Introduction
The complex landscape of embedded systems in medical devices introduces a distinct array of challenges that can profoundly affect patient safety and device reliability. Developers, as they navigate the intricacies of debugging these systems, can derive critical insights into effective troubleshooting strategies. These strategies not only enhance device performance but also ensure adherence to regulatory standards. However, engineers face significant obstacles, including:
- Limited visibility
- Real-time constraints
- The essential requirement for comprehensive documentation
How can they surmount these hurdles to achieve optimal debugging outcomes?
Understand the Unique Challenges of Embedded System Debugging
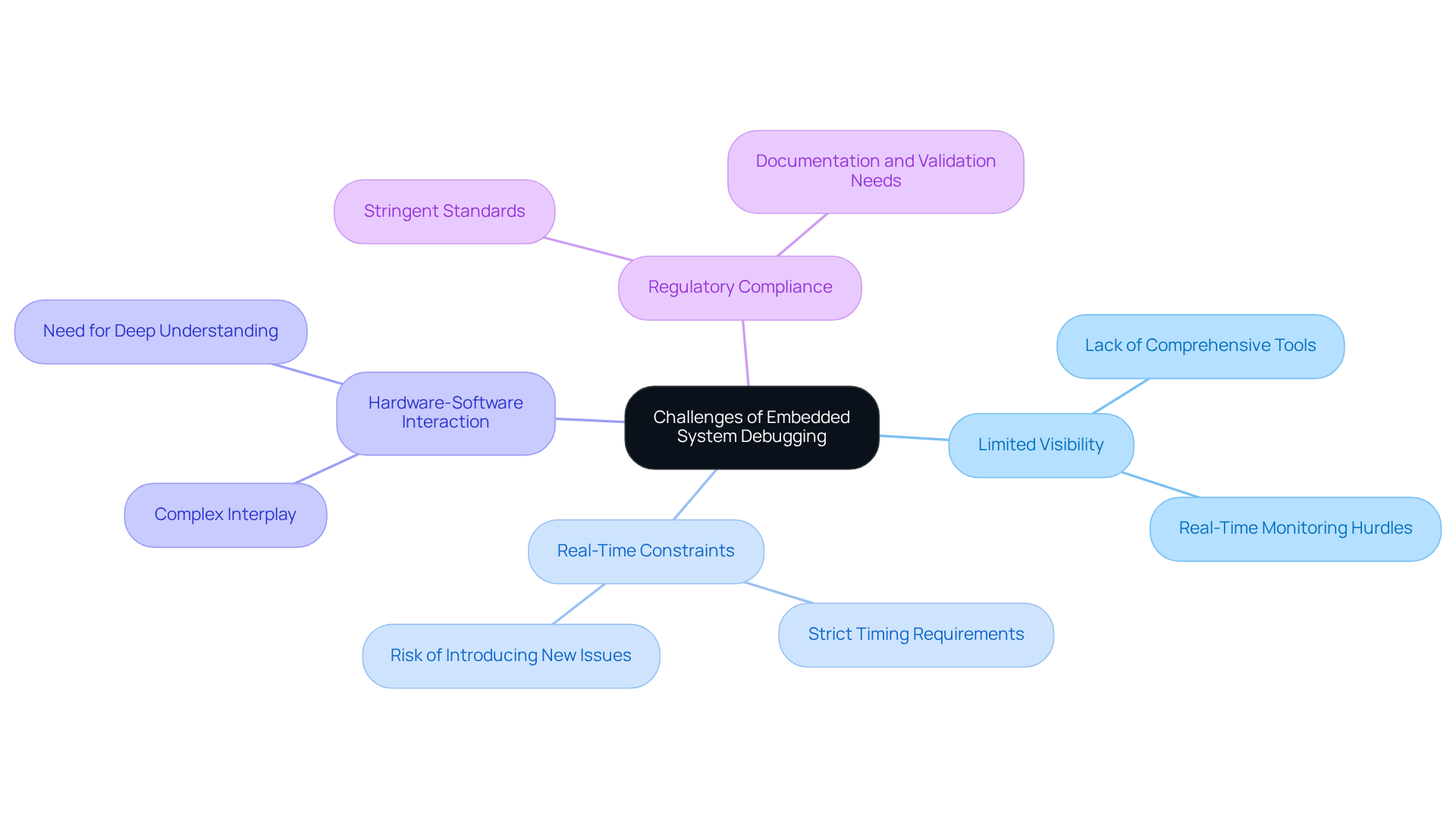
The use of a debugger in embedded systems for medical equipment presents unique challenges due to their inherent complexity and the critical nature of their applications. The primary challenges include:
- Limited Visibility: Unlike traditional software environments, embedded systems often lack comprehensive user interfaces and debugging tools. This absence makes real-time monitoring of system behavior a significant hurdle, potentially leading to undetected issues that compromise functionality.
- Real-Time Constraints: Many medical devices operate under strict timing requirements, where even minor delays can lead to critical failures. This urgency complicates the with the debugger in embedded systems, as developers must ensure that fixes do not introduce new timing issues.
- Hardware-Software Interaction: The intricate interplay between hardware and software can create problems that are difficult to isolate. A deep understanding of both domains is essential for effective troubleshooting using a debugger in embedded systems, as issues may arise from either side of the system.
- Regulatory Compliance: Debugging processes must comply with stringent regulatory standards, requiring thorough documentation and validation. Adhering to these standards is vital for ensuring the safety and reliability of healthcare instruments, as failures can result in significant legal and financial repercussions.
Addressing these challenges is crucial for developing effective troubleshooting strategies that enhance the reliability and safety of healthcare equipment, ultimately leading to improved patient outcomes.
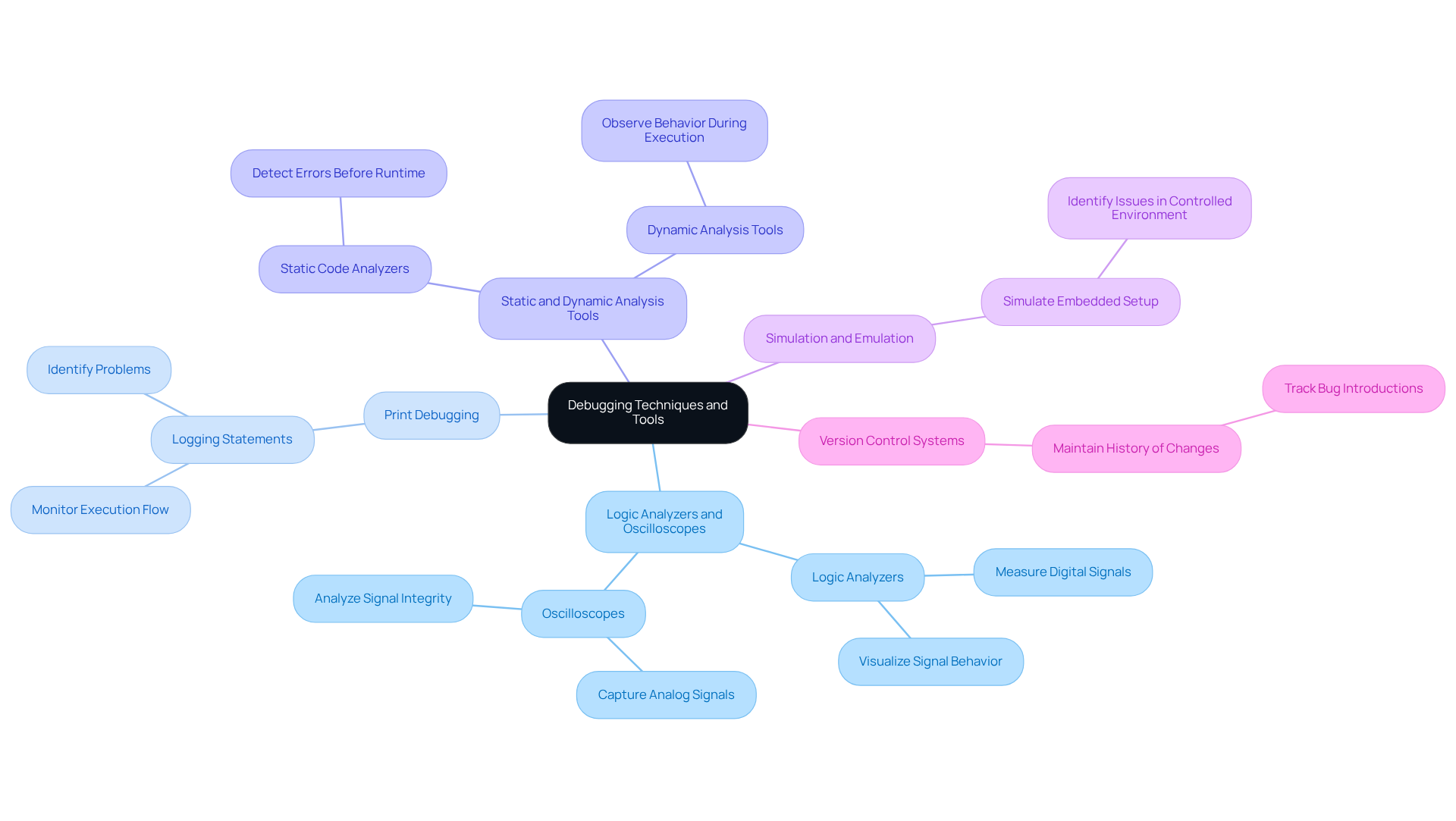
To enhance the debugging process in embedded systems for medical devices, several techniques and tools can be employed:
- Use of Logic Analyzers and Oscilloscopes: These essential tools visualize signal behavior and timing issues, providing insights that software debugging alone may overlook. Logic analyzers measure digital signals, representing them as binary values, while oscilloscopes capture and display analog signals as waveforms. This makes them ideal for analyzing signal integrity and timing.
- Print Debugging: Incorporating logging statements in the code assists in monitoring the flow of execution and identifying where problems occur. This practice leads to enhanced reliability in embedded devices.
- Static and Dynamic Analysis Tools: Employing static code analyzers can detect potential errors before runtime, while dynamic analysis tools observe behavior during execution. This dual approach ensures that issues are recognized early in the development process.
- Simulation and Emulation: Prior to deploying on real hardware, simulating the embedded setup allows for the identification of issues in a controlled environment. This reduces the risk of failures after deployment.
- Version Control Systems: Maintaining a history of changes facilitates tracking down when a bug was introduced, making it easier to isolate and resolve issues.
By utilizing these methods and tools, engineers can significantly decrease troubleshooting time and improve the reliability of their medical devices with the help of a debugger in embedded system.
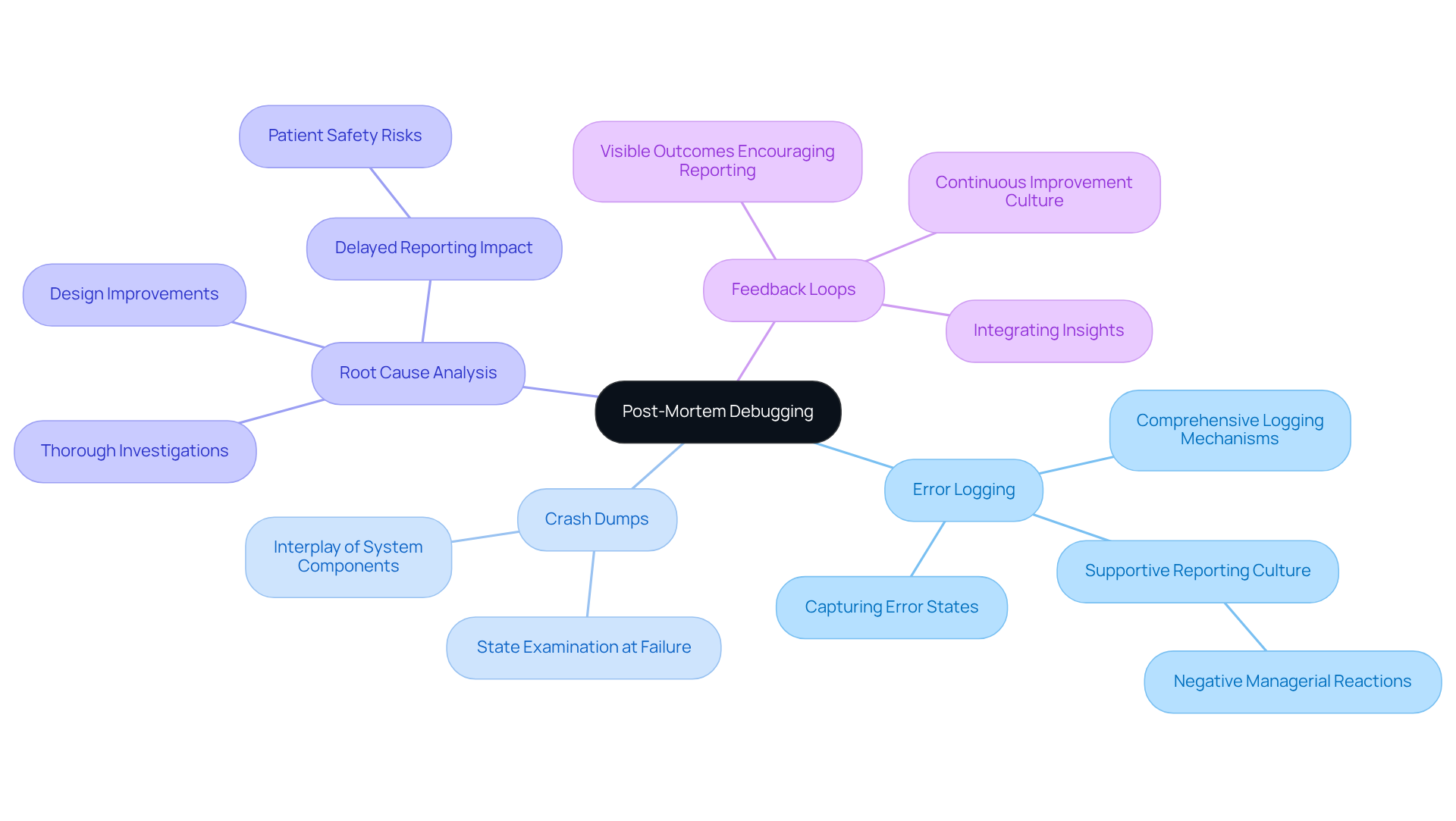
Leverage Post-Mortem Debugging for Enhanced Insights
Post-mortem debugging is essential for examining frameworks after failures, as it provides crucial insights into root causes. Key practices include:
- Error Logging: Implementing comprehensive logging mechanisms is vital for capturing error states and system behavior leading up to a failure. This data is invaluable for diagnosing issues post-failure, offering a detailed account of performance prior to the incident. A risk management officer has noted that negative managerial reactions can deter staff from reporting incidents, underscoring the importance of fostering a supportive reporting culture.
- Crash Dumps: Utilizing crash dump analysis enables engineers to examine the state of the platform at the time of failure, aiding in the identification of the exact conditions that led to the issue. This practice is critical for of various system components during a malfunction.
- Root Cause Analysis: Conducting thorough investigations into failures is necessary to determine underlying causes. This analysis informs design improvements and helps prevent future occurrences, ensuring that similar issues do not arise again. For instance, a study highlighted that delayed adverse event reporting by manufacturers of health equipment can jeopardize patient safety, emphasizing the necessity for prompt and precise error recording.
- Feedback Loops: Establishing processes for integrating insights gained from post-mortem analyses into future design and development cycles fosters a culture of continuous improvement. By learning from past failures, teams can enhance product reliability and safety. A nurse observed that when staff see actions taken from reports, they are more inclined to report, reinforcing the value of feedback in the reporting process.
Effectively utilizing post-mortem analysis through these practices allows engineers to enhance equipment performance, ultimately resulting in improved patient outcomes.
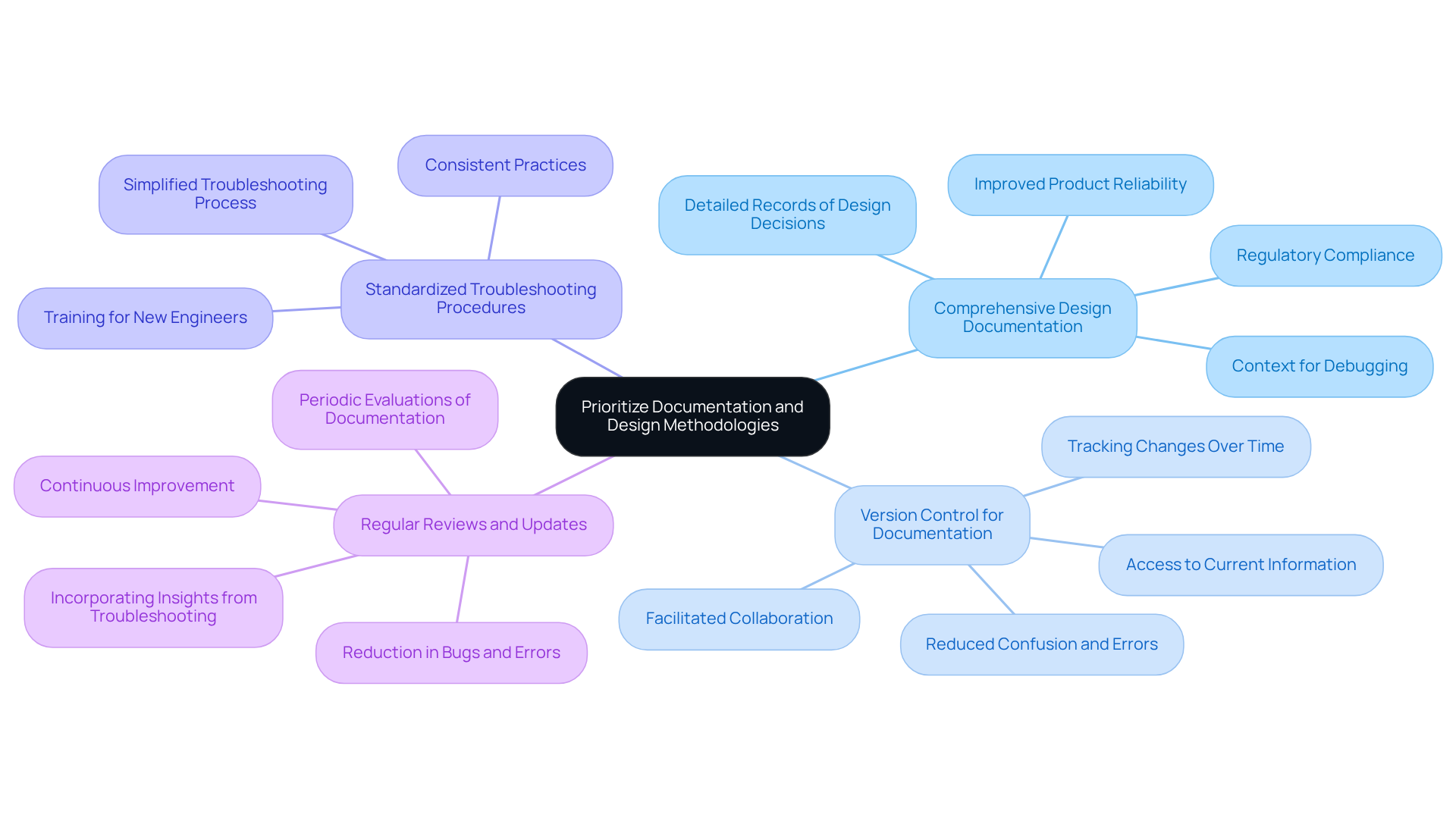
Prioritize Documentation and Design Methodologies
Efficient documentation and organized design approaches are essential for successful troubleshooting in embedded technologies, particularly within the medical device industry. Key practices include:
- Comprehensive Design Documentation: It is vital to maintain detailed records of design decisions, system architecture, and interfaces. This thorough documentation provides context during the use of a debugger in embedded systems, aiding in issue identification and enhancing understanding of system behavior. Ultimately, this contributes to and ensures compliance with regulatory standards.
- Version Control for Documentation: Just as code changes are tracked, documentation must also be versioned to reflect updates over time. Implementing version control ensures that all team members have access to the most current information, thereby reducing confusion and errors during the development process. For example, utilizing tools like Git allows teams to manage changes effectively, facilitating collaboration and maintaining a clear history of modifications.
- Standardized Troubleshooting Procedures: Establishing and recording standardized methods for troubleshooting is crucial. This ensures that all team members adhere to consistent and efficient practices. Such methods not only simplify the troubleshooting process but also assist in training new engineers, enabling them to quickly adapt to established protocols.
- Regular Reviews and Updates: It is important to schedule periodic evaluations of documentation and methodologies to ensure their relevance and effectiveness. Incorporating insights gained from previous troubleshooting efforts fosters continuous improvement in documentation practices, which can lead to a significant reduction in bugs and errors. Studies indicate that proper documentation can lower maintenance costs by up to 50% and reduce bugs by 40% (Microsoft).
By prioritizing documentation and structured methodologies, device engineers can establish a robust framework that supports effective use of a debugger in embedded systems and compliance with regulatory standards. This approach ultimately contributes to the successful development of innovative health technologies, aligning with the broader context of engineering design projects as outlined in the table of contents, and exemplified by the case study of documentation compliance support for startups in the medical technology sector.
Conclusion
Mastering the art of debugging in embedded systems for medical devices is crucial for ensuring the safety and effectiveness of healthcare technologies. The complexities inherent in these systems - such as limited visibility, real-time constraints, hardware-software interactions, and stringent regulatory compliance - present unique challenges. Effectively addressing these issues through robust debugging strategies is essential for enhancing the reliability of medical devices and ultimately improving patient outcomes.
Key techniques and tools have been highlighted throughout this discussion, including:
- Logic analyzers
- Print debugging
- Simulation
- Post-mortem analysis
Each of these methods contributes to a comprehensive understanding of system behavior and aids in identifying issues before they escalate. Moreover, prioritizing documentation and standardized procedures cultivates a culture of continuous improvement and compliance, which is vital in the highly regulated medical device industry.
In conclusion, adopting best practices for debugging not only mitigates risks associated with embedded systems but also reinforces the commitment to patient safety and quality care. By implementing these strategies, engineers can significantly enhance the development process of medical devices, ensuring they meet the highest standards of reliability and performance. The journey toward mastering debugging in embedded systems is ongoing, and the insights gained from both successes and failures will pave the way for future innovations in healthcare technology.
Frequently Asked Questions
What are the unique challenges of debugging embedded systems in medical equipment?
The unique challenges include limited visibility, real-time constraints, hardware-software interaction, and regulatory compliance.
Why is limited visibility a challenge in embedded system debugging?
Limited visibility occurs because embedded systems often lack comprehensive user interfaces and debugging tools, making real-time monitoring of system behavior difficult and potentially leading to undetected issues.
How do real-time constraints affect embedded system debugging?
Real-time constraints are critical because many medical devices have strict timing requirements, where even minor delays can result in critical failures. This urgency complicates the debugging process, as developers must ensure that fixes do not introduce new timing issues.
What role does hardware-software interaction play in debugging embedded systems?
The intricate interplay between hardware and software can create problems that are challenging to isolate. Effective troubleshooting requires a deep understanding of both domains, as issues may arise from either side of the system.
What is the importance of regulatory compliance in embedded system debugging?
Debugging processes must comply with stringent regulatory standards, which require thorough documentation and validation. Adhering to these standards is essential for ensuring the safety and reliability of healthcare instruments, as failures can lead to significant legal and financial repercussions.
Why is it crucial to address the challenges of embedded system debugging in medical devices?
Addressing these challenges is vital for developing effective troubleshooting strategies that enhance the reliability and safety of healthcare equipment, ultimately leading to improved patient outcomes.
List of Sources
- Understand the Unique Challenges of Embedded System Debugging
- Medical technology is unthinkable without embedded systems | embedded world (https://embedded-world.de/en/knowledge/2026/03/medical-technology-is-unthinkable-without-embedded-systems)
- Embedded Software Testing for Medical Devices - Parasoft (https://parasoft.com/blog/medical-devices-embedded-software-testing)
- AI-Assisted Debugging 2026: Anomaly Detection in Firmware (https://promwad.com/news/ai-assisted-debugging-2026-anomaly-detection-firmware)
- From AI to CRA: The trends shaping the future of embedded development at embedded world 2026 (https://iar.com/blog/the-trends-shaping-the-future-of-embedded-development-at-embedded-world-2026)
- Top 10 Challenges in the MedTech industry (https://ptc.com/en/blogs/medtech/medical-device-industry-challenges?srsltid=AfmBOoq-zW7U--CUV3rghEmH5tJ5ReZSLruKGOUJA5wfd74krv2598xZ)
- Implement Effective Debugging Techniques and Tools
- Best Practices for Selecting Embedded Tools in Medical Device Design (https://volersystems.com/blog/best-practices-for-selecting-embedded-tools-in-medical-device-design)
- Logic Analyzer vs Oscilloscope - Keysight Technologies (https://keysight.com/used/us/en/knowledge/guides/logic-analyzer-vs-oscilloscope)
- Embedded Software Testing for Medical Devices - Parasoft (https://parasoft.com/blog/medical-devices-embedded-software-testing)
- Oscilloscope vs. Logic Analyzer vs. Protocol Analyzer - Understanding Their Roles in Debugging (https://totalphase.com/blog/2025/02/oscilloscope-vs-logic-analyzer-vs-protocol-analyzer-understanding-roles-debugging?srsltid=AfmBOoqcofP1eMa7r-oIFwSG7foKlsyGK_aTJ0uLjdDZnks1HKo_qPIe)
- Top Tools for Embedded System Debugging and Monitoring in 2025 (https://promwad.com/news/embedded-debugging-tools-2025)
- Leverage Post-Mortem Debugging for Enhanced Insights
- 2026 US Medical Device & Diagnostic Post-Market Surveillance, Vigilance Conference (https://lne-gmed.com/news/2026-us-ttl-pms-conference)
- Medical device manufacturers often delay reporting adverse safety events, study says (https://healthjournalism.org/blog/2025/05/medical-device-manufacturers-often-delay-reporting-adverse-safety-events-study-says)
- Challenges and dynamics in reporting medical device incidents: a qualitative study - PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC12727643)
- Prioritize Documentation and Design Methodologies
- A Method for Debugging Medical Device Software (https://innolitics.com/articles/skuash-debugging)
- Statistics and Facts Supporting the Importance of Software Documentation - Away with words (https://awaywithwords.co/2024/09/18/statistics-and-facts-supporting-the-importance-of-software-documentation-away-with-words)
- Why Documentation Debt Will Be the New Technical Debt in 2026 (https://techscribehub.medium.com/why-documentation-debt-will-be-the-new-technical-debt-in-2026-232676ddacc8)
- Prepare your medical device software for the new FDA cybersecurity guidance (https://parasoft.com/blog/prepare-your-medical-device-software-for-the-new-fda-cybersecurity-guidance)
- FDA Medical Device Inspections: What Changed and Why It Matters (https://nsf.org/life-science-regulatory-news/fda-medical-device-inspections-what-changed-and-why-it-matters)





