Prototype Definition Engineering: Key Insights for Medical Devices
Discover the essentials of prototype definition engineering for effective medical device...

In an era where precision and efficiency in medical technology are critical, the role of IoT simulation tools has become increasingly significant. At the heart of this transformation is the application of IoT simulation tools that enable engineers to develop precise virtual models of medical devices prior to testing. This article examines the key benefits of these tools, revealing how they streamline product development and enhance compliance with stringent regulatory standards. As the medical device development landscape grows more complex, teams must determine how to select and integrate these simulation resources effectively to maximize benefits and minimize risks.
IoT modeling resources play a pivotal role in advancing medical equipment design, allowing engineers to create virtual representations prior to physical prototyping. These resources facilitate the assessment of various scenarios, including:
While mitigating the costs and risks associated with real-world testing. For instance, models can identify potential design or functionality issues early in the development process, enabling necessary adjustments. This proactive strategy not only reduces time-to-market but also improves the quality and reliability of the final product.
Furthermore, modeling resources assist in fulfilling regulatory standards by offering detailed documentation of testing procedures and results, which is essential for healthcare products that must comply with strict safety requirements. Real-world applications, such as the Living Heart Project by Dassault Systèmes, demonstrate how digital twins can personalize cardiac care by simulating patient responses to various treatments, optimizing care without risk.
Moreover, the incorporation of modeling technologies has been shown to represent around 45% of preclinical testing protocols for Class II and III healthcare instruments in North America and Europe, highlighting their significance in ensuring compliance and improving product effectiveness. The integration of modeling technologies not only streamlines compliance but also elevates the effectiveness of healthcare products in a competitive market.
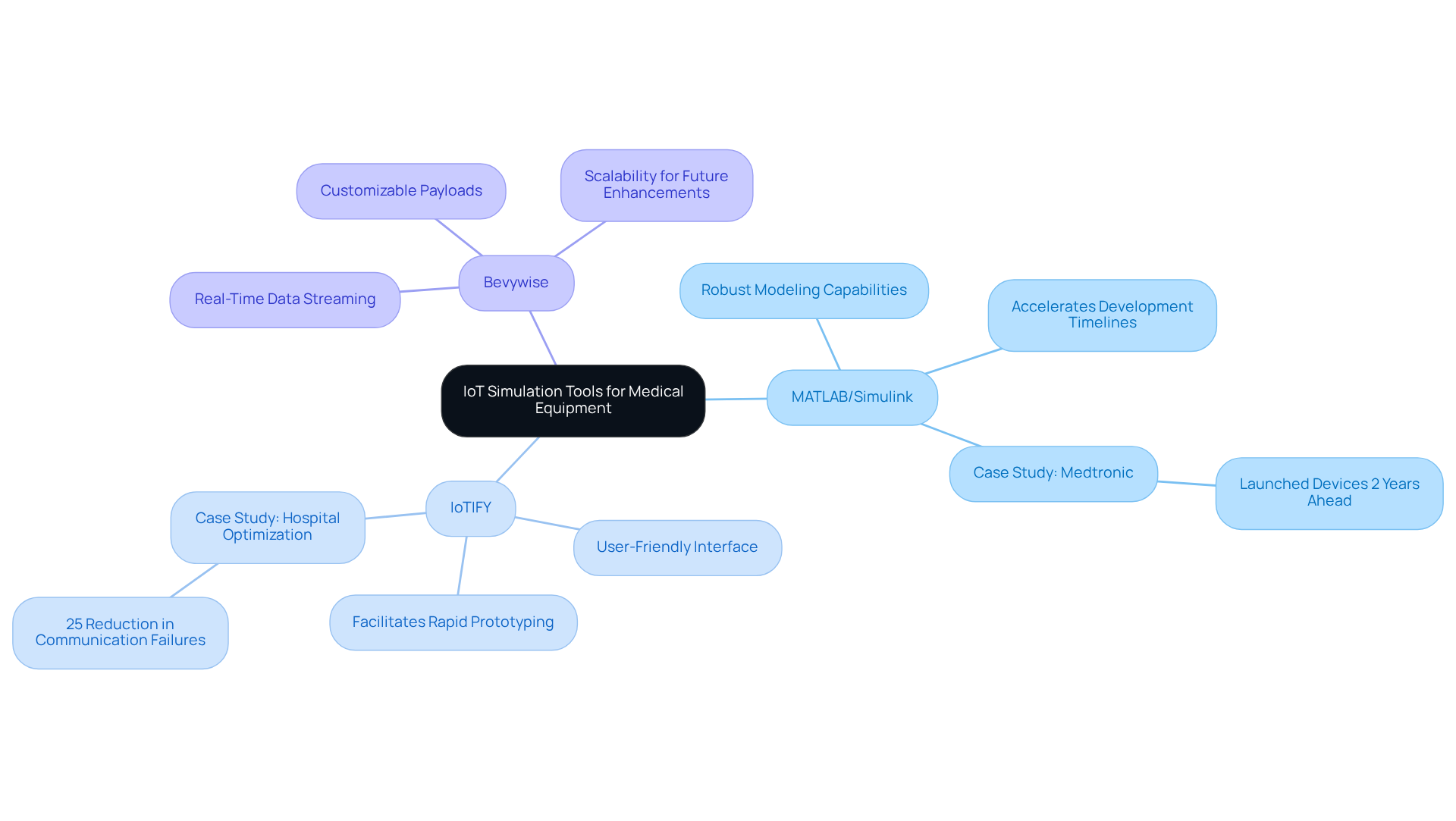
Selecting the right IoT simulation tools for the development of medical equipment is a complex task that demands careful consideration of various factors. MATLAB/Simulink is recognized for its robust modeling capabilities, making it particularly suitable for complex systems requiring detailed modeling. Research indicates that using MATLAB/Simulink can significantly accelerate development timelines, with companies such as Medtronic leveraging these tools to launch devices two years ahead of competitors.
In this context, IoTIFY and Bevywise are essential platforms that function as IoT simulation tools, providing user-friendly interfaces that facilitate rapid prototyping and testing. Bevywise, for example, supports real-time data streaming and customizable payloads, improving the efficiency and accuracy of simulations for developers. Evaluating the scalability of these resources is vital, as they must accommodate future enhancements and additional functionalities as projects evolve.
Engaging with case studies from successful healthcare product developers can provide valuable insights into the practical effectiveness of specific resources. For instance, a hospital employed IoT simulation tools to optimize its operations, achieving a 25% reduction in communication failures. Such examples underscore the importance of selecting appropriate modeling resources to ensure reliability and compliance in healthcare product development. Ultimately, the right choice of modeling resources can significantly influence the success and efficiency of healthcare product development.
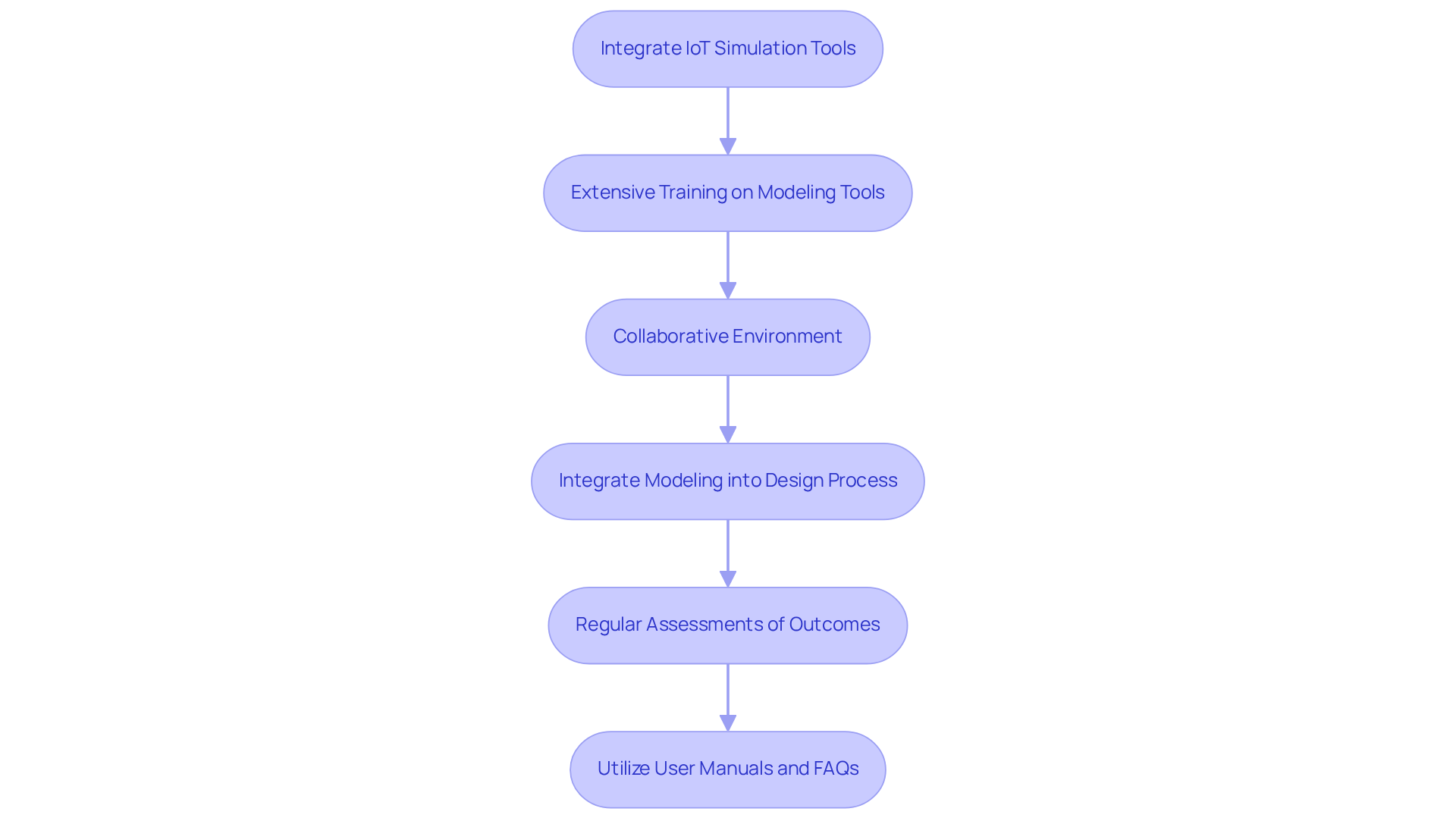
A systematic strategy is crucial for effectively incorporating IoT simulation tools into the medical device development process. Extensive training on selected modeling tools is essential for team members to fully leverage their capabilities. A collaborative environment, where engineers, designers, and regulatory experts work together, fosters innovative solutions and accelerates problem resolution. Integrating modeling into the iterative design process enables ongoing feedback and enhancement, ensuring that the final product meets user requirements and regulatory standards. Regular assessments of modeling outcomes help identify trends and pinpoint areas for improvement, promoting ongoing improvement within the development team. This structured approach not only improves product quality but also significantly reduces time to market, as manufacturers often struggle with safety, efficacy, and compliance in a competitive landscape. For further guidance, user manuals and FAQs can provide valuable insights into common errors in manufacturing tests, ensuring that teams are well-equipped to navigate the complexities of documentation compliance with increased confidence.
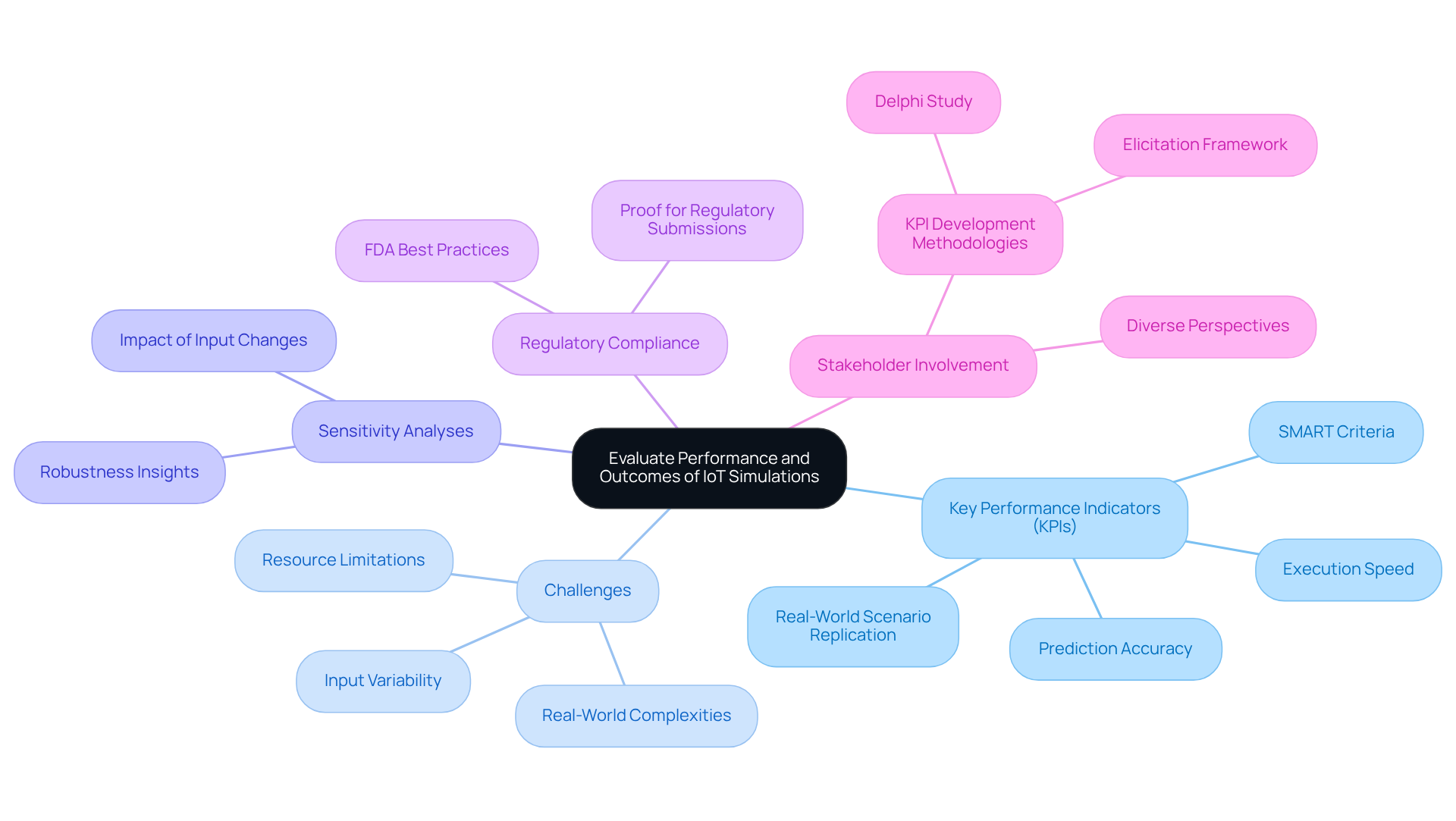
Evaluating the performance of IoT models at Voler Systems requires a meticulous examination of the accuracy and reliability of our IoT simulation tools. Establishing key performance indicators (KPIs) is essential. These indicators must adhere to the SMART criteria: they should be specific, measurable, attainable, relevant, and time-bound. KPIs such as execution speed, prediction accuracy, and the capacity to replicate real-world scenarios should be established to evaluate the effectiveness of the models.
Consistently contrasting modeled results with real test data can assist in recognizing differences and opportunities for enhancement. However, achieving accurate simulations with IoT simulation tools can be challenging due to varying input parameters and real-world complexities. Additionally, conducting sensitivity analyses can reveal how changes in input parameters affect outcomes, providing deeper insights into the robustness of the design.
Recording these assessments not only helps in improving the modeling process but also acts as essential proof for regulatory submissions, showing that the apparatus has been comprehensively tested and validated prior to entering the market. At Voler Systems, we prioritize quality and timely delivery, ensuring that our testing practices align with FDA best practices for measuring and evaluating the performance of AI-enabled health technologies.
Ongoing performance monitoring is emphasized, and stakeholder involvement in developing KPIs for digital health interventions is crucial, as it ensures that the indicators reflect diverse perspectives and needs. This rigorous approach is particularly important given that the accuracy of IoT simulation tools can significantly impact the reliability of medical devices. Inaccurate simulations not only jeopardize patient safety but also risk non-compliance with industry regulations, underscoring the critical need for rigorous testing and validation.
The integration of IoT simulation tools is crucial for the advancement of medical device development, as it allows for thorough testing and refinement before physical production. Adopting these tools enables organizations to identify design flaws, ensure regulatory compliance, and deliver higher-quality products to market more efficiently.
Throughout the article, key insights have been shared regarding the selection and integration of IoT simulation tools. From understanding their critical role in product development to evaluating their performance through established metrics, the discussion emphasizes the importance of choosing the right tools - such as MATLAB/Simulink, IoTIFY, and Bevywise - to meet specific project needs. Furthermore, the necessity of a collaborative approach among teams and ongoing assessments of simulation outcomes has been highlighted as vital for achieving successful results.
In summary, integrating IoT simulation tools is a strategic necessity for medical device developers in a competitive landscape. By embracing these technologies, organizations can improve patient safety, enhance product reliability, and ensure compliance with industry regulations. The commitment to advanced simulation tools will define the future of innovation and excellence in medical device development, shaping the industry's landscape for years to come.
What is the role of IoT simulation tools in product development?
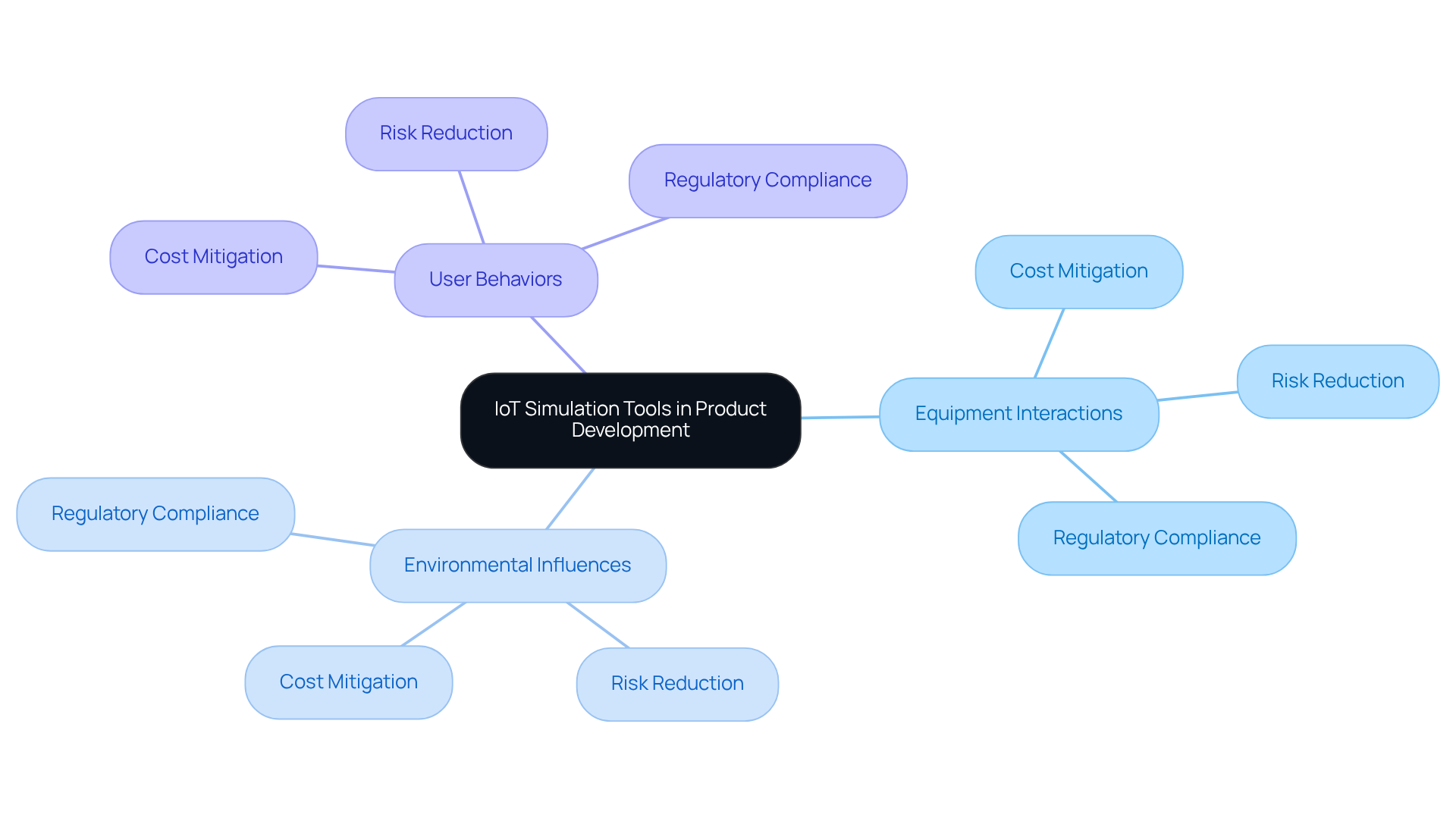
IoT simulation tools are essential in advancing medical equipment design by allowing engineers to create virtual representations of products before physical prototyping, enabling the assessment of equipment interactions, environmental influences, and user behaviors.
How do IoT modeling resources benefit the product development process?
They help identify potential design or functionality issues early in the development process, reducing costs and risks associated with real-world testing, and ultimately improving the quality and reliability of the final product.
In what ways do modeling resources assist with regulatory compliance?
Modeling resources provide detailed documentation of testing procedures and results, which is crucial for healthcare products that must meet strict safety requirements.
Can you provide an example of a real-world application of IoT simulation tools?
The Living Heart Project by Dassault Systèmes is an example where digital twins are used to personalize cardiac care by simulating patient responses to various treatments, optimizing care without risk.
What percentage of preclinical testing protocols do modeling technologies represent for Class II and III healthcare instruments?
Modeling technologies represent around 45% of preclinical testing protocols for Class II and III healthcare instruments in North America and Europe.
How do modeling technologies impact the effectiveness of healthcare products?
The integration of modeling technologies streamlines compliance with regulatory standards and enhances the effectiveness of healthcare products in a competitive market.
