Create an Effective Prototyping Plan for Medical Devices
Learn how to create an effective prototyping plan for medical devices to enhance design...

In the rapidly evolving landscape of healthcare technology, the design of Internet of Things (IoT) devices is crucial for enhancing patient care and operational efficiency. Effective IoT device design relies not only on the integration of essential components but also on strict adherence to regulatory standards and innovative testing methodologies.
As the market for IoT healthcare solutions is projected to grow significantly, a key challenge arises: how can designers ensure that their devices are both functional and compliant while remaining user-friendly?
This article explores best practices for creating effective IoT devices in the medical sector, providing insights that can lead to improved patient outcomes and increased market confidence.
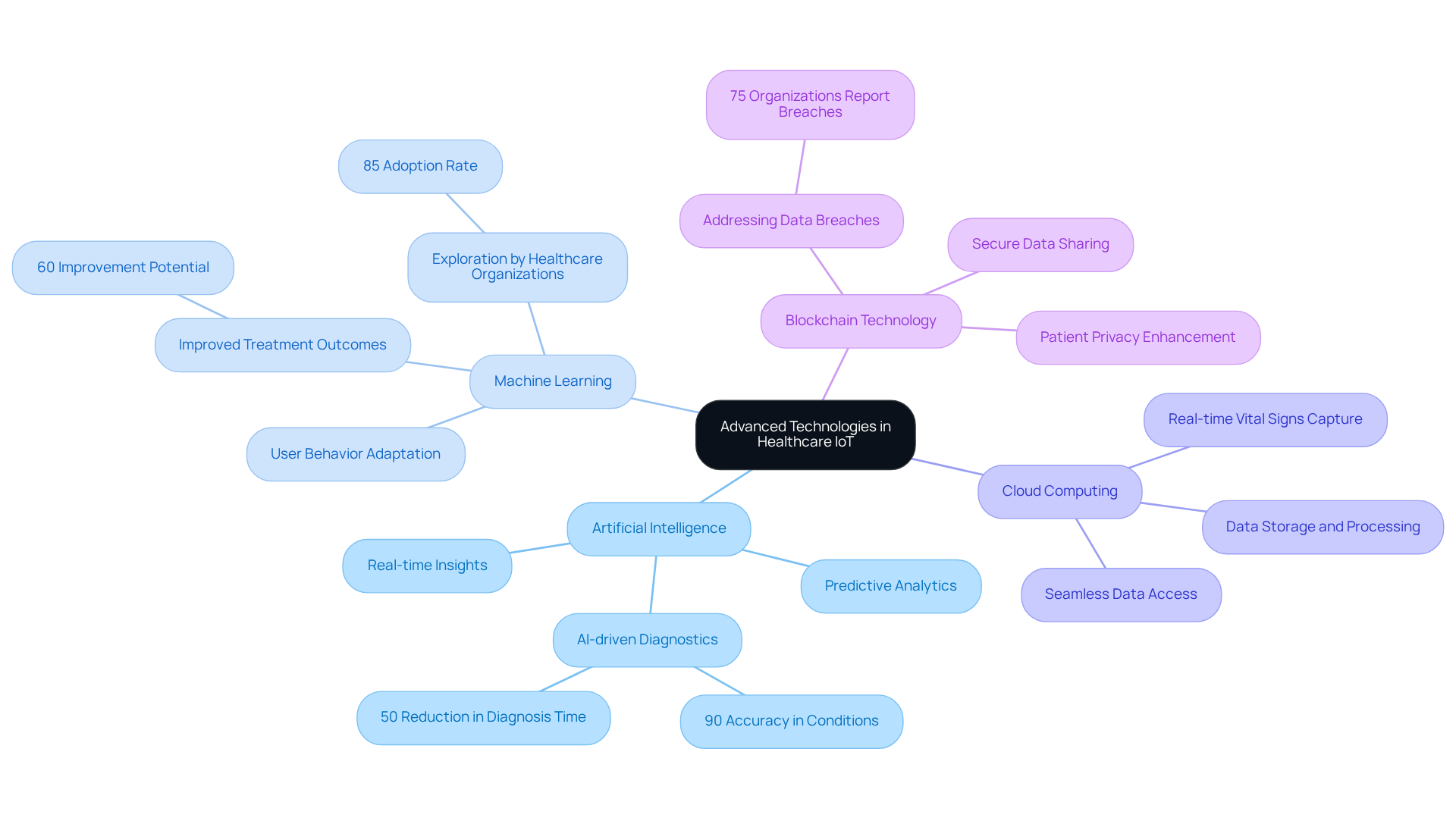
In designing IoT tools for healthcare applications, incorporating essential components is vital for ensuring both functionality and reliability. Key components include:
By carefully selecting and integrating these elements, designers can achieve IoT device design that adheres to the stringent standards of the healthcare sector, ensuring safety, effectiveness, and improved patient outcomes. Voler Systems' aids manufacturers in navigating these challenges, providing the expertise necessary to develop innovative and secure healthcare products.
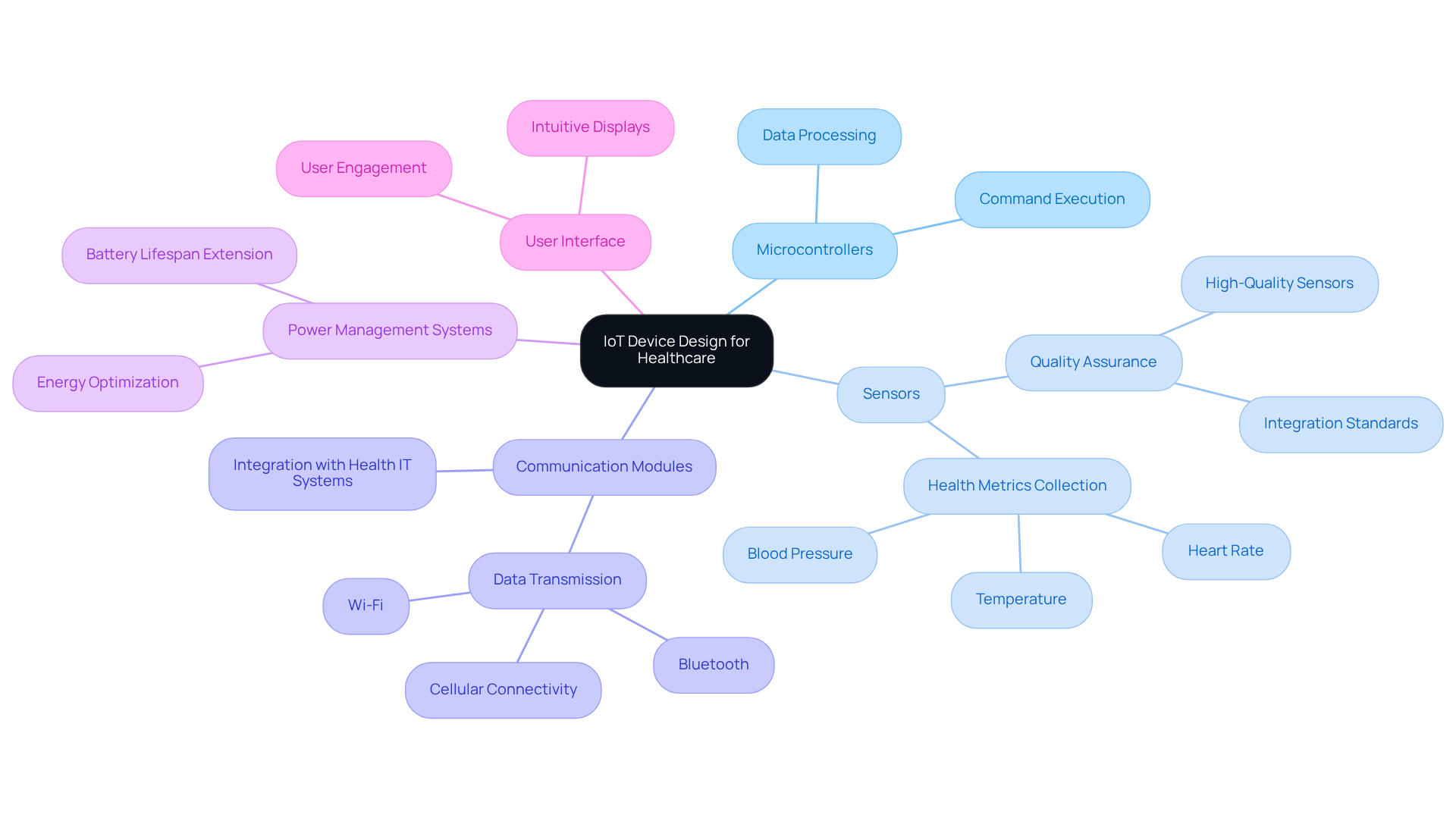
Ensuring adherence to regulations and maintaining quality control are essential aspects of IoT device design for healthcare instruments. Key practices include:
By emphasizing regulatory adherence and quality assurance in IoT device design, companies can reduce risks and boost the reliability of their IoT healthcare products, ultimately resulting in better patient outcomes and market confidence.
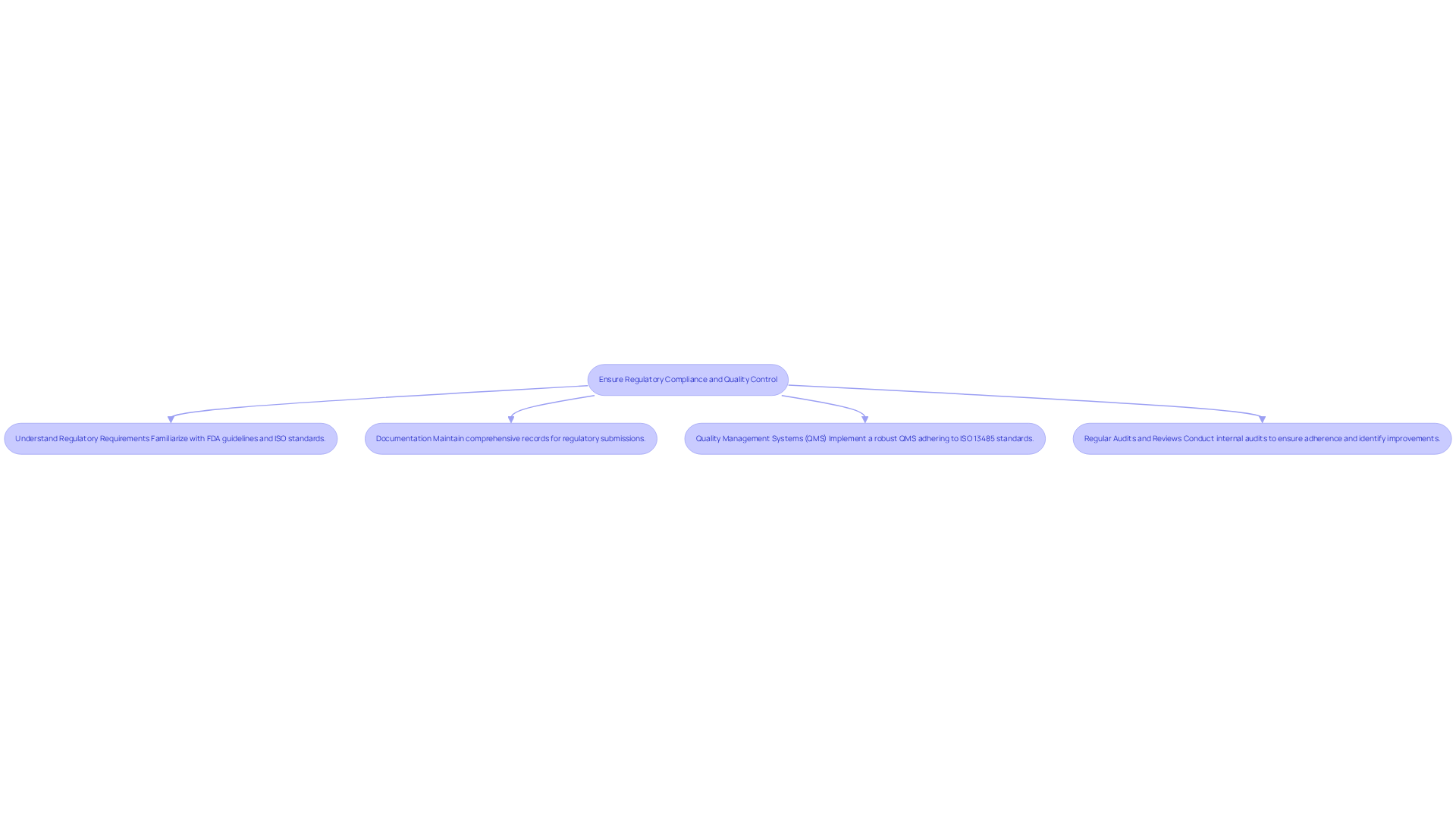
Prototyping and testing are critical components of the IoT device design process, particularly in the medical equipment sector, where adherence to standards and quality are paramount. Effective strategies include:
By implementing these strategies, designers can enhance the reliability and user-friendliness of their IoT device design for healthcare instruments, ultimately leading to improved patient outcomes and ensuring compliance with industry regulations.
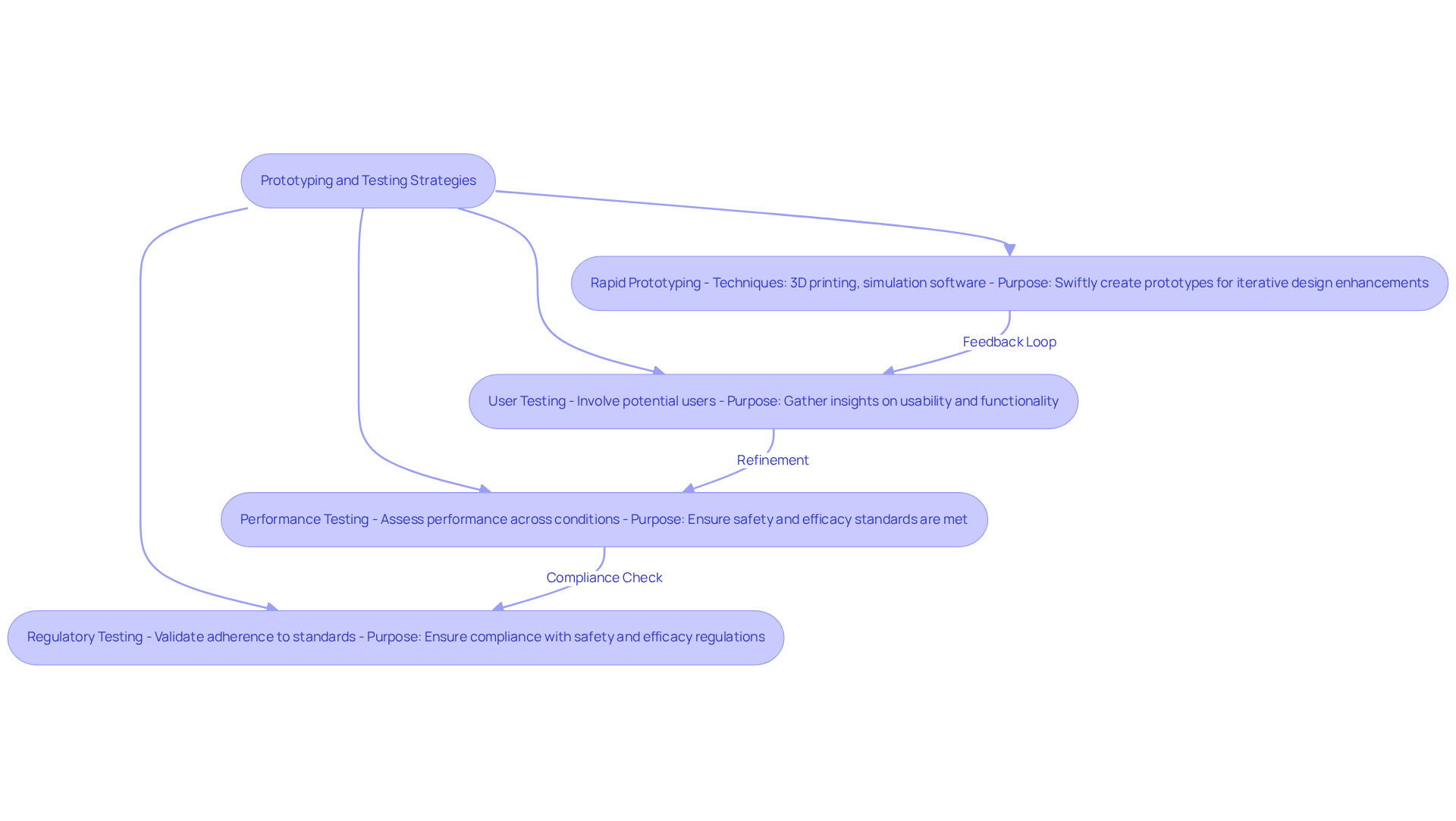
Integrating advanced technologies into IoT device design for healthcare equipment can lead to significant improvements in functionality and user experience. The following technologies play a crucial role in this transformation:
By leveraging these advanced technologies, medical device designers can focus on IoT device design to develop innovative solutions that not only address current healthcare needs but also anticipate future demands, ultimately transforming patient management and treatment planning.
Effective IoT device design in the medical field relies on a thorough understanding of essential components, regulatory compliance, and the integration of advanced technologies. Incorporating microcontrollers, sensors, communication modules, power management systems, and user interfaces is fundamental to creating reliable and functional healthcare devices. Moreover, adherence to regulatory requirements and quality control measures is critical for ensuring safety and efficacy, ultimately leading to improved patient outcomes.
Key insights from this discussion highlight the importance of:
Additionally, the necessity of prototyping and testing strategies, such as user testing and performance evaluations, is emphasized as integral to refining designs and ensuring compliance with regulatory expectations. The incorporation of cutting-edge technologies like AI, machine learning, cloud computing, and blockchain not only enhances the functionality of these devices but also addresses the evolving needs of healthcare delivery.
In conclusion, the landscape of IoT device design for medical applications is rapidly evolving, driven by technological advancements and stringent regulatory demands. Embracing these best practices is essential for manufacturers aiming to develop innovative, secure, and effective healthcare solutions. By prioritizing these strategies, stakeholders can significantly improve patient care and contribute to a more efficient healthcare system, underscoring the vital role of IoT in transforming medical device design and functionality.
What are the key components of IoT device design for healthcare applications?
The key components include microcontrollers, sensors, communication modules, power management systems, and user interfaces.
What role do microcontrollers play in IoT devices?
Microcontrollers act as the brain of the device, processing data and executing commands, making them essential for medical IoT devices.
Why are sensors important in healthcare IoT devices?
Sensors are crucial for collecting health metrics such as heart rate, temperature, and blood pressure. High-quality sensors ensure precise data gathering, which is vital for effective health monitoring.
How do communication modules function in IoT devices?
Communication modules facilitate data transmission between the device and external systems, such as cloud servers or healthcare providers, using options like Wi-Fi, Bluetooth, and cellular connectivity.
What is the significance of power management systems in IoT devices?
Power management systems optimize energy usage for battery-powered equipment, extending battery lifespan and addressing the demand for ultra-low power solutions in medical applications.
How does Voler Systems contribute to IoT device design in healthcare?
Voler Systems provides expertise in sensor integration and advanced power management strategies, ensuring that healthcare instruments meet rigorous standards for safety and effectiveness.
What is the importance of user interfaces in IoT devices?
Effective user interfaces allow users to engage with the device intuitively, enhancing user experience and compliance with health monitoring protocols.
How can manufacturers benefit from Voler Systems' IoT design consulting?
Manufacturers can navigate the challenges of IoT device design with Voler Systems' consulting, which provides the expertise necessary to develop innovative and secure healthcare products.
