Introduction
Creating embedded systems for medical devices is a complex endeavor that requires precision and strict adherence to established standards. Engineers and designers encounter the challenge of integrating essential components while ensuring compliance with regulatory requirements and safety protocols. This article examines best practices for developing robust embedded systems in healthcare, highlighting critical components, advanced technologies, and rigorous testing methods that can enhance patient care and operational efficiency.
How can engineers effectively navigate these multifaceted challenges to devise innovative solutions that not only address current needs but also anticipate future healthcare demands?
Identify Key Components of Embedded Systems in Medical Devices
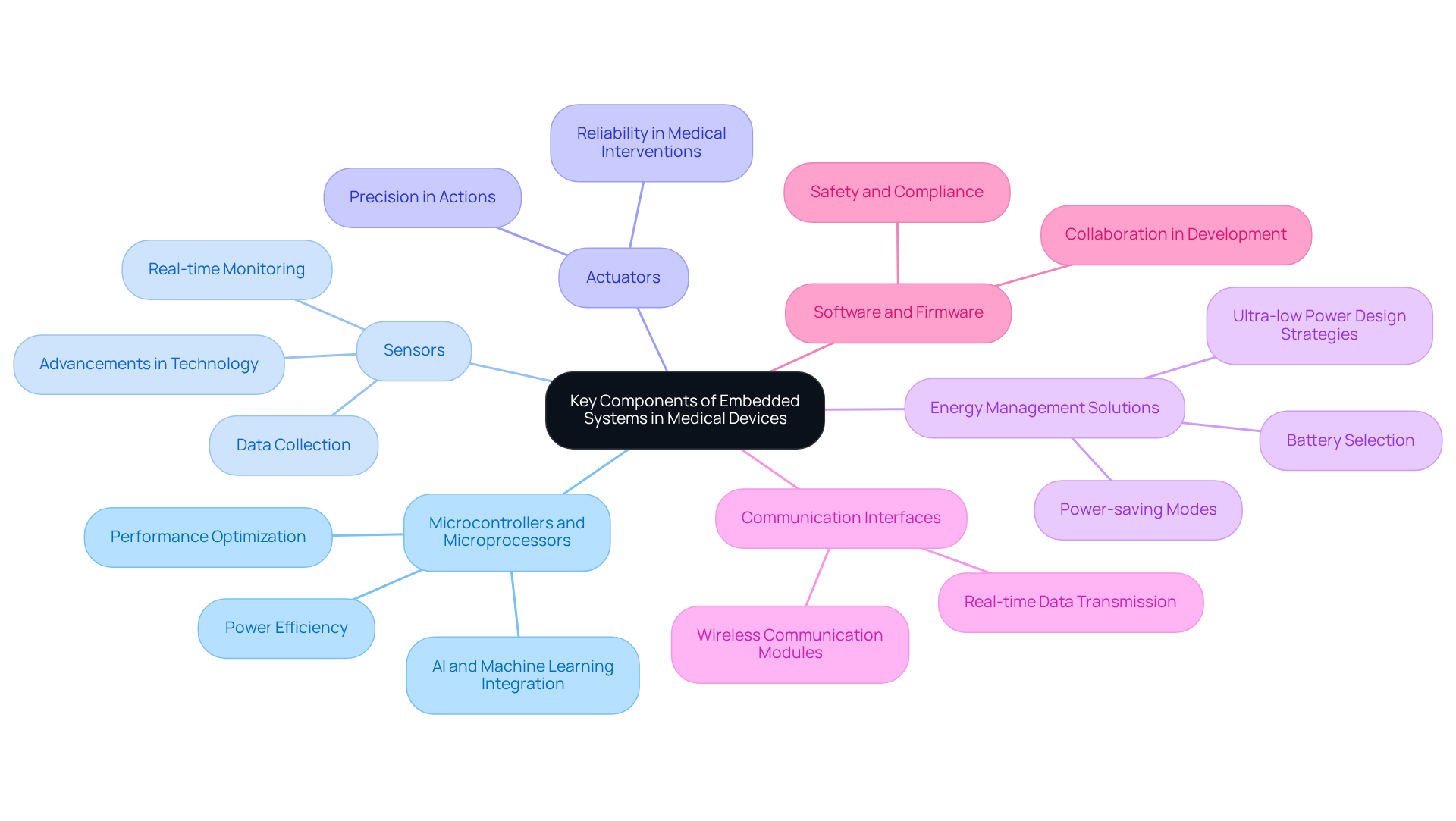
Creating efficient embedded systems basic for medical equipment requires the careful identification and integration of essential components that ensure functionality, reliability, and compliance with industry standards. Since 1979, Voler Systems has committed to excellence in electronics product design, with a focus on wearable and IoT technology. The primary components include:
- Microcontrollers and Microprocessors: These components serve as the brains of the embedded system, executing instructions and managing information. Selecting the right microcontroller is vital for optimizing performance and power efficiency, particularly in battery-operated devices. The incorporation of AI and machine learning can further enhance these components' capabilities, leading to improved diagnostic accuracy and better patient care outcomes.
- Sensors: Critical for collecting data from the environment or the human body, sensors in medical devices include temperature sensors, heart rate monitors, and glucose sensors. Recent advancements in sensor technology are improving the accuracy and responsiveness of these devices, facilitating real-time monitoring and enhancing patient outcomes. Voler's expertise in sensor integration ensures that these instruments meet the highest performance standards.
- Actuators: These components perform actions based on processed data, such as administering medication or adjusting equipment settings. The integration of advanced actuators can enhance the precision and reliability of medical interventions, a key focus for Voler in their design processes.
- Energy Management Solutions: Effective power management is crucial, especially for portable devices. This involves selecting appropriate batteries and implementing power-saving modes to extend device longevity, which is increasingly important as the demand for continuous monitoring grows. Voler Systems employs ultra-low power design strategies to optimize battery life in their medical equipment.
- Communication Interfaces: These interfaces enable devices to communicate with other systems, utilizing wireless communication modules like Bluetooth and Wi-Fi for data transmission. Enhanced connectivity is expected to improve patient care and facilitate real-time information sharing among healthcare providers. As healthcare devices increasingly need to interact and share data in real time, Voler Systems addresses this challenge through their comprehensive IoT design consulting.
- Software and Firmware: Software governs hardware components and processes data, necessitating design considerations for safety and compliance. Adhering to industry standards, such as ISO 14971 for risk management, is essential to ensure the safety and efficacy of healthcare devices. Voler Systems prioritizes collaboration in software development to effectively navigate compliance from prototype to production.
By thoughtfully selecting and integrating these elements, engineers can create robust systems that meet the stringent requirements of healthcare products, ultimately leading to improved patient care and operational efficiency in embedded systems basic design.
Address Regulatory Compliance and Safety Standards
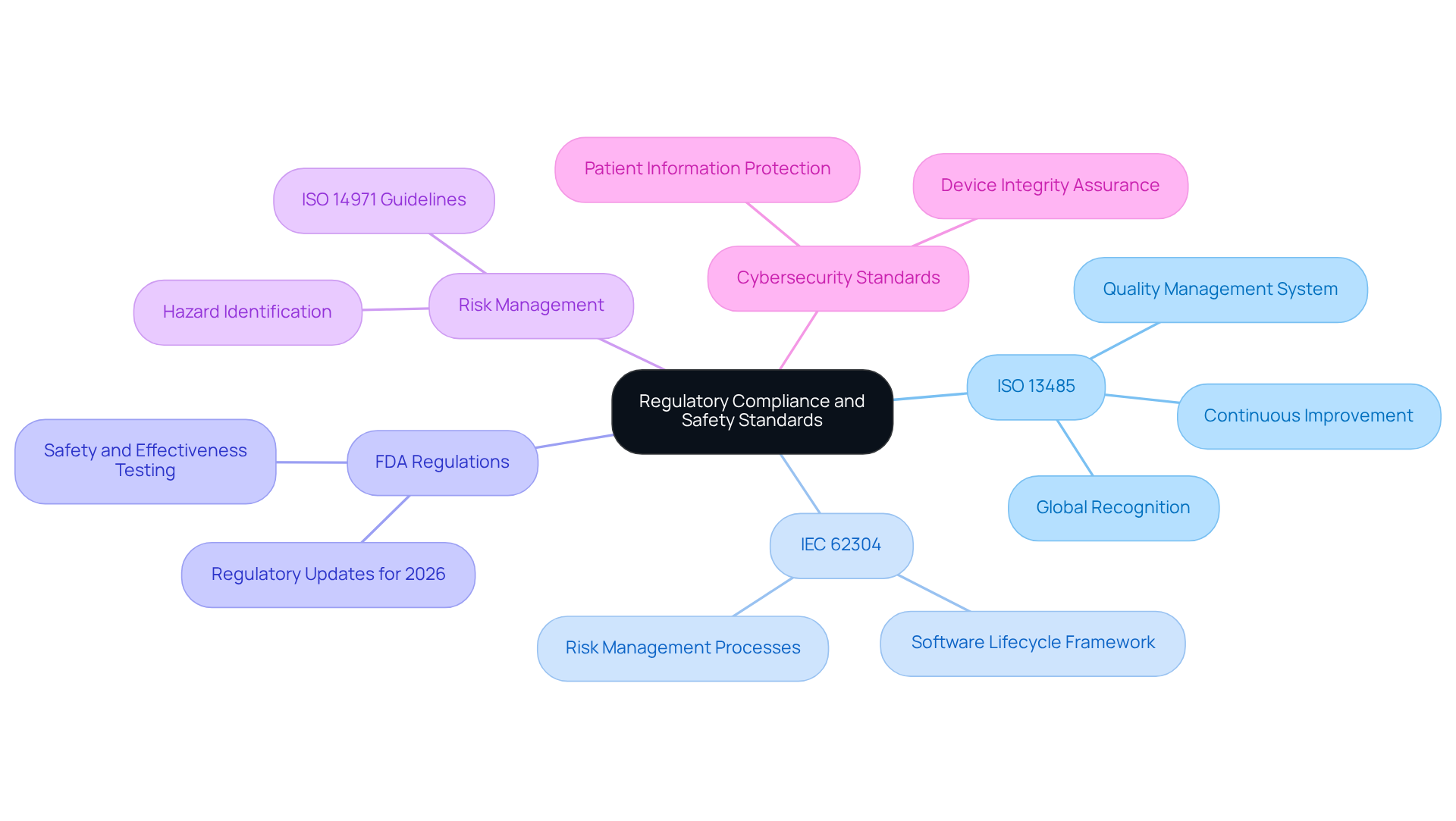
Adhering to regulatory standards is essential in the development of healthcare instruments. The key regulations include:
- ISO 13485: This standard specifies the criteria for a quality management system tailored for healthcare products, ensuring that items consistently meet customer and regulatory expectations.
- IEC 62304: This standard establishes a framework for the life cycle of healthcare technology software, encompassing development, maintenance, and risk management processes.
- FDA Regulations: In the United States, the FDA mandates that manufacturers of health-related instruments demonstrate safety and effectiveness through comprehensive testing and documentation, in line with the latest regulatory updates for 2026.
- Risk Management: Implementing a risk management process as outlined in ISO 14971 is vital for identifying and mitigating potential hazards associated with healthcare products, thereby enhancing overall safety.
- Cybersecurity Standards: Given the increasing connectivity of healthcare devices, adherence to cybersecurity guidelines is crucial for safeguarding patient information and ensuring the integrity of the equipment.
Integrating these compliance measures into the design and development process enables engineers to enhance the safety and reliability of healthcare products, ultimately protecting patients and facilitating successful market access.
Incorporate Advanced Technologies for Enhanced Functionality
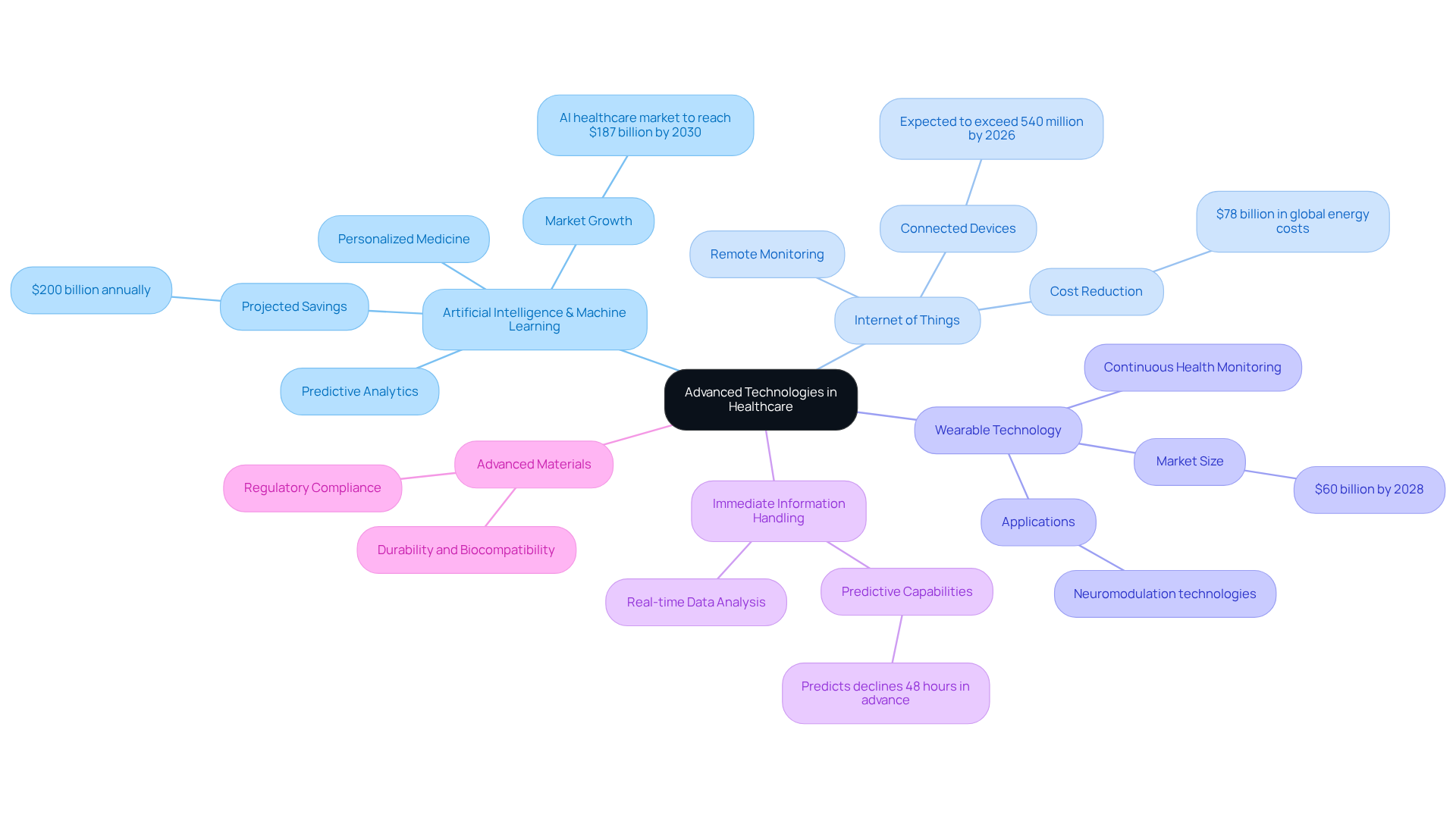
To remain competitive in the healthcare equipment market, it is essential to incorporate advanced technologies into embedded systems basic. Key technologies to consider include:
- Artificial Intelligence (AI) and Machine Learning (ML): These technologies enhance the functionality of tools through predictive analytics, personalized medicine, and improved diagnostics. AI-driven applications, such as those developed by Voler Systems, are projected to save the healthcare sector an estimated $200 billion annually by optimizing various processes, including Edge AI applications that improve real-time decision-making in medical tools.
- Internet of Things (IoT): IoT connectivity enables devices to interact with one another and healthcare providers, facilitating remote monitoring and information gathering. By 2026, the number of connected healthcare devices is expected to exceed 540 million units, significantly enhancing patient monitoring capabilities. Voler Technologies' innovative IoT solutions, including cost-effective low data rate links utilizing TI CC3200, improve connectivity and operational efficiency. Furthermore, IoT-enabled optimization is anticipated to reduce global energy costs by $78 billion, underscoring its advantages in healthcare.
- Wearable Technology: The integration of wearable sensors allows for continuous health monitoring, which enhances patient engagement and outcomes. Voler Systems plays a pivotal role in advancing wearable neuromodulation technologies, particularly in FDA-approved medical products for neurological pain management. The market for AI-powered wearable health products is projected to reach $60 billion by 2028, emphasizing their growing importance in preventive healthcare, including solutions for sleep apnea.
- Immediate Information Handling: Establishing immediate information handling capabilities ensures that instruments can respond swiftly to patient needs, thereby improving safety and efficiency. AI-supported tools can analyze patient data instantaneously, predicting declines 48 hours before human intervention is feasible, marking a significant advancement in healthcare technology.
- Advanced Materials: The use of innovative materials can enhance equipment durability, biocompatibility, and overall performance, which is crucial for meeting stringent regulatory standards in the healthcare sector.
By adopting these advanced technologies, engineers at Voler Systems can develop innovative healthcare tools that not only meet current market demands but also anticipate future health requirements.
Implement Rigorous Testing and Validation Protocols
Testing and validation are essential components in the development of embedded systems. These practices ensure that devices function reliably and meet regulatory standards.
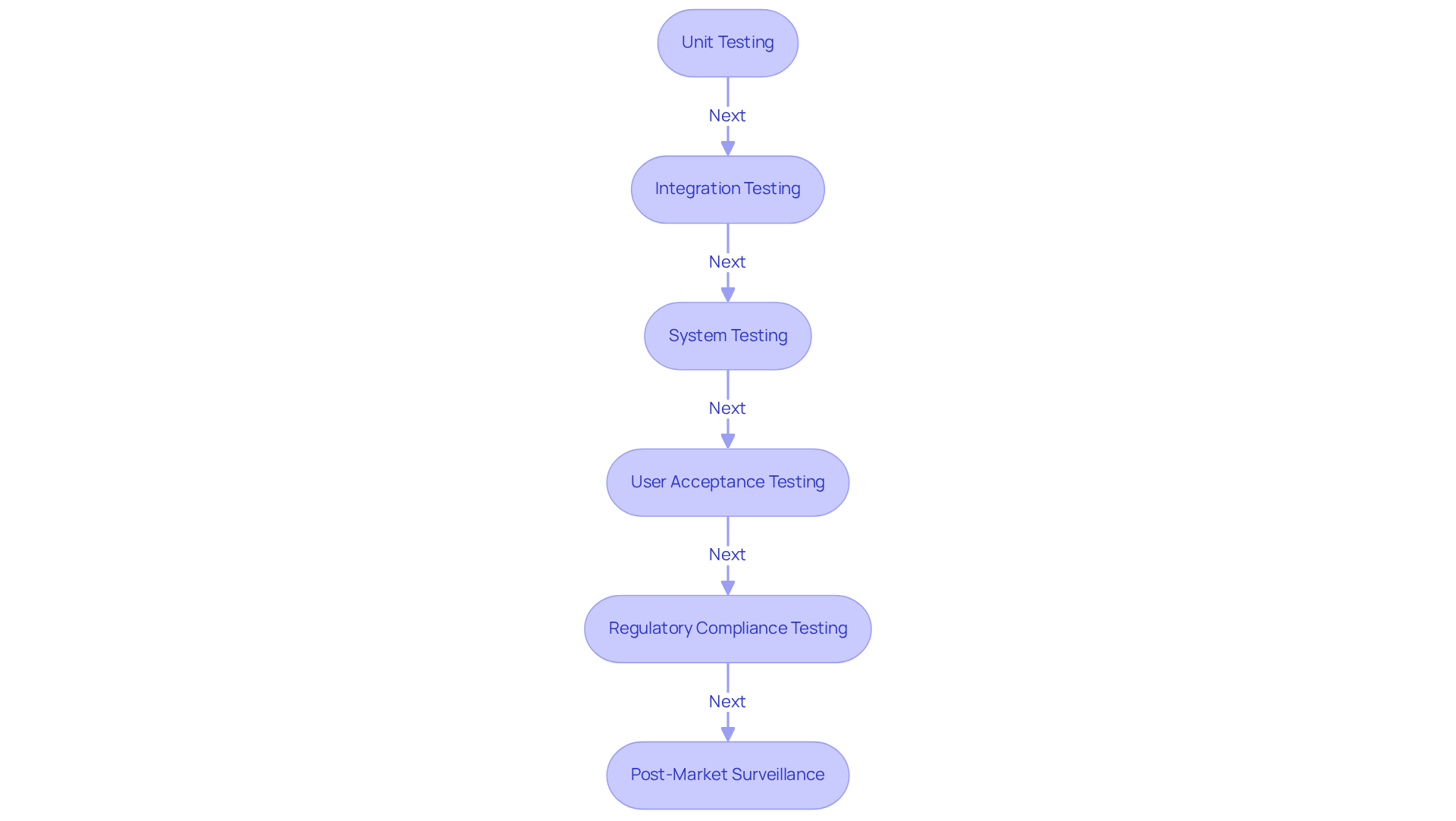
Unit Testing involves assessing individual components to confirm their correct operation in isolation. This early identification of issues is critical; for example, unit tests can validate the accuracy of functions like medication dosage calculations, ensuring dependable performance.
Integration Testing is crucial for verifying that different components work together as intended. This step is vital for the smooth operation of the entire system. For instance, successful integration testing in patient record management systems ensures that data flows correctly between modules, thereby enhancing overall functionality.
System Testing encompasses comprehensive evaluation of the complete system to ensure it meets all specifications and requirements. In the case of a dialysis system, this testing includes verifying the user interface, measurement accuracy, and alarm management, confirming that the apparatus operates reliably under real-world conditions.
User Acceptance Testing (UAT) involves end-users in the testing process, allowing for the collection of feedback on usability and functionality. This step is particularly important in telemedicine software, especially in the context of embedded systems basic, where healthcare professionals assess the software's ergonomics and functionalities during virtual consultations.
Regulatory Compliance Testing ensures that the equipment adheres to all regulatory requirements through thorough documentation and validation processes. Expertise from organizations like Voler Systems is invaluable in navigating these regulatory challenges, underscoring the importance of compliance in the healthcare equipment sector. The FDA's emphasis on a risk-based strategy for testing highlights the critical nature of adherence in the medical equipment industry. Additionally, common errors in manufacturing tests, such as inadequate documentation or failure to follow established protocols, can result in significant setbacks during development.
Post-Market Surveillance is vital after product launch, as ongoing monitoring of performance in the field helps identify potential issues and ensures continued compliance with regulatory standards.
By implementing these rigorous testing and validation protocols, engineers can significantly mitigate the risk of device failures and enhance patient safety. This aligns with insights from industry leaders who emphasize that quality is a continuous process. Furthermore, addressing common errors in manufacturing tests can streamline the development process, ensuring that new hardware meets the highest standards of quality and efficiency.
Conclusion
Creating effective embedded systems for medical devices necessitates a thorough understanding of various components, strict compliance with regulatory standards, and the integration of advanced technologies. By concentrating on microcontrollers, sensors, actuators, energy management, communication interfaces, and robust software, engineers can design systems that not only fulfill industry demands but also enhance patient care outcomes. The importance of adhering to safety standards and rigorous testing protocols is paramount, as these practices ensure that devices are reliable and effective in real-world applications.
This article underscores key best practices, including the meticulous selection of components, adherence to regulatory frameworks such as ISO 13485 and IEC 62304, and the incorporation of cutting-edge technologies like AI, IoT, and wearable devices. Each of these elements is crucial in developing medical devices that are not only functional but also safe and compliant with the latest industry standards. Moreover, implementing thorough testing and validation processes is essential to identify potential issues early, ensuring that devices perform reliably and efficiently throughout their lifecycle.
Ultimately, advancements in embedded systems for medical devices present a significant opportunity for enhancing healthcare delivery. By adopting these best practices, engineers and developers can contribute to the creation of innovative solutions that improve patient monitoring, streamline healthcare processes, and ensure safety and compliance. As the demand for sophisticated medical technologies continues to rise, a commitment to excellence in embedded systems design will be vital for addressing the evolving challenges within the healthcare sector.
Frequently Asked Questions
What are the key components of embedded systems in medical devices?
The key components include microcontrollers and microprocessors, sensors, actuators, energy management solutions, communication interfaces, and software and firmware.
What role do microcontrollers and microprocessors play in embedded systems?
Microcontrollers and microprocessors serve as the brains of the embedded system, executing instructions and managing information, which is crucial for optimizing performance and power efficiency in devices.
How do sensors contribute to medical devices?
Sensors collect data from the environment or the human body, such as temperature, heart rate, and glucose levels, improving accuracy and responsiveness for real-time monitoring and enhancing patient outcomes.
What function do actuators serve in medical devices?
Actuators perform actions based on processed data, such as administering medication or adjusting equipment settings, enhancing the precision and reliability of medical interventions.
Why is energy management important for medical devices?
Effective power management is crucial for portable devices to extend battery life, especially as the demand for continuous monitoring grows. This involves selecting appropriate batteries and implementing power-saving modes.
How do communication interfaces enhance medical devices?
Communication interfaces enable devices to communicate with other systems using wireless communication modules like Bluetooth and Wi-Fi, improving connectivity and facilitating real-time information sharing among healthcare providers.
What is the significance of software and firmware in embedded systems?
Software governs hardware components and processes data, requiring design considerations for safety and compliance with industry standards, such as ISO 14971 for risk management, to ensure the safety and efficacy of healthcare devices.
How does Voler Systems ensure the performance of medical devices?
Voler Systems focuses on the thoughtful selection and integration of essential components, employing strategies like ultra-low power design and comprehensive IoT design consulting to meet stringent healthcare product requirements.
List of Sources
- Identify Key Components of Embedded Systems in Medical Devices
- Navigate 2026's Medical Device Manufacturing Trends (https://mddionline.com/medical-device-regulations/navigate-2026-s-medical-device-manufacturing-trends)
- Medical technology is unthinkable without embedded systems | embedded world (https://embedded-world.de/en/knowledge/2026/03/medical-technology-is-unthinkable-without-embedded-systems)
- Medical Devices Microcontrollers (MCU) Market Overview and Forecast 2026 - 2033: A Detailed Study of MARKET TYPE and MARKET APPLICATION (https://linkedin.com/pulse/medical-devices-microcontrollers-mcu-market-overview-forecast-lxtrf)
- Embedded Systems for Smart Medical Wearables in 2026 | Design and Connectivity Strategies (https://promwad.com/news/embedded-systems-smart-medical-wearables-2026-design-and-connectivity-strategies)
- What’s New in MedTech in 2026 — And What It Means for Converge (https://linkedin.com/pulse/whats-new-medtech-2026-what-means-converge-lq2te)
- Address Regulatory Compliance and Safety Standards
- FDA Implements Quality Management System Regulation for Medical Devices (https://aabb.org/news-resources/news/article/2026/02/04/fda-implements-quality-management-system-regulation-for-medical-devices)
- 2026 Medical Device Regulations: EUDAMED, FDA QMSR, and Compliance Risk - The Richmond Group USA (https://richgroupusa.com/2026-medical-device-regulations-eudamed-fda-qmsr)
- Top Benefits of ISO 13485: Real-World Examples (https://theknowledgeacademy.com/blog/benefits-of-iso-13485)
- 8 Benefits of ISO 13485 Certification for Medical Device Manufacturing (https://amtivo.com/us/resources/insights/iso-13485-medical-device-certification-benefits)
- What the FDA’s New Quality Requirements Mean for U.S. Medical Device Companies (https://amtivo.com/us/resources/insights/fdas-new-quality-requirements-mean-for-us-medical-device-companies)
- Incorporate Advanced Technologies for Enhanced Functionality
- Number of connected IoT devices growing 14% to 21.1 billion (https://iot-analytics.com/number-connected-iot-devices)
- IoT Device Growth Statistics & Trends (2026) (https://thenetworkinstallers.com/blog/iot-device-growth-statistics)
- What’s New in MedTech in 2026 — And What It Means for Converge (https://linkedin.com/pulse/whats-new-medtech-2026-what-means-converge-lq2te)
- Machine Learning Statistics for 2026: The Ultimate List (https://itransition.com/machine-learning/statistics)
- The State of AI in Healthcare: Market Growth and Key Stats (https://patentpc.com/blog/the-state-of-ai-in-healthcare-market-growth-and-key-stats)
- Implement Rigorous Testing and Validation Protocols
- 21 inspirational quotes about software testing (https://testlio.com/blog/21-inspirational-quotes-about-software-testing)
- Medical Device Testing in Transition (https://mpo-mag.com/medical-device-testing-in-transition)
- 62 Software testing quotes to inspire you (https://globalapptesting.com/blog/software-testing-quotes)
- 50 Inspirational Quotes About Software Testing - QA Madness (https://qamadness.com/inspirational-quotes-about-software-testing)
- Software Testing: A Component of Medical Device Quality (https://cleio.com/insights/blog/software-testing-a-component-of-medical-device-quality)





