the role of IP QAM modulators in medical devices?
IP QAM modulators play a crucial role in medical devices by converting digital information into analog signals, enabling efficient communication and high-speed data transmission between devices and systems.
Why is timely information transfer important in healthcare?
Timely and accurate information transfer is essential in healthcare to ensure that healthcare providers receive substantial volumes of diagnostic details, telemetry, and patient monitoring insights without delays, which could compromise patient care.
How do IP QAM modulators enhance performance in healthcare applications?
IP QAM modulators enhance performance by boosting transmission rates and reducing latency, allowing for real-time medical responses and ensuring that patient information reaches healthcare providers promptly.
What skills must engineers develop to work with IP QAM modulators in healthcare?
Engineers must develop skills in planning and executing project strategies, assessing project viability, and recognizing when a project is in trouble to intervene effectively.
How does Voler Systems contribute to healthcare technology?
Voler Systems utilizes AI-driven engineering and advanced FPGA design solutions to develop health-related products, ensuring that projects are completed on schedule and within budget, thereby revolutionizing contemporary healthcare.
Why is understanding the operational principles of IP QAM modulators important for engineers?
Understanding the operational principles is vital for engineers to select the appropriate modulator for specific applications and to troubleshoot integration challenges effectively, ensuring optimal device performance and patient care.
List of Sources
- Understand IP QAM Modulators and Their Role in Medical Devices
- Multifunctional Modular IPQAM Modulator Market Set to Surge - Key Insights You Must Know (https://openpr.com/news/4474426/multifunctional-modular-ipqam-modulator-market-set-to-surge)
- Top Five Health Care Trends for 2026: How New Technology Is Transforming Patient Care (https://aanp.org/news-feed/top-five-health-care-trends-for-2026-how-new-technology-is-transforming-patient-care)
- 6 healthcare tech trends for 2026 (https://medtronic.com/en-us/our-company/stories/6-healthcare-tech-trends-for-2026.html)
- Quantum technology opens new frontiers in medical devices - Medical Technology | Issue 86 | May 2025 (https://medical-technology.nridigital.com/medical_technology_may25/quantum_technology_opens_new_frontiers_in_medical_devices)
- 8 Breakthrough Technology Trends That Will Transform Healthcare In 2026 (https://forbes.com/sites/bernardmarr/2025/10/27/the-8-biggest-healthcare-technology-trends-to-watch-in-2026)
- Implement Best Practices for Integration and Compliance
- FDA Implements Quality Management System Regulation for Medical Devices (https://aabb.org/news-resources/news/article/2026/02/04/fda-implements-quality-management-system-regulation-for-medical-devices)
- QMSR 2026 Explained: FDA Quality Management System Regulation & ISO 13485 Alignment Guide (https://sushvin.com/qmsr-2026-fda-quality-management-system-regulation.html)
- FDA’s New QMSR Rule for 2026: What Medical Device Manufacturers Must Do Now (https://theoris.com/fdas-new-qmsr-rule-for-2026-what-medical-device-manufacturers-must-do-now)
- U.S. FDA Reissues Medical Device Cybersecurity Guidance to Align with QMSR (https://intertek.com/blog/2026/04-15-medical-device-cybersecurity-guidance)
- FDA Quality Management System Regulation 2026: Changes to 21 CFR Part 820 and Compliance Requirements (https://proximacro.com/news/fda-quality-management-system-regulation-2026-changes-to-21-cfr-part-820-and-compliance-requirements)
- Leverage Benefits of IP QAM Modulators for Enhanced Device Performance
- Performance Analysis on Modulation Techniques for Medical Devices Sensitivity in Wireless NB-IoT Network (https://papers.ssrn.com/sol3/papers.cfm?abstract_id=3852562)
- Transforming healthcare: The impact of telehealth and telemedicine - Mayo Clinic Press (https://mcpress.mayoclinic.org/research-innovation/transforming-healthcare-the-impact-of-telehealth-and-telemedicine)
- Connected Nation's newest study reveals impact of remote monitoring in patients' perceptions of telehealth (https://prnewswire.com/news-releases/connected-nations-newest-study-reveals-impact-of-remote-monitoring-in-patients-perceptions-of-telehealth-302589188.html)
- Troubleshoot Common Integration Challenges Effectively
- Medical Device Integration: Progress & Challenges (https://signifyresearch.net/insights/medical-device-integration-progress-potential-and-persistent-challenges)
- The Biggest Quality Challenges for Medical Device Companies in 2025 - Corena (https://co-re-na.com/the-biggest-quality-challenges-for-medical-device-companies-in-2025)
- All About Signal Distortion in Transmission Impairment (https://resources.pcb.cadence.com/blog/2022-all-about-signal-distortion-in-transmission-impairment)
- US government probes medical devices for possible cyber flaws (https://foxnews.com/tech/us-government-probes-medical-devices-for-possible-cyber-flaws)
- Navigating Key Challenges in Medical Device Design (https://medtechbusinessrevieweurope.com/news/navigating-key-challenges-in-medical-device-design-nwid-555.html)





