Essential Best Practices for Embedded Systems in Medical Devices
Explore essential best practices for embedded systems basic in medical devices to enhance...

The integration of wireless technologies into medical devices signifies a fundamental transformation in healthcare delivery. This article outlines best practices for ensuring compliance with regulatory standards while improving patient safety and device functionality. However, rapid technological advancements present challenges in selecting appropriate wireless solutions and establishing rigorous testing protocols. Manufacturers must navigate these complexities to effectively leverage wireless innovations while ensuring patient outcomes are safeguarded.
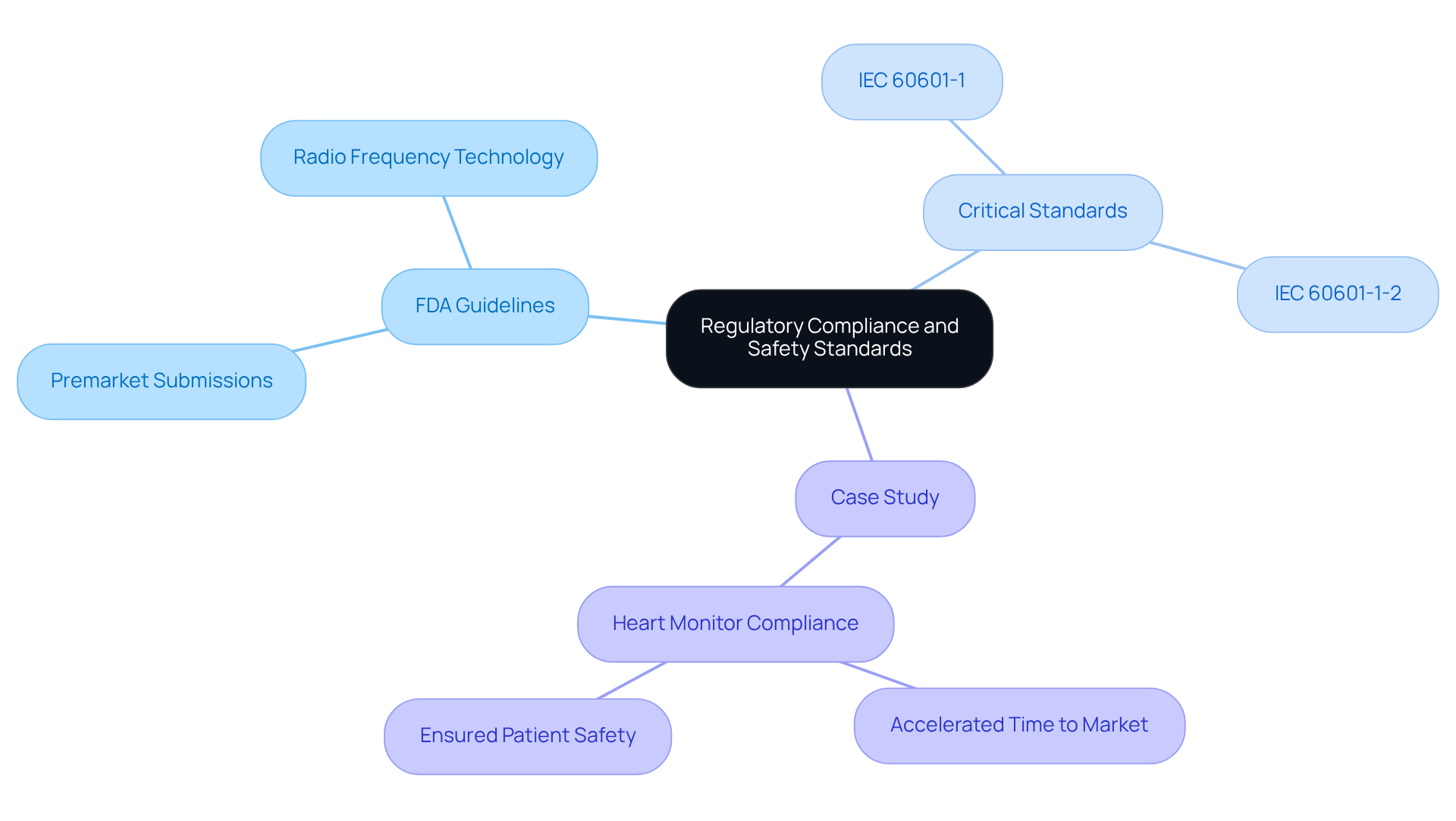
Navigating the integration of wireless technologies into medical instruments presents significant regulatory and safety challenges that manufacturers must address. The FDA outlines essential guidelines, including the requirement for premarket submissions that demonstrate the safety and effectiveness of the product.
Critical standards include:
Manufacturers must also consider specific requirements for radio technologies, as detailed in the FDA's guidance on radio frequency technology. Adhering to these standards mitigates legal risks and enhances the product's reputation within the industry.
For instance, a case study involving a heart monitor demonstrated that compliance with these standards greatly accelerated the time to market while ensuring patient safety. By 2026, the evolving IEC 60601-1 standards will further underscore the critical importance of safety and effectiveness in healthcare products, reflecting the industry's commitment to high-quality healthcare solutions.
Failure to comply with these standards can result in delayed product launches and increased scrutiny from regulatory bodies.
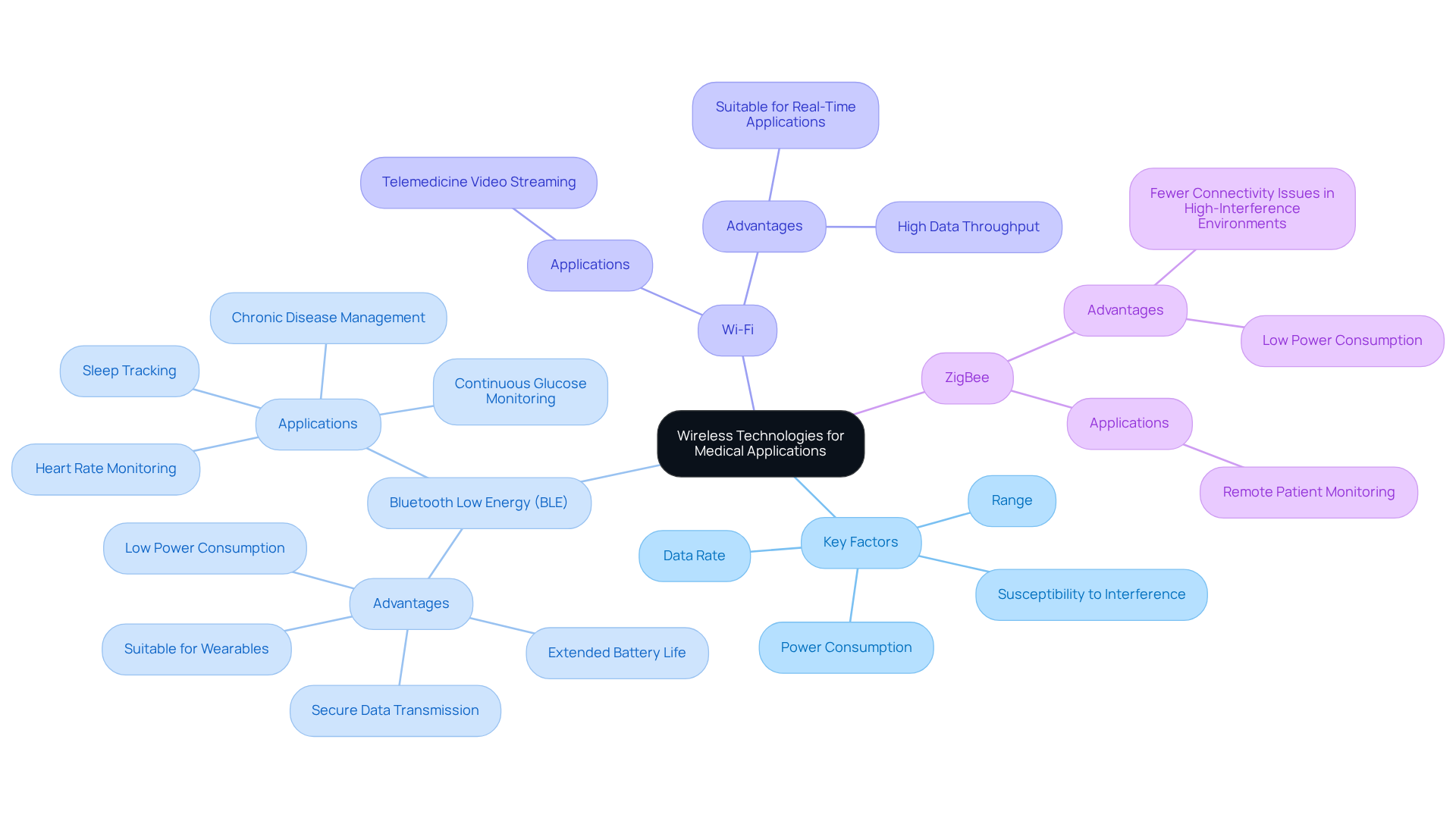
Selecting the appropriate wireless technologies is critical for ensuring the reliability and effectiveness of medical devices across diverse healthcare settings. Key factors to consider in this selection process are:
Bluetooth Low Energy (BLE) is an ideal choice for wearables due to its low power requirements, allowing for extended operation without frequent recharging. Conversely, Wi-Fi is better suited for applications that require high data throughput, such as real-time video streaming in telemedicine.
Moreover, the operational environment significantly influences the selection of communication technology. Research indicates that remote patient monitoring tools using ZigBee technology experience fewer connectivity issues in high-interference environments than those relying on standard Wi-Fi. ZigBee's effectiveness in these settings underscores the importance of thoroughly analyzing the operational context and specific equipment requirements when choosing the right communication technology for healthcare applications. Furthermore, companies like Voler Systems are leveraging AI-driven power management solutions to extend battery life, thereby improving the performance of wearable technology.
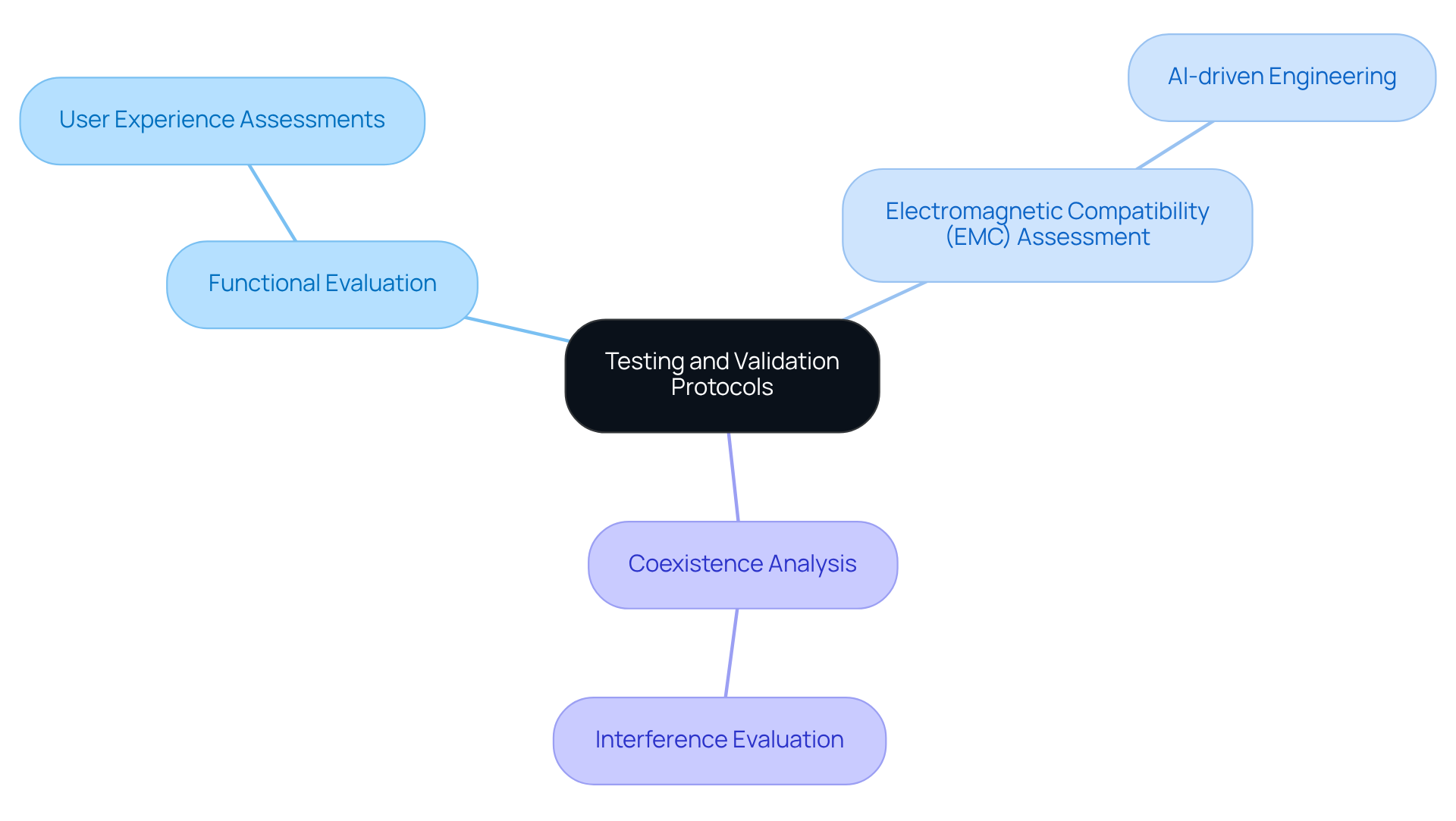
Ensuring the reliability and safety of remote health instruments is paramount for manufacturers in the medical device industry. To achieve this, producers must implement rigorous evaluation and confirmation procedures. This includes:
A significant case study on wireless technologies, particularly a wireless insulin pump, highlighted the importance of evaluating interference from other medical equipment, as such disruptions could jeopardize functionality. Additionally, validation must include user experience assessments to ensure the device is intuitive and meets the needs of healthcare providers and patients.
By adhering to established evaluation standards, such as those from the FDA and IEC, manufacturers can mitigate the risk of equipment failure and enhance patient safety. In 2026, the FDA continues to enforce strict evaluation standards, underscoring the significance of EMC assessment in ensuring the safety and effectiveness of health-related equipment. Industry leaders advocate for extensive EMC evaluations, recognizing wireless technologies as a fundamental aspect of healthcare product safety, especially in a progressively interconnected health environment.
Furthermore, the incorporation of AI-driven engineering in the design process can improve the precision of testing and validation, while Voler Systems assists healthcare product firms in managing compliance and optimizing manufacturing tests, including legacy test equipment, ensuring quality and efficiency in electronic product design. As the industry evolves, the integration of advanced technologies and stringent evaluation processes will be crucial in safeguarding patient outcomes.
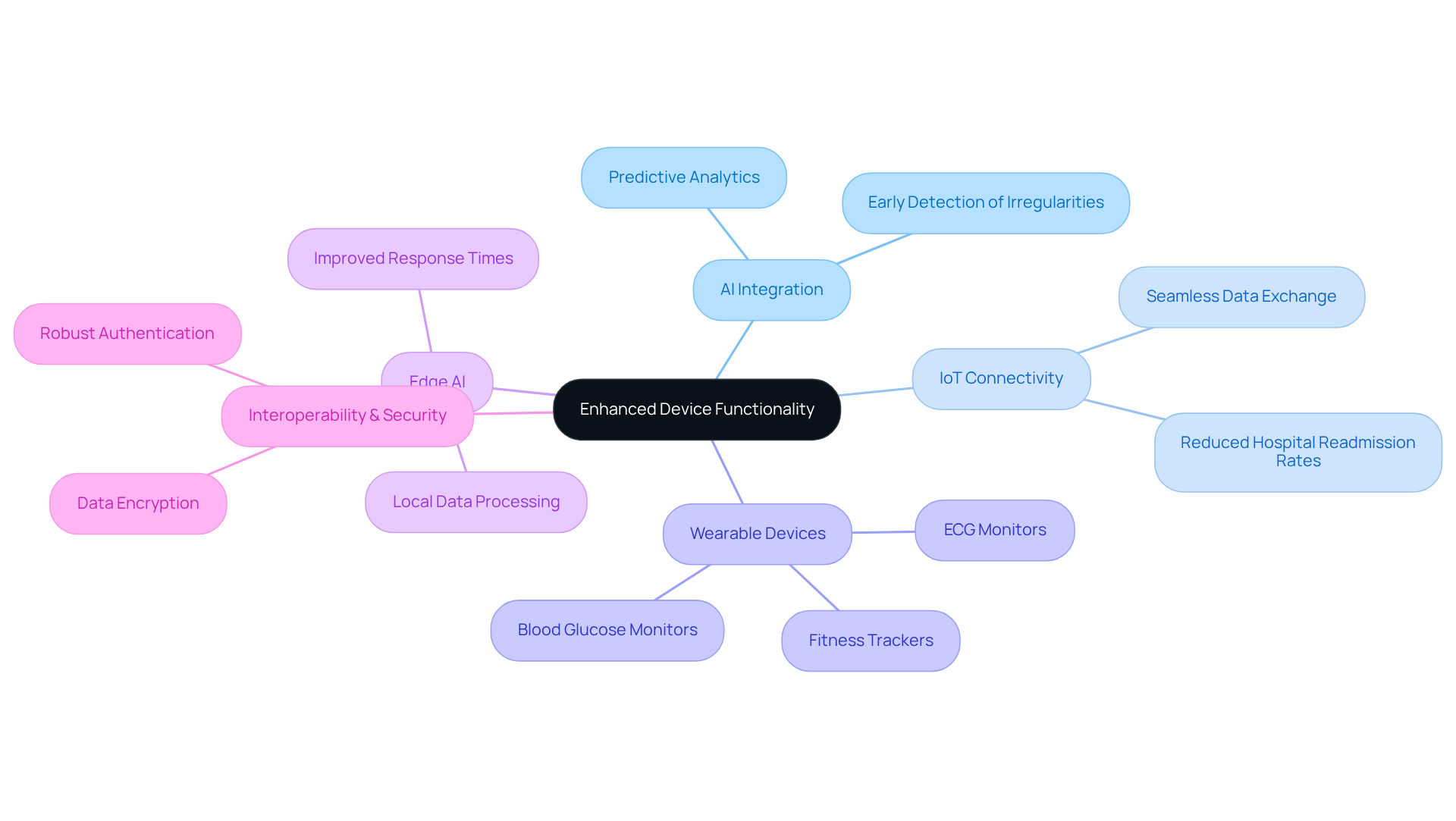
The integration of wireless technologies, AI, and IoT into medical equipment presents a transformative opportunity for enhancing patient care. Voler Systems employs artificial intelligence for predictive analytics. This capability allows equipment to anticipate patient needs and adapt accordingly. For instance, intelligent wearable devices, such as blood glucose monitors and ECG monitors, utilize AI algorithms to detect irregularities and promptly notify healthcare professionals.
Furthermore, the integration of Edge AI enables these devices to process data locally, reducing latency and improving response times. Voler Systems excels in developing ultra-low-power wearable sensors, ensuring efficiency and effectiveness across various environments, even in challenging conditions like moisture and temperature.
Wireless technologies enable IoT connectivity, facilitating seamless data exchange between devices and healthcare systems, which is crucial for remote monitoring and enhancing patient engagement. Research demonstrates that IoT-enabled gadgets, especially those for tracking respiratory conditions, significantly reduce hospital readmission rates through continuous monitoring and timely interventions, with patients using wearables being 38% less likely to be hospitalized for heart failure within 90 days.
Additionally, the shift from tethered to truly wireless technologies in cardiac monitoring exemplifies Voler Systems' impact on enhancing patient care. Manufacturers must prioritize interoperability and data security to effectively incorporate these technologies, ensuring devices communicate efficiently while safeguarding patient information through end-to-end data encryption and robust authentication.
Integrating wireless technologies into medical devices presents significant challenges that require meticulous attention to detail. This process is not merely about enhancing functionality; it demands careful consideration of regulatory compliance, technology selection, rigorous testing, and the incorporation of advanced innovations like AI and IoT. Adhering to safety standards and regulatory guidelines is crucial for ensuring the safety and effectiveness of medical devices while fostering trust within the healthcare industry.
The article highlights critical factors involved in this integration process, from understanding regulatory frameworks such as the FDA guidelines and IEC standards to selecting the most suitable wireless technologies based on specific application needs. It emphasizes the necessity of implementing thorough testing and validation protocols to mitigate risks and enhance patient safety. The integration of AI and IoT technologies exemplifies how these advancements can enhance patient care through predictive analytics and efficient data exchange, contributing to improved health outcomes.
As the healthcare landscape continues to evolve, manufacturers must prioritize effective strategies to navigate the challenges of wireless technology integration effectively. Embracing these strategies not only ensures compliance and safety but also positions medical devices at the forefront of innovation, paving the way for improved patient engagement and care. The commitment to excellence in integrating these technologies will ultimately shape the future of healthcare delivery and patient outcomes.
What are the main regulatory challenges in integrating wireless technologies into medical instruments?
The main regulatory challenges include navigating essential guidelines set by the FDA, which require premarket submissions that demonstrate the safety and effectiveness of the product.
What critical standards must manufacturers adhere to for medical instruments?
Manufacturers must adhere to IEC 60601-1 for electrical safety and IEC 60601-1-2 for electromagnetic compatibility.
What specific requirements should manufacturers consider for radio technologies?
Manufacturers should refer to the FDA's guidance on radio frequency technology to understand the specific requirements for radio technologies.
How does compliance with regulatory standards impact a medical product's market readiness?
Compliance with regulatory standards can greatly accelerate the time to market while ensuring patient safety, as demonstrated by a case study involving a heart monitor.
What will be the impact of the evolving IEC 60601-1 standards by 2026?
The evolving IEC 60601-1 standards will further emphasize the importance of safety and effectiveness in healthcare products, reflecting the industry's commitment to high-quality healthcare solutions.
What are the potential consequences of failing to comply with regulatory standards?
Failure to comply with these standards can result in delayed product launches and increased scrutiny from regulatory bodies.
