Understanding Prototype Development: Definition and Importance for Medical Devices
Introduction Prototyping serves as a cornerstone in medical device development, where...

Prototype Definition Engineering serves as a foundational element in the medical device development process, where rigorous testing and validation are paramount to meeting user needs and regulatory standards. This discussion will explore the complexities inherent in this discipline, highlighting how innovative techniques like 3D modeling and rapid prototyping not only optimize design workflows while ensuring compliance with quality and safety standards but also contribute to improved product outcomes.
Despite the critical nature of prototype definition engineering, teams often encounter significant challenges that hinder their progress. What strategies can be employed to mitigate these challenges and effectively harness prototype definition engineering to enhance healthcare outcomes?
Prototype Definition Engineering serves as a foundational approach in developing healthcare equipment, ensuring that initial models undergo rigorous testing and validation. This discipline involves several key stages. These include:
In the medical device sector, this process is particularly vital, as it ensures prototypes not only comply with stringent regulatory standards but also effectively meet user requirements. Techniques such as 3D modeling and quick modeling enable engineers to swiftly explore various design iterations, greatly reducing project timelines. Statistics suggest that quick modeling can result in faster design iterations and early validation, which are essential for improving product quality and safety.
Successful examples, such as the SleeveSense project, demonstrate how effective prototype definition engineering can alleviate burdens on healthcare professionals while improving patient care. Ultimately, a structured design process not only enhances product quality but also fosters trust among healthcare professionals and patients alike.
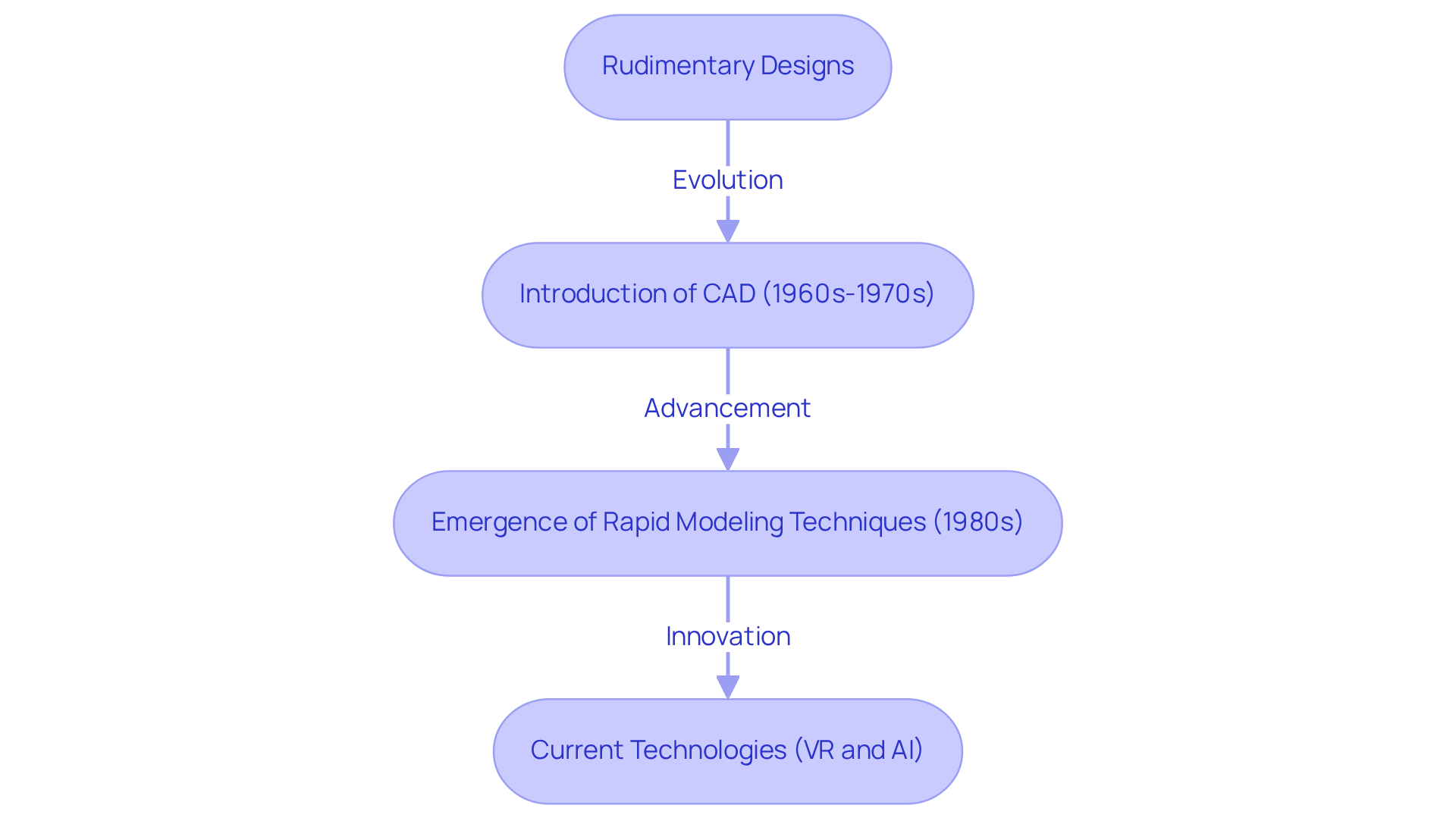
The evolution of model-making in healthcare reflects a significant shift from rudimentary designs to sophisticated technologies. The contemporary age of model development began in the 20th century with the introduction of computer-aided design (CAD) during the 1960s and 1970s. This innovation allowed engineers to create more complex and accurate prototypes, demonstrating the principles of prototype definition engineering and significantly reducing the time and expenses related to product creation.
Prior to CAD, engineers faced challenges in prototype definition engineering, which often led to increased time and costs. The emergence of rapid modeling techniques in the 1980s, particularly through the rise of 3D printing technology, transformed the landscape by facilitating quicker iterations and more accessible testing processes.
Currently, the initial phase of healthcare product creation employs advanced technologies such as virtual reality and artificial intelligence to enhance simulation capabilities and user interaction. These advancements are crucial for guaranteeing the safety and efficiency of healthcare instruments. As a result, the healthcare industry has seen a significant enhancement in the reliability of its instruments, reshaping the future of healthcare innovation.
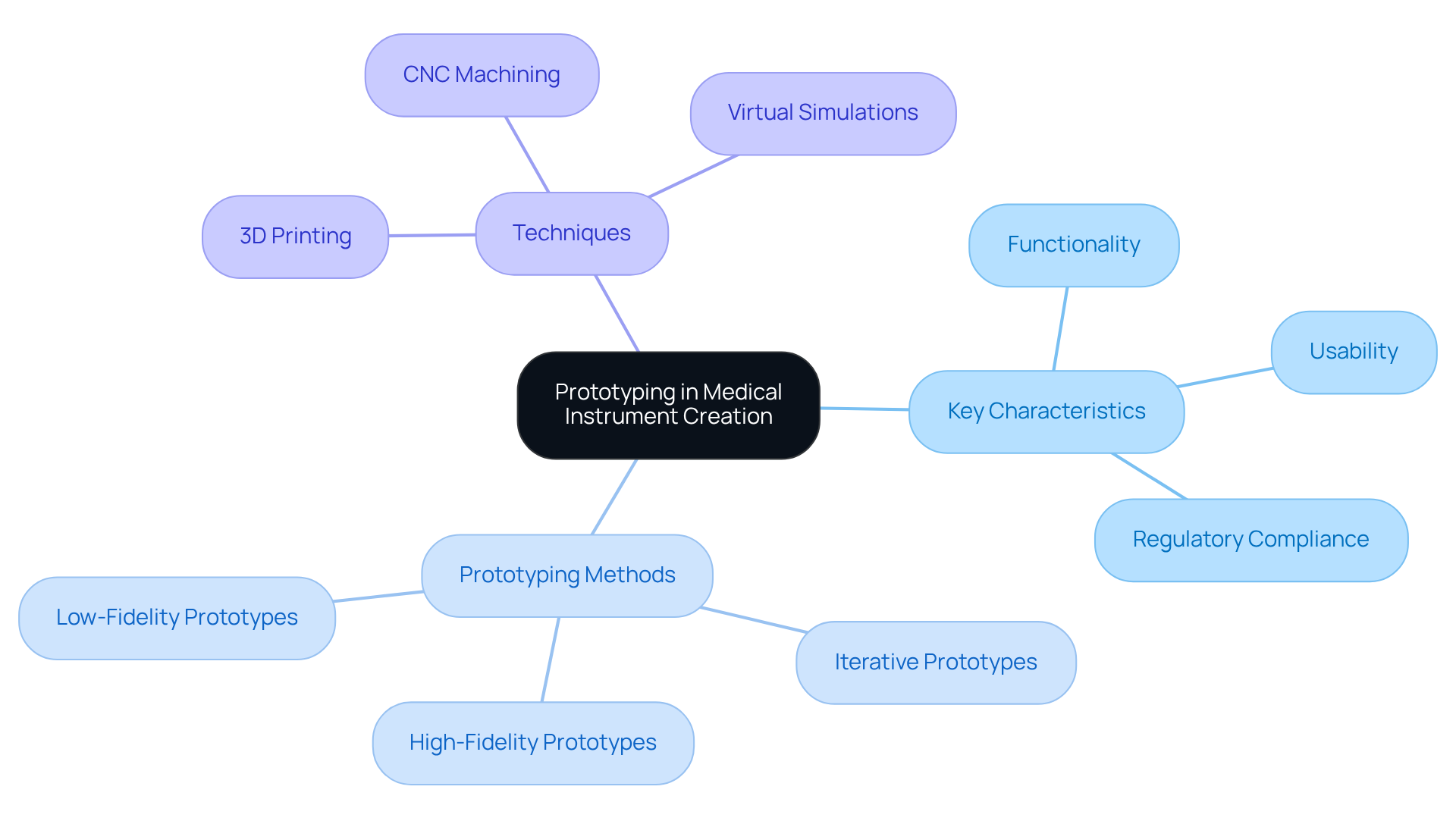
Successful modeling in medical instrument creation hinges on three essential traits: functionality, usability, and regulatory compliance. In prototype definition engineering, prototyping methods include:
Common techniques for creating prototypes include:
Each approach serves a specific aim, from exploring design options to testing usability and ensuring that the product meets safety requirements. Rapid modeling plays an essential role in speeding up progress and minimizing risk, allowing teams to enhance their designs based on real-world testing and feedback. Furthermore, incorporating mock-ups throughout the development lifecycle is crucial for delivering compliant and commercially successful products to market more quickly.
Voler Systems offers documentation compliance assistance, ensuring that all prototyping efforts conform to regulatory standards, which is essential as products become more intricate and regulated. Cleanroom environments are also crucial for ensuring compliance and quality in healthcare product manufacturing. As the complexity of medical devices increases, ensuring compliance and quality becomes essential through prototype definition engineering.
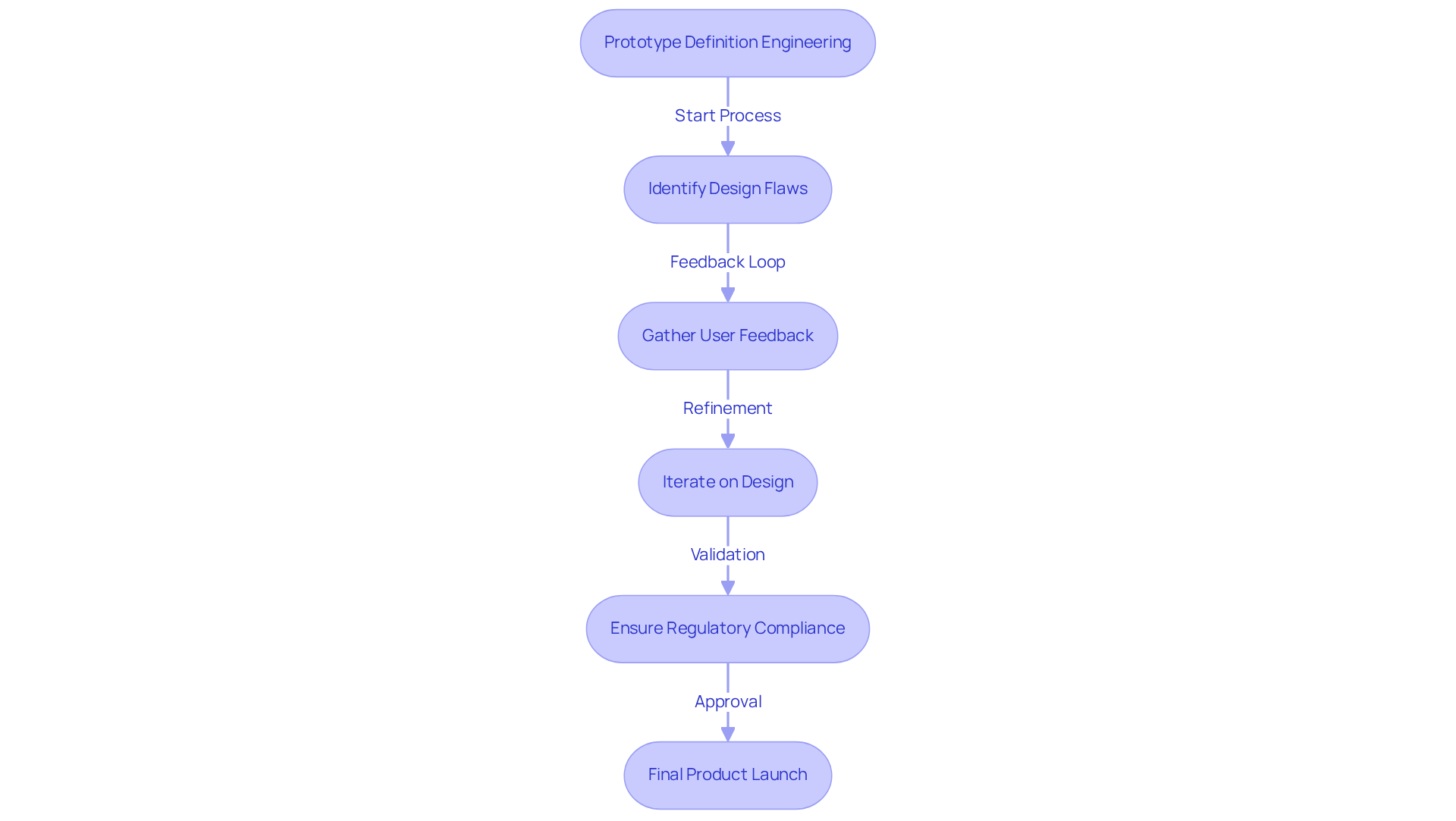
In the medical device sector, prototype definition engineering is essential for identifying design flaws early and facilitating user testing. By employing prototype definition engineering to create prototypes, teams can gather invaluable feedback from healthcare professionals and potential users, ensuring that the final product aligns with their needs and expectations. This iterative process enhances product quality and accelerates time-to-market by incorporating prototype definition engineering to refine development stages. Furthermore, model development plays a crucial role in regulatory compliance, as it provides documented evidence of testing and validation that is essential for obtaining necessary approvals from agencies like the FDA and CE.
However, common errors in manufacturing tests, such as inadequate planning, failure to simulate real-world conditions, and neglecting user feedback, can significantly hinder prototype definition engineering. It is essential to identify these pitfalls early on to ensure that prototype definition engineering is tested effectively, leading to better design outcomes. For instance, during the prototype definition engineering process of a new insulin pump, repeated modeling allowed engineers to enhance the product's usability and safety features, ultimately resulting in a successful market launch. Mark Hill notes that 'Prototype definition engineering is vital for medical devices, impacting patient outcomes and quality of care.' The investment in prototype definition engineering thus translates into reduced risks, lower costs, and improved patient outcomes. Statistics suggest that digital modeling can save up to 35% of total development expenses, while in silico simulations accelerate regulatory reviews by reducing the necessity for extensive physical trials. Ultimately, the commitment to rigorous prototype definition engineering processes can significantly enhance patient care and safety outcomes.
Prototype Definition Engineering is not merely a step in medical device development; it is a foundational element that ensures safety and efficacy. This structured approach ensures compliance with regulatory standards and aligns the final product with user needs. Healthcare engineers can enhance safety and usability by effectively iterating on designs through advanced modeling techniques.
Throughout the article, the significance of Prototype Definition Engineering has been highlighted through its key characteristics, methods, and historical evolution. From the advent of computer-aided design to modern rapid modeling techniques, each advancement has contributed to a more efficient and effective prototyping process. However, without a structured approach, gathering user feedback can be inconsistent, leading to potential risks. The ability to gather user feedback and refine designs iteratively has proven essential in minimizing risks and enhancing product quality, ultimately leading to better patient outcomes.
A rigorous prototyping process is essential for stakeholders in the healthcare industry to consider. Investing in Prototype Definition Engineering not only streamlines product development but also fosters trust among healthcare professionals and patients. Neglecting this process risks not only product quality but also patient safety. As the landscape of medical technology evolves, the commitment to rigorous prototyping will determine the success of future innovations.
What is Prototype Definition Engineering?
Prototype Definition Engineering is a foundational approach in developing healthcare equipment, focusing on rigorous testing and validation of initial models.
What are the key stages involved in Prototype Definition Engineering?
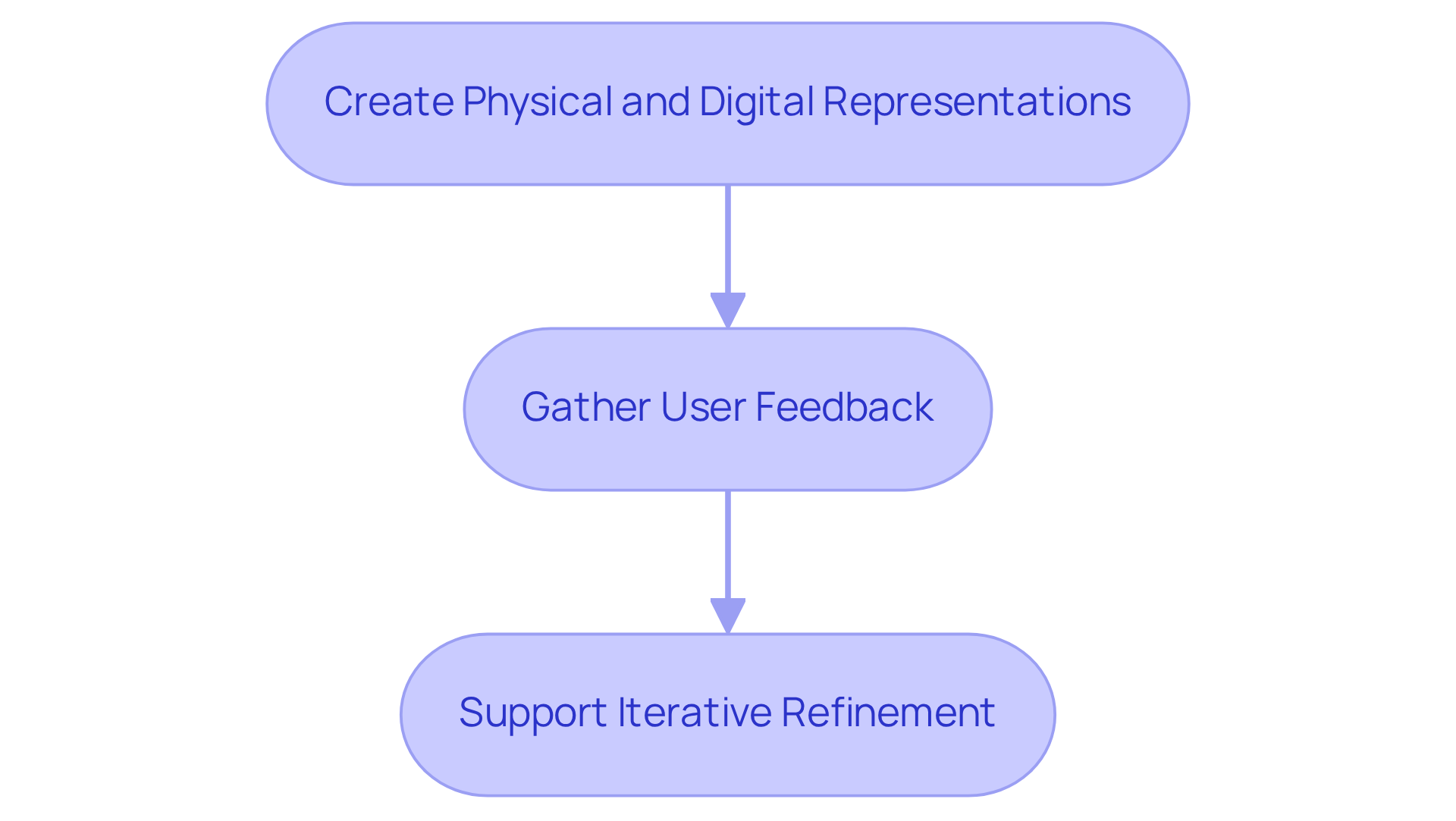
The key stages include creating physical and digital representations for functional testing, gathering user feedback, and supporting iterative refinement.
Why is Prototype Definition Engineering important in the medical device sector?
It ensures that prototypes comply with stringent regulatory standards and effectively meet user requirements, which is crucial for the safety and quality of medical devices.
What techniques are commonly used in Prototype Definition Engineering?
Techniques such as 3D modeling and quick modeling are commonly used, allowing engineers to explore various design iterations quickly.
How does quick modeling impact the design process?
Quick modeling can lead to faster design iterations and early validation, which are essential for improving product quality and safety.
Can you provide an example of successful Prototype Definition Engineering?
The SleeveSense project is an example that demonstrates how effective prototype definition engineering can alleviate burdens on healthcare professionals while enhancing patient care.
What are the overall benefits of a structured design process in Prototype Definition Engineering?
A structured design process enhances product quality and fosters trust among healthcare professionals and patients.
