Introduction
The rapid evolution of technology in healthcare is fundamentally transforming the communication and operation of medical devices, with Machine-to-Machine (M2M) communication leading this change. This technology significantly enhances monitoring capabilities and streamlines operations, ultimately aiming to improve patient outcomes.
However, as M2M integration in medical devices becomes increasingly prevalent, challenges emerge regarding:
- Regulatory compliance
- Effective testing
- The implementation of advanced features such as artificial intelligence
Manufacturers must navigate these complexities to fully harness the potential of M2M technology in healthcare.
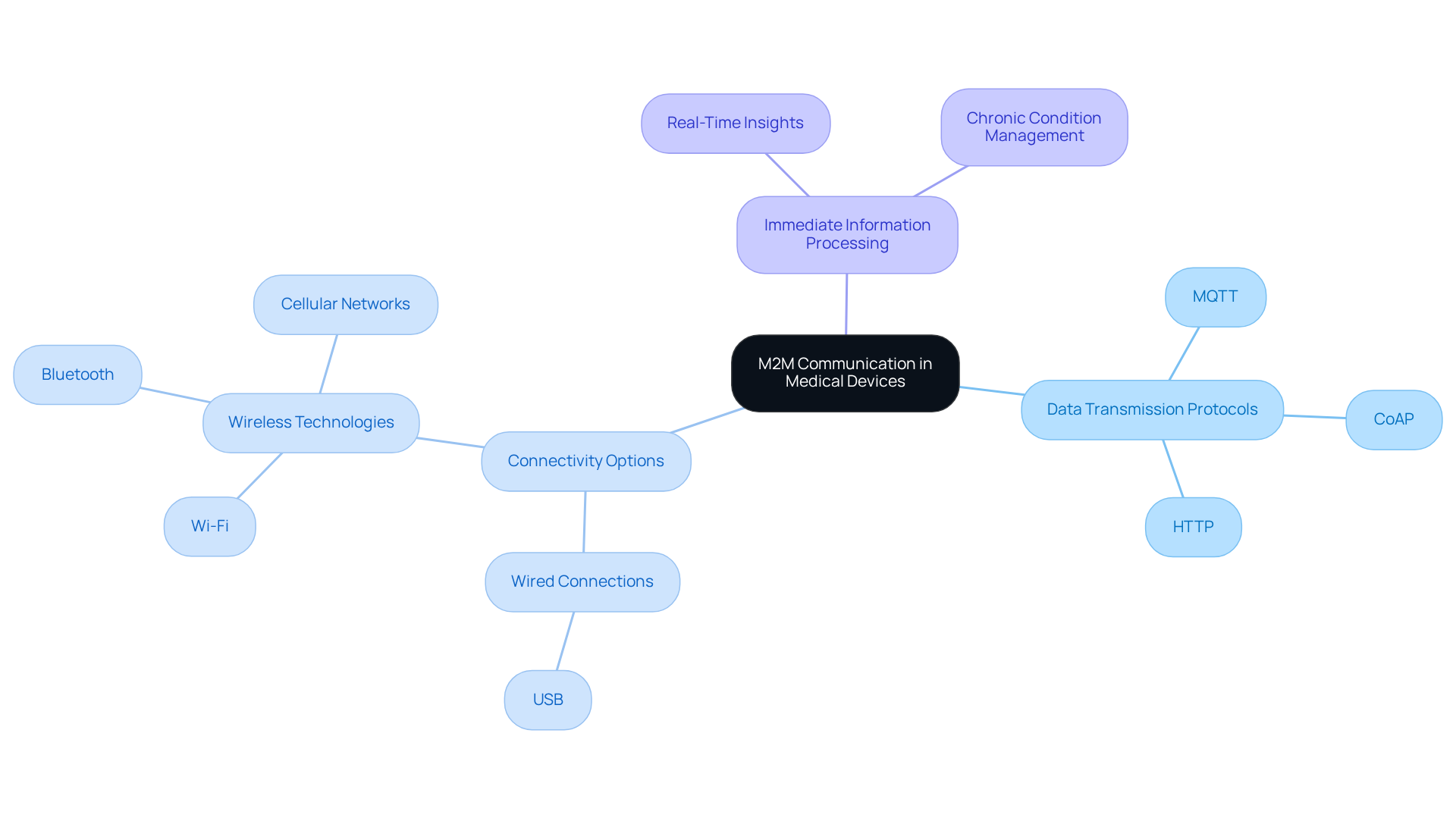
Understand M2M Communication Fundamentals in Medical Devices
Machine-to-Machine (M2M) communication facilitates the automated transfer of information between units without human intervention, significantly enhancing monitoring capabilities and operational efficiency in . This technology allows for seamless information exchange among devices, sensors, and systems, which is crucial for .
Key components of include:
- : Protocols such as MQTT, CoAP, and HTTP are essential for ensuring reliable data exchange. These protocols safeguard the integrity and security of transmitted data, which is particularly important in , where patient information is sensitive.
- : can employ various , including wired connections like USB and wireless technologies such as Wi-Fi, Bluetooth, and cellular networks. The choice of connectivity affects device performance and its ability to integrate into existing infrastructures.
- : M2M systems often necessitate real-time information processing capabilities to deliver timely insights for . This immediacy is especially beneficial for managing chronic conditions, where daily data can inform treatment adjustments and enhance care management.
By mastering these fundamentals, engineers can develop devices that not only comply with regulatory standards but also leverage enhanced connectivity and data utilization to foster improved . The integration of is revolutionizing the field, enabling more intelligent and responsive solutions. As the sector evolves, the application of M2M will be instrumental in shaping the future of patient care.
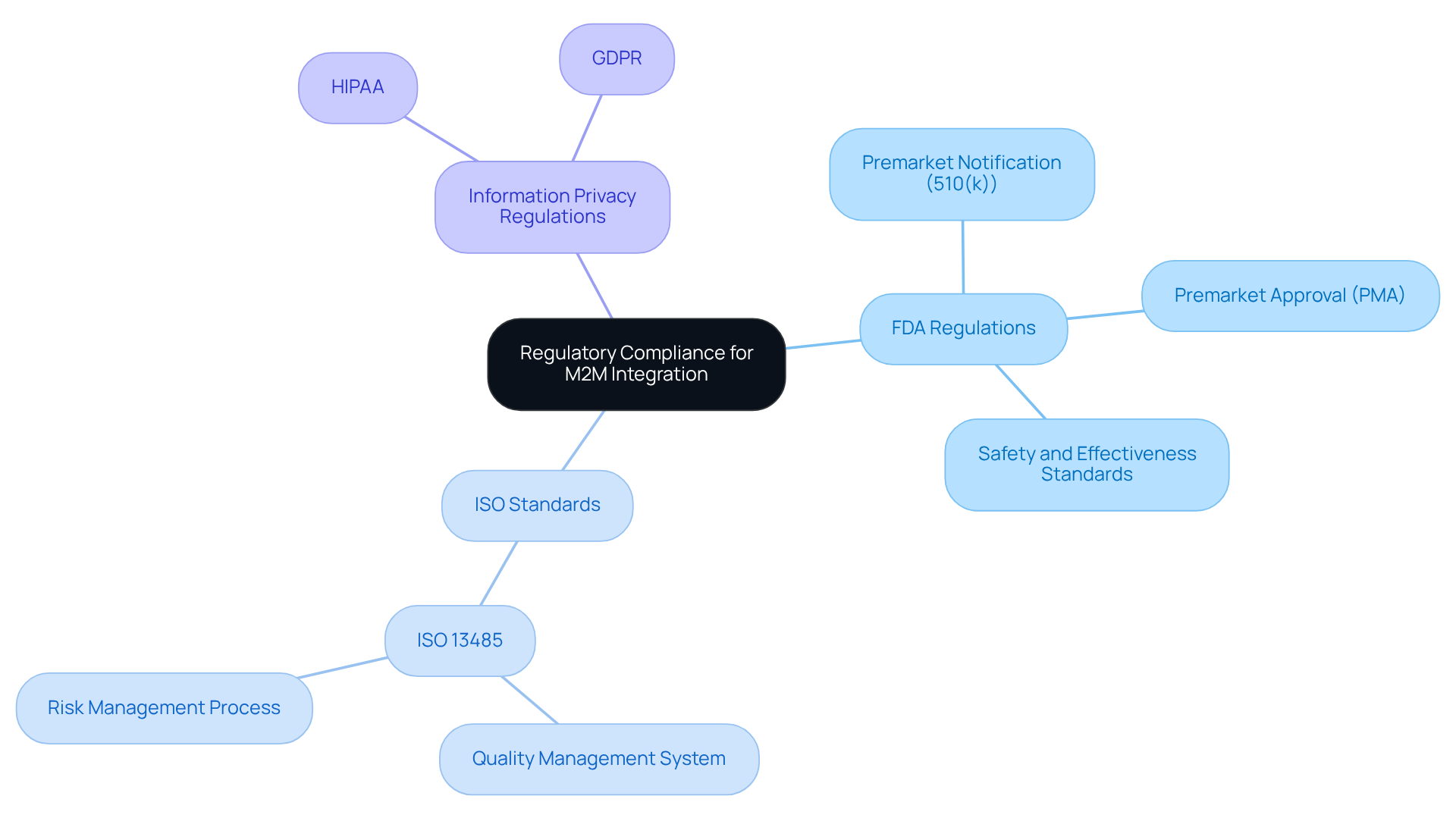
Ensure Regulatory Compliance for M2M Integration
plays a vital role in the application of in . The following key regulations must be considered:
- : The FDA requires that all meet specific safety and effectiveness standards. Depending on the classification of the device, manufacturers are obligated to submit either a premarket notification (510(k)) or a premarket approval (PMA).
- ISO Standards: Compliance with is critical, as it ensures that manufacturers implement a throughout the product lifecycle, thereby enhancing reliability and safety.
- : The application of M2M is essential for compliance with HIPAA and GDPR, safeguarding patient information transmitted via M2M systems and ensuring that privacy is upheld during data exchange.
To achieve compliance, manufacturers should perform thorough , maintain detailed documentation, and actively engage with regulatory bodies throughout the development process.
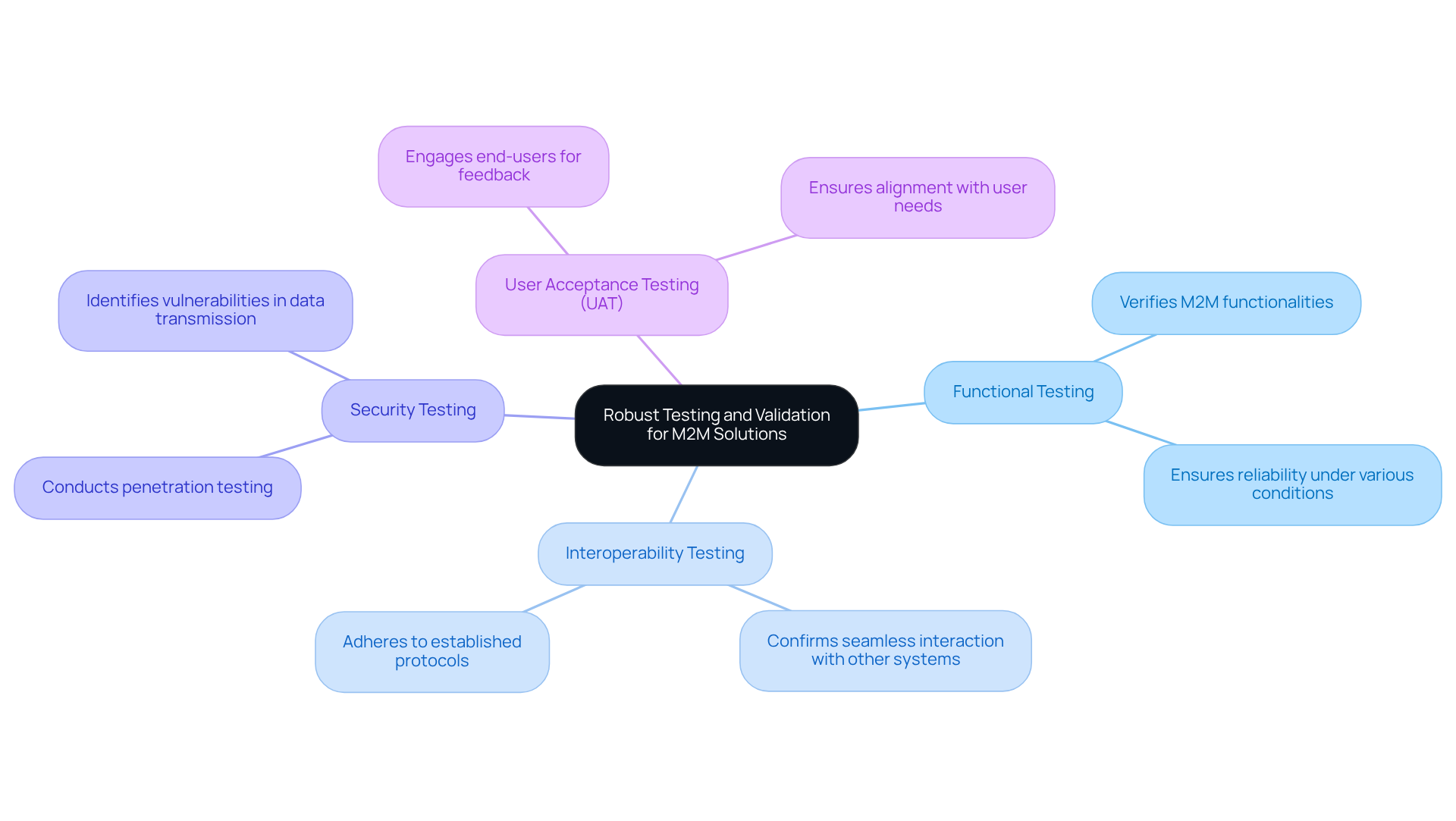
Implement Robust Testing and Validation for M2M Solutions
Robust testing and validation are crucial for , ensuring compliance with . The following key practices are essential:
- : This involves verifying that all operate as intended under various conditions, thereby ensuring reliability in real-world applications.
- : It is vital to confirm that devices can interact seamlessly with other systems and components, adhering to established protocols to facilitate .
- : Conducting penetration testing is necessary to identify and mitigate vulnerabilities in data transmission and storage, thereby protecting sensitive patient information.
- : Engaging end-users in the testing process is important for gathering valuable feedback on usability and functionality, ensuring that the product aligns with user needs.
By implementing these , manufacturers can enhance the safety, effectiveness, and market readiness of their health-related products.
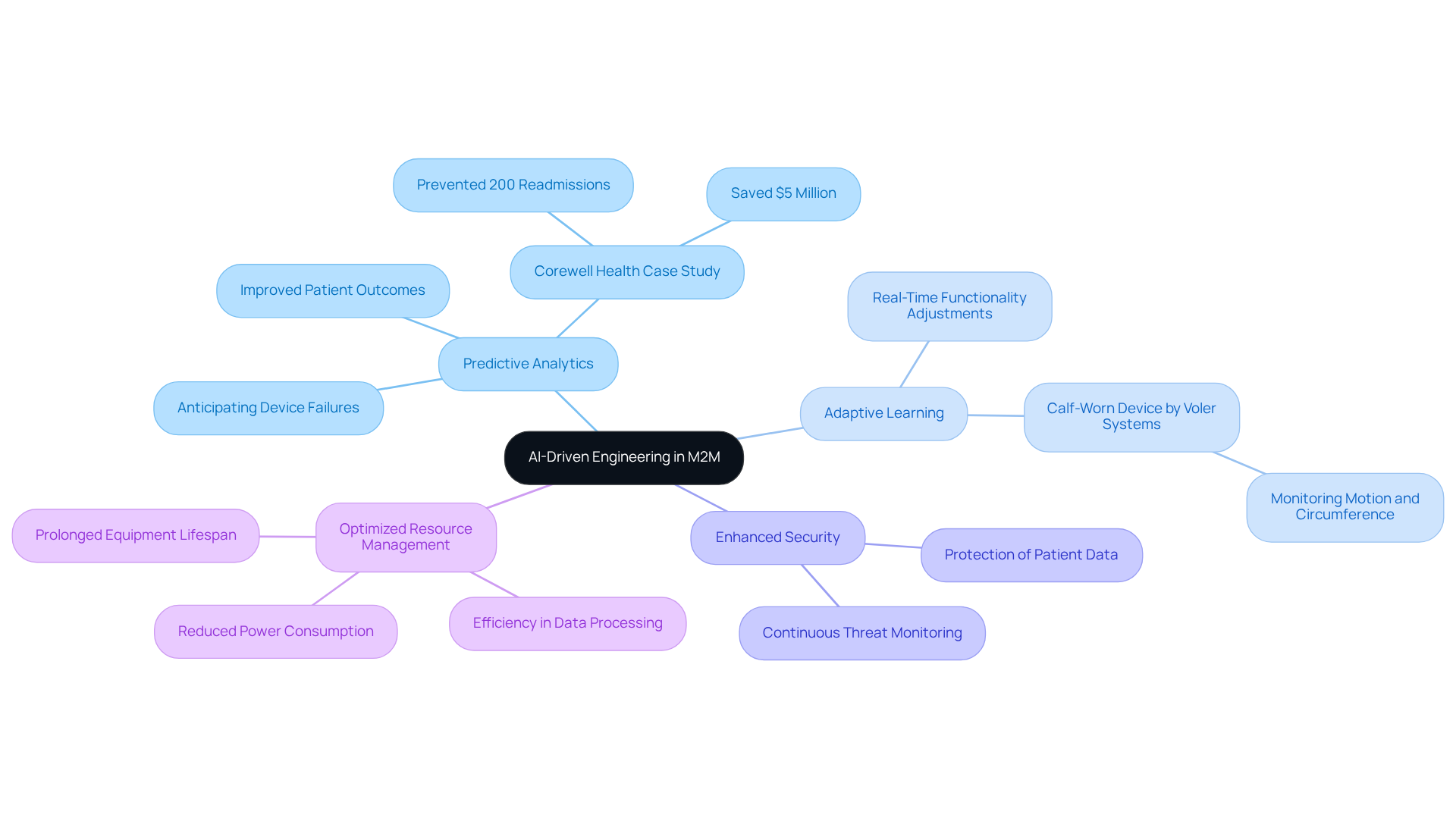
is transforming in medical devices through several key advancements:
- : Leveraging AI algorithms to analyze data patterns allows manufacturers to anticipate device failures or potential patient health issues, facilitating . This capability is crucial, as significantly enhances healthcare equipment performance, leading to improved outcomes for patients and reduced operational costs. For example, Corewell Health successfully prevented 200 readmissions and saved $5 million through , illustrating the practical application of AI in optimizing resource management.
- Adaptive Learning: AI systems with can evolve based on user interactions and environmental changes. This adaptability enables medical devices, such as the innovative calf-worn device developed by for monitoring motion and circumference during knee replacement rehabilitation, to adjust their functionalities in real-time, ensuring they meet the changing needs of patients and healthcare providers.
- : The integration of AI strengthens security protocols by continuously monitoring for potential threats and responding in real-time. This is particularly vital in the healthcare sector, where information breaches can compromise patient safety and privacy. AI's role in bolstering security measures is essential, especially in light of increasing concerns regarding privacy in healthcare.
- : AI enhances data processing efficiency and reduces power consumption, which is especially beneficial for . This optimization not only prolongs the lifespan of equipment but also .
By incorporating AI into M2M systems, the application of M2M allows manufacturers like to create smarter, more efficient medical devices that enhance patient care and drive operational excellence. Including testimonials and success stories can further illustrate the impact of these innovations.
Conclusion
The integration of Machine-to-Machine (M2M) communication in medical devices signifies a pivotal advancement in enhancing patient care and operational efficiency within healthcare. By leveraging this technology, healthcare providers can facilitate seamless data exchange, improve monitoring capabilities, and ultimately achieve superior patient outcomes. Understanding the fundamentals of M2M, along with regulatory compliance, rigorous testing, and the incorporation of artificial intelligence, is essential for the successful application of this innovative approach.
Key insights from the discussion underscore the necessity of:
- Mastering data transmission protocols and connectivity options
- Ensuring adherence to regulatory standards such as FDA and ISO
- Implementing robust testing methodologies to validate device performance
Furthermore, the role of AI in enhancing predictive analytics, adaptive learning, and security is critical, as these advancements significantly bolster the overall effectiveness of M2M solutions in medical devices.
As the healthcare landscape continues to evolve, embracing best practices for M2M technology is vital for both manufacturers and providers. By concentrating on these practices, stakeholders can not only fulfill regulatory requirements but also foster innovation that enhances patient safety and care quality. The future of healthcare relies on the successful integration of M2M communication, making it imperative for industry professionals to remain informed and proactive in adopting these advancements.
Frequently Asked Questions
What is M2M communication in the context of medical devices?
M2M communication refers to the automated transfer of information between medical devices without human intervention, enhancing monitoring capabilities and operational efficiency in healthcare.
Why is M2M communication important for patient outcomes?
M2M communication allows for seamless information exchange among devices, sensors, and healthcare systems, which is crucial for improving patient outcomes.
What are the key components of M2M communication?
The key components include data transmission protocols, connectivity options, and immediate information processing capabilities.
What data transmission protocols are used in M2M communication?
Essential data transmission protocols include MQTT, CoAP, and HTTP, which ensure reliable data exchange and safeguard the integrity and security of transmitted data.
What connectivity options are available for medical devices?
Medical devices can use wired connections like USB and wireless technologies such as Wi-Fi, Bluetooth, and cellular networks, affecting their performance and integration with healthcare infrastructures.
Why is immediate information processing important in M2M systems?
Immediate information processing is crucial for delivering timely insights in healthcare, particularly for managing chronic conditions where daily data can inform treatment adjustments.
How can engineers benefit from understanding M2M communication fundamentals?
By mastering these fundamentals, engineers can develop healthcare devices that comply with regulatory standards and leverage enhanced connectivity and data utilization to improve patient outcomes.
What role does artificial intelligence play in M2M communication for healthcare?
The integration of artificial intelligence in healthcare devices is revolutionizing the field, enabling more intelligent and responsive solutions to enhance patient care.
How will M2M communication shape the future of patient care?
As the healthcare sector evolves, the application of M2M communication will be instrumental in shaping the future of patient care through improved connectivity and data utilization.
List of Sources
- Understand M2M Communication Fundamentals in Medical Devices
- 12 Best Quotes on the Future of Healthcare (https://klara.com/blog/12-best-quotes-on-the-future-of-healthcare)
- Healthcare: Medical Device Manufacturer Archives - IoT M2M Council (https://iotm2mcouncil.org/iot-library/case-studies/healthcare-medical-device-manufacturer)
- 6 Important Quotes from Medtech Leaders (https://mddionline.com/business/6-important-quotes-from-medtech-leaders)
- M2M Healthcare Market Size, Share & Growth Report, 2034 (https://marketdataforecast.com/market-reports/global-m2m-healthcare-market)
- deliberatedirections.com (https://deliberatedirections.com/quotes-future-of-healthcare)
- Ensure Regulatory Compliance for M2M Integration
- How ISO 13485 Boosts Medical Device Quality - Dot Compliance (https://dotcompliance.com/blog/medical-device-manufacturing/how-iso-13485-boosts-medical-device-quality)
- FDA's AI Medical Device List: Stats, Trends & Regulation | IntuitionLabs (https://intuitionlabs.ai/articles/fda-ai-medical-device-tracker)
- FDA’s 2026 Guidance Expands Pathway for Low-Risk Digital Health Products—But Caution Remains Essential | Berkley Lifesciences (https://berkleyls.com/blog/fdas-2026-guidance-expands-pathway-low-risk-digital-health-products-caution-remains-essential)
- Duane Morris LLP - FDA Adopts ISO Standard for Device Makers in New Rule (https://duanemorris.com/alerts/fda_adopts_iso_standard_device_makers_new_rule_0324.html)
- #iso13485 #medicaldevices #qualitymanagement #qms #regulatoryaffairs #medtech #qualityassurance | Uma Gaur, PhD, RAC (US, Europe, Canada) | 14 comments (https://linkedin.com/posts/uma-gaur-phd-rac-us-europe-canada-03334629_iso13485-medicaldevices-qualitymanagement-activity-7389968813863231488-e4Mj)
- Implement Robust Testing and Validation for M2M Solutions
- 6 Important Quotes from Medtech Leaders (https://mddionline.com/business/6-important-quotes-from-medtech-leaders)
- Statistical Methods are also important for Medical Devices - ECA Academy (https://gmp-compliance.org/gmp-news/statistical-methods-are-also-important-for-medical-devices)
- Medical Device Validation & Verification Market Report, 2030 (https://grandviewresearch.com/industry-analysis/medical-device-verification-validation-market)
- Understanding Medical Device Validation Testing Stages (https://highpowervtls.com/2025/12/understanding-medical-device-validation-testing-stages)
- 7 quotes from 2023 to guide you on the medtech market (https://tiinatyni.com/blogi/7-quotes-from-2023-to-guide-you-on-the-medtech-market)
- Leverage AI-Driven Engineering for Enhanced M2M Performance
- 9 notable quotes from 1 big day at an epicenter of AI in healthcare – RamaOnHealthcare (https://ramaonhealthcare.com/9-notable-quotes-from-1-big-day-at-an-epicenter-of-ai-in-healthcare)
- AI in Healthcare 2025 Statistics: Market Size, Adoption, Impact (https://ventionteams.com/healthtech/ai/statistics)
- Impact of Predictive Analytics in Healthcare: Revolutionizing Patient Care (https://intone.com/impact-of-predictive-analytics-in-healthcare-revolutionizing-patient-care)
- How AI Agents and Tech Will Transform Health Care in 2026 (https://bcg.com/publications/2026/how-ai-agents-will-transform-health-care)





