Introduction
In the rapidly evolving medical device industry, the stakes are higher than ever due to increasingly stringent regulatory standards. The pre-silicon design phase is a critical foundation for both compliance and innovation, providing manufacturers with a unique opportunity to streamline development while ensuring patient safety. However, a significant challenge persists: how can teams effectively navigate the complexities of regulatory requirements and quality assurance without compromising creativity and efficiency? This article examines four best practices that not only enhance compliance but also foster collaboration and leverage advanced technologies, thereby setting the stage for successful medical device development.
Establish Compliance Standards Early in Pre-Silicon Design
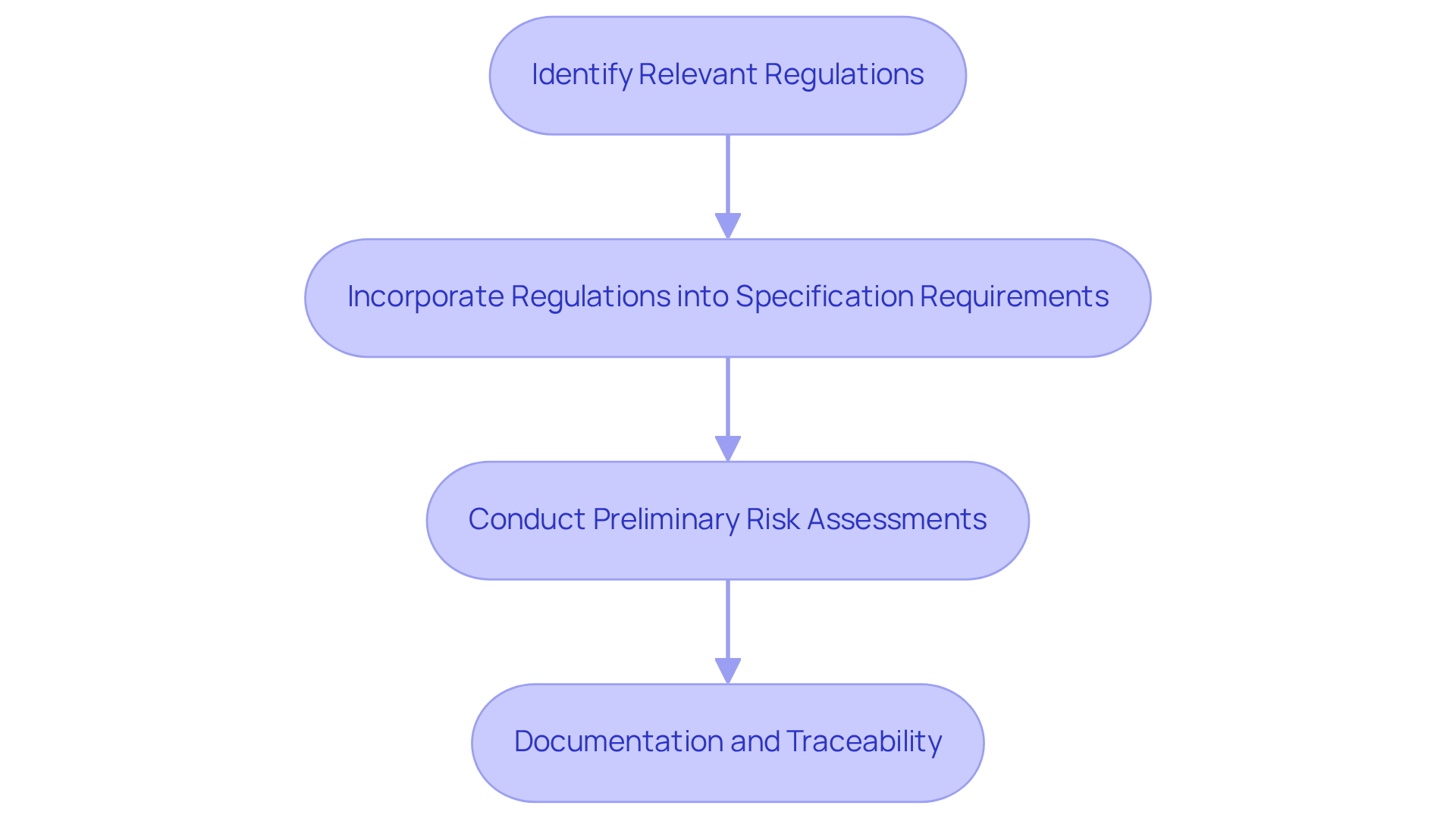
In the , adherence to is crucial. Establishing these standards early in the creation phase can significantly streamline the . The following key steps are essential for implementation:
- Identify Relevant Regulations: Begin by pinpointing the specific regulations applicable to your device, including the , and updated ISO standards. This alignment ensures that your creation process adheres to essential regulatory requirements.
- Incorporate Regulations into Specification Requirements: Integrate regulatory needs into your specification requirements from the outset. This involves defining performance metrics, safety standards, and usability criteria that meet regulatory expectations, particularly in light of the new , which emphasizes .
- Conduct Preliminary Risk Assessments: Early risk assessments are vital for identifying potential compliance issues before they escalate. Employ tools such as to evaluate risks associated with design choices, as the FDA's focus on risk management will be a key aspect of inspections under the new regulations.
- Documentation and Traceability: Maintain comprehensive records of and modifications. This practice not only facilitates but also ensures traceability throughout the development process, which is increasingly scrutinized during FDA inspections.
By embedding adherence into the structural framework, teams can avoid costly revisions and ensure a more seamless path to market approval, ultimately enhancing and operational efficiency.
Implement Rigorous Quality Assurance Protocols
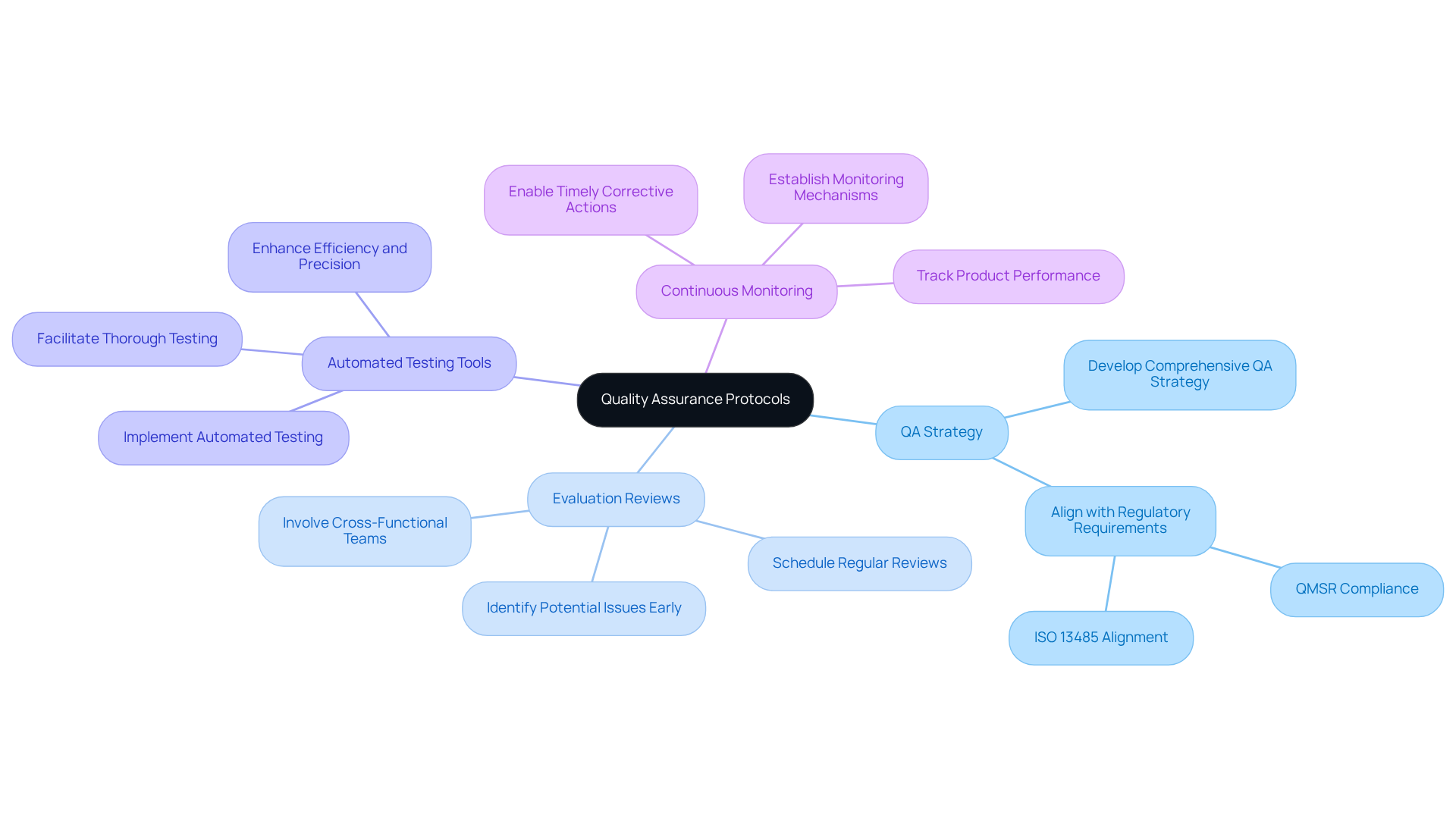
Quality assurance (QA) is crucial in the development of medical devices, ensuring compliance with safety and efficacy standards. To implement effective QA protocols, consider the following best practices:
- Create a Thorough : Develop a comprehensive QA strategy that delineates processes, responsibilities, and metrics for quality assurance throughout the creation and development phases. This plan must align with , including the new (QMSR) effective February 2, 2026, which emphasizes risk management and lifecycle accountability. can assist startups in navigating these regulatory challenges, ensuring compliance with documentation from the outset.
- Conduct Regular : Schedule frequent evaluation reviews to assess adherence to QA standards. Involving cross-functional teams in these reviews integrates diverse perspectives and expertise, thereby enhancing the overall quality of the device. This collaborative approach is essential for early identification of potential issues during the design phase, a critical step in successful engineering design projects.
- Utilize Automated Testing Tools: Implement automated testing tools to optimize the QA process. These tools enhance efficiency and precision, facilitating thorough testing of functionality and performance, which is vital for meeting the stringent standards established by the . Leveraging innovative technology in testing can significantly improve the reliability of electronic equipment.
- Implement : Establish mechanisms for continuous monitoring to track product performance and quality metrics throughout the development lifecycle. This proactive approach enables early identification of issues and timely corrective actions, ultimately enhancing product reliability and mitigating the risk of post-market complications. By incorporating continuous monitoring into the QA framework, organizations can ensure consistent compliance with quality standards and regulatory requirements.
By emphasizing stringent QA protocols, organizations can significantly enhance product reliability and ensure adherence to evolving regulatory standards, thereby safeguarding patient safety and equipment effectiveness. Furthermore, addressing common errors in manufacturing tests can further strengthen the quality assurance process. [Voler Systems](https://volersystems.com)' expertise in documentation adherence and quality assurance can provide invaluable support to medical equipment manufacturers striving for success in their projects.
Foster Cross-Functional Collaboration for Effective Design
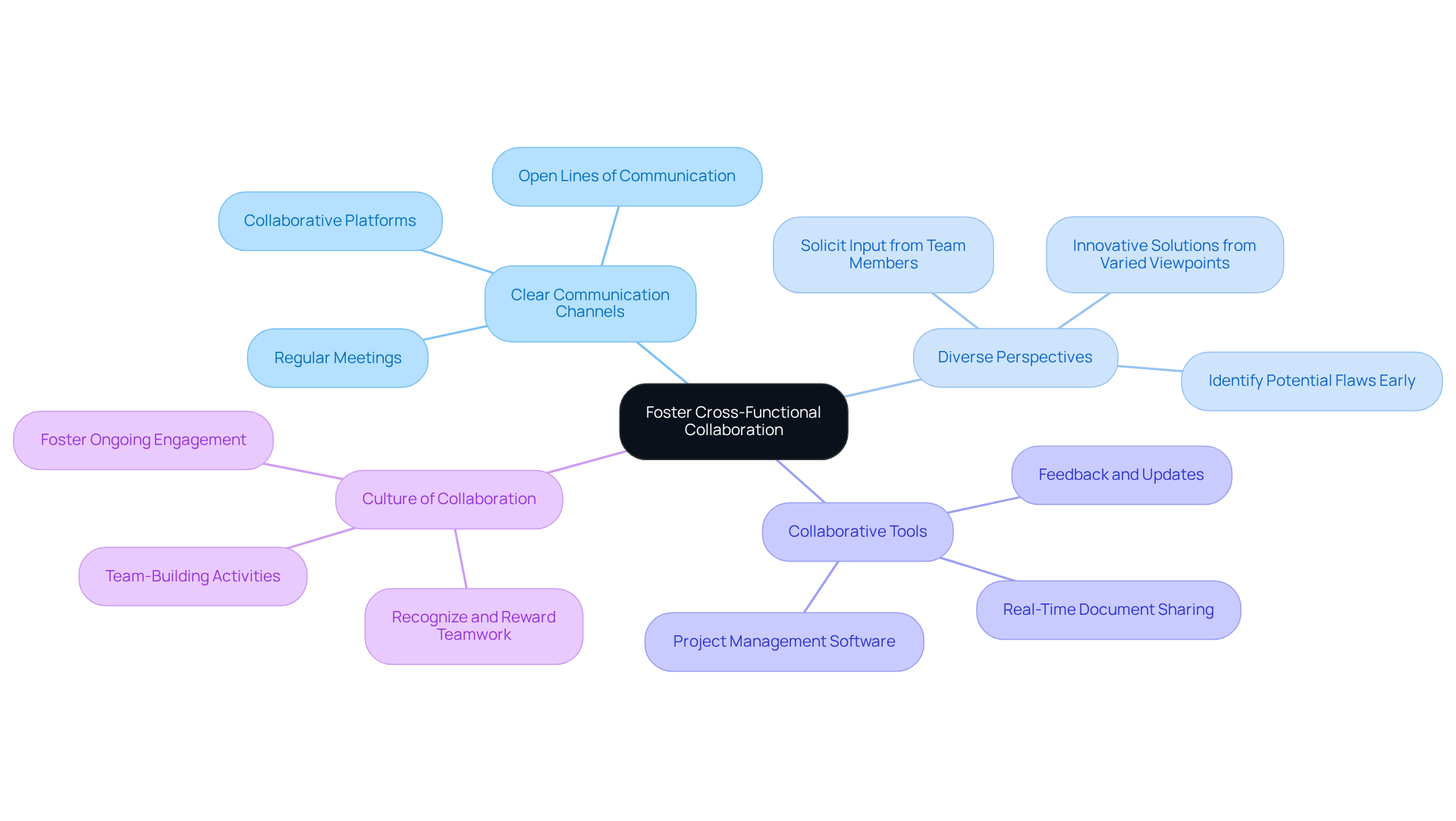
Successful creation in medical instruments necessitates contributions from diverse fields, including engineering, regulatory affairs, and clinical expertise. To enhance , consider the following strategies:
- Establish : Develop open lines of communication among team members from various disciplines. Regular meetings and collaborative platforms facilitate information sharing and alignment on project goals, significantly improving .
- : Actively solicit input from team members with differing backgrounds and expertise. Varied viewpoints can lead to and help identify potential flaws early in the development process, ultimately enhancing device quality and compliance with regulatory standards.
- Implement : Employ collaborative tools and software that enable real-time sharing of project documents, feedback, and updates. This enhances clarity and ensures all team members are informed about project advancements, which is crucial for maintaining momentum during the creation phase.
- Promote a : Cultivate a culture that values teamwork and collaboration. Recognizing and rewarding collaborative efforts fosters ongoing engagement and cooperation among team members, which is essential for navigating the complexities of .
By prioritizing cross-functional cooperation, organizations can significantly enhance the efficiency of their development efforts, leading to improved product outcomes and adherence to industry standards.
Leverage Advanced Technologies for Enhanced Design Efficiency
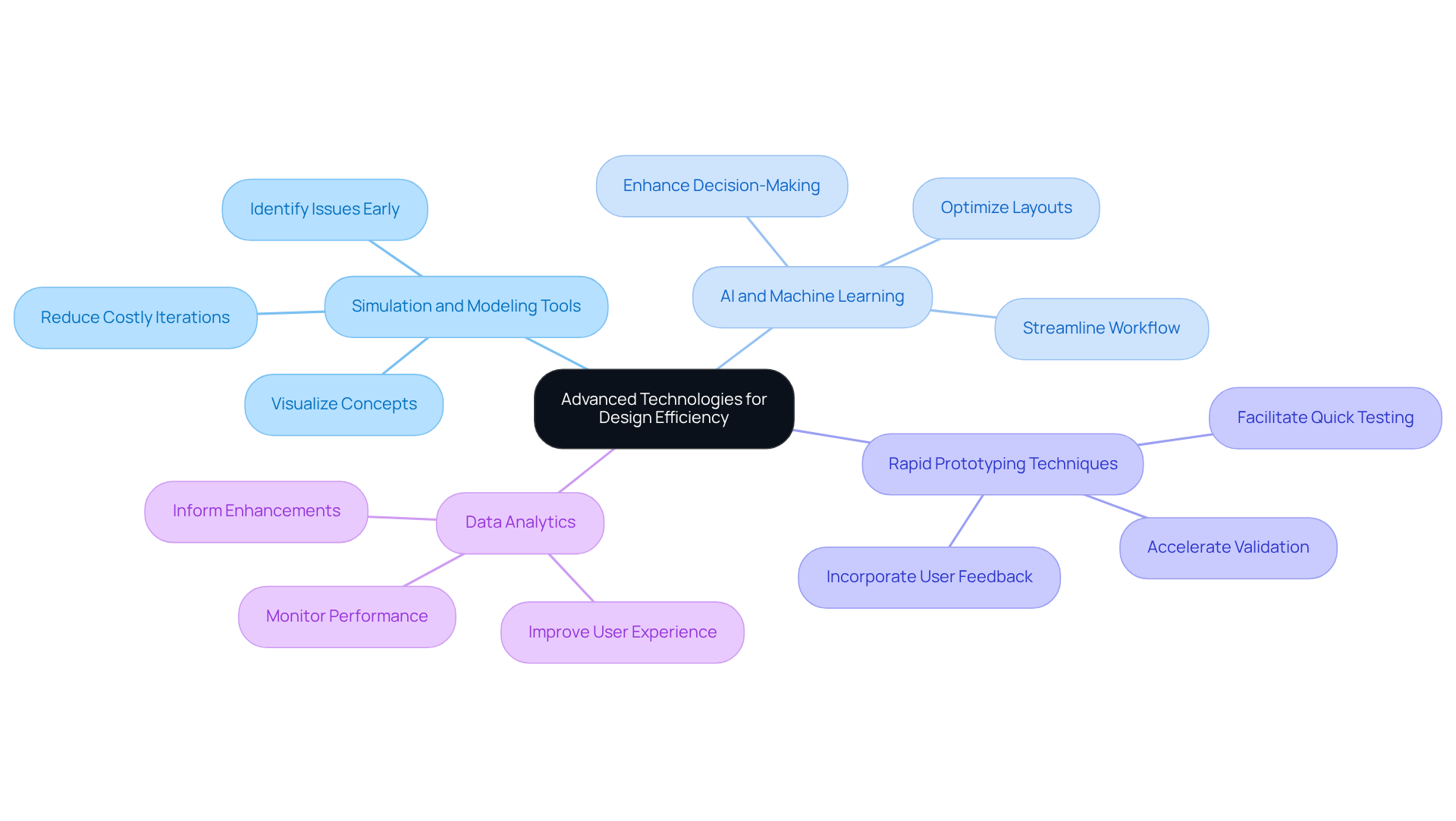
Integrating into the process can lead to significant improvements in both efficiency and effectiveness. Key technologies to consider include:
- : Utilizing allows teams to visualize and test concepts prior to physical prototyping. This approach helps identify potential issues early, thereby significantly reducing the need for costly iterations.
- : The implementation of AI and machine learning algorithms can analyze data related to layout optimization. These technologies provide valuable insights that enhance decision-making and streamline the creation workflow, ultimately leading to more innovative solutions.
- : The use of rapid prototyping techniques, such as 3D printing, facilitates the quick creation and testing of physical models. This accelerates the validation process and allows for faster iterations based on user feedback, ensuring that products meet effectively.
- : Employing data analytics to monitor performance and user interactions is crucial. Analyzing this data informs enhancements and improves user experience, making products more intuitive and effective.
By adopting these advanced technologies, organizations can improve , shorten time to market, and enhance overall product quality, positioning themselves for success in the competitive medical device landscape. Furthermore, considering environmental factors and user adoption can further enhance the effectiveness of , ensuring they address the diverse needs of users.
Conclusion
Establishing effective pre-silicon design practices in medical devices is essential for ensuring compliance, quality, and overall success in the development process. By prioritizing regulatory standards from the outset, implementing rigorous quality assurance protocols, fostering cross-functional collaboration, and leveraging advanced technologies, organizations can significantly enhance their design efficiency and product reliability. These practices not only streamline the development workflow but also contribute to improved patient safety and operational excellence.
Key insights from the article underscore the importance of early compliance with regulations, such as FDA guidelines and ISO standards, which lay the groundwork for a successful design process. The integration of quality assurance strategies and continuous monitoring further ensures that products meet safety and efficacy standards. Moreover, fostering collaboration among diverse teams can lead to innovative solutions, while advanced technologies like AI and rapid prototyping can accelerate the design timeline and enhance product quality.
In conclusion, embracing these best practices in pre-silicon design is crucial for medical device manufacturers navigating the complexities of regulatory compliance and market demands. By committing to these strategies, organizations can achieve regulatory success and position themselves as leaders in the competitive medical device landscape, ultimately delivering safer and more effective products to the market.
Frequently Asked Questions
Why is it important to establish compliance standards early in pre-silicon design for medical equipment?
Establishing compliance standards early is crucial as it significantly streamlines the pre-silicon development workflow and ensures adherence to regulatory requirements, enhancing patient safety and operational efficiency.
What are the first steps to take in establishing compliance standards?
The first steps include identifying relevant regulations, such as the latest FDA guidelines and updated ISO standards, and incorporating these regulations into specification requirements from the outset.
How should regulations be incorporated into specification requirements?
Regulations should be integrated by defining performance metrics, safety standards, and usability criteria that meet regulatory expectations, particularly in light of the new Quality Management System Regulation (QMSR) that emphasizes risk management.
What is the role of preliminary risk assessments in compliance?
Preliminary risk assessments are vital for identifying potential compliance issues early on. Tools like Failure Mode and Effects Analysis (FMEA) can be used to evaluate risks associated with design choices, which is important given the FDA's focus on risk management.
Why is documentation and traceability important in the compliance process?
Maintaining comprehensive records of compliance-related decisions and modifications is important as it facilitates regulatory submissions and ensures traceability throughout the development process, which is crucial during FDA inspections.
What benefits can teams expect from embedding compliance into their structural framework?
By embedding compliance into the structural framework, teams can avoid costly revisions and ensure a more seamless path to market approval, ultimately enhancing patient safety and operational efficiency.
List of Sources
- Establish Compliance Standards Early in Pre-Silicon Design
- FDA’s 2026 Expectations: Clinical Decision Support Compliance (https://about.citiprogram.org/blog/clinical-decision-support-compliance-fdas-2026-expectations)
- FDA Aligns U.S. Medical Device Rules with Global Standards (https://mddionline.com/regulatory-quality/medical-device-companies-must-navigate-historic-fda-rule-change)
- FDA Tightens Its Medical Device Cybersecurity Guidance (https://fedtechmagazine.com/article/2026/03/fda-tightens-its-medical-device-cybersecurity-guidance-perfcon)
- Key Updates in FDA’s 2026 General Wellness and Clinical Decision Support Software Guidance | Faegre Drinker Biddle & Reath LLP (https://faegredrinker.com/en/insights/publications/2026/1/key-updates-in-fdas-2026-general-wellness-and-clinical-decision-support-software-guidance)
- A QMSR State of Mind: FDA Adopts New Inspection Approach for Medical Devices as Quality Management System Regulation Takes Effect | Insights | Ropes & Gray LLP (https://ropesgray.com/en/insights/alerts/2026/02/a-qmsr-state-of-mind-fda-adopts-new-inspection-approach-for-medical-devices)
- Implement Rigorous Quality Assurance Protocols
- Top Medical Device Regulations to Watch in 2026 (https://xtalks.com/top-medical-device-regulations-to-watch-in-2026-4549)
- QMSR 2026 Explained: FDA Quality Management System Regulation & ISO 13485 Alignment Guide (https://sushvin.com/qmsr-2026-fda-quality-management-system-regulation.html)
- Overview of the FDA's new Quality Management System Regulation: effective from 2 February 2026 - ECA Academy (https://gmp-compliance.org/gmp-news/overview-of-the-fdas-new-quality-management-system-regulation-effective-from-2-february-2026)
- A QMSR State of Mind: FDA Adopts New Inspection Approach for Medical Devices as Quality Management System Regulation Takes Effect | Insights | Ropes & Gray LLP (https://ropesgray.com/en/insights/alerts/2026/02/a-qmsr-state-of-mind-fda-adopts-new-inspection-approach-for-medical-devices)
- FDA QMSR & ISO 13485: Key Changes Effective 2026 | IntuitionLabs (https://intuitionlabs.ai/articles/fda-qmsr-iso-13485-changes-2026)
- Foster Cross-Functional Collaboration for Effective Design
- Want to Drive Innovation? You Need Cross-Functional Collaboration (https://americanhealthcareleader.com/2025/drive-innovation-with-cross-functional-collaboration)
- The Importance of Collaboration Within Medical Device Manufacturing | Ranfac (https://ranfac.com/the-importance-of-collaboration-within-medical-device-manufacturing)
- The Importance of Collaboration in Medical Device Manufacturing. - PolarSeal (https://polarseal.net/blog/collaboration-medical-device-manufacturing)
- Cross-Disciplinary Collaboration Enhances Medical Device Design (https://blackhagendesign.com/2024/07/18/cross-disciplinary-collaboration-enhances-medical-device-design)
- The Role of Collaboration in Medical Device Innovation: Partnering with Experts for Breakthrough Solutions (https://occamdesign.com/the-role-of-collaboration-in-medical-device-innovation-partnering-with-experts-for-breakthrough-solutions)
- Leverage Advanced Technologies for Enhanced Design Efficiency
- 6 healthcare tech trends for 2026 (https://medtronic.com/en-us/our-company/stories/6-healthcare-tech-trends-for-2026.html)
- CES 2026: Medical technologies you need to know (https://medicaldesignandoutsourcing.com/ces-2026-medical-technologies-you-need-to-know)
- 4 medtech topics to watch in 2026 (https://medtechdive.com/news/4-medtech-topics-trends-2026/809091)
- MedTech Trends Shaping 2026 (https://iqvia.com/blogs/2026/02/medtech-trends-shaping-2026)
- Medtech trends 2026: From AI strategy to execution | ZS (https://zs.com/insights/medtech-trends-2026-industry-strategy-and-execution)
![]()



