10 Essential Elements of Product Design for Medical Devices
Introduction The complex realm of medical device design integrates innovation, precision,...

Mixed signal design stands as a cornerstone of modern medical devices, integrating analog and digital components to enhance both functionality and performance. By mastering the fundamentals of this intricate field, engineers can achieve significant benefits, including improved signal integrity and adherence to stringent regulatory standards. However, the journey presents numerous challenges.
How can teams effectively navigate the complexities of mixed signal design while fostering innovation and collaboration? This article explores best practices that not only address these challenges but also enhance the reliability and effectiveness of healthcare technology.
serves as a crucial element in the development of by integrating both analog and digital inputs within a single system. To effectively create mixed analog and digital circuits, engineers must grasp essential principles such as , signal integrity, and . Key practices include:
By mastering these fundamentals, engineers can develop robust systems through that meet the stringent requirements of the healthcare sector, ultimately enhancing the reliability and effectiveness of . has pioneered a range of innovative health products, including wearable sensors and heart pumps, demonstrating how and can lead to improved performance. Moreover, the importance of test systems in ensuring quality and reliability in electronic equipment development is underscored by case studies such as 'Resolving EMI and PCB Routing Issues in a Medical Optical Scanner.
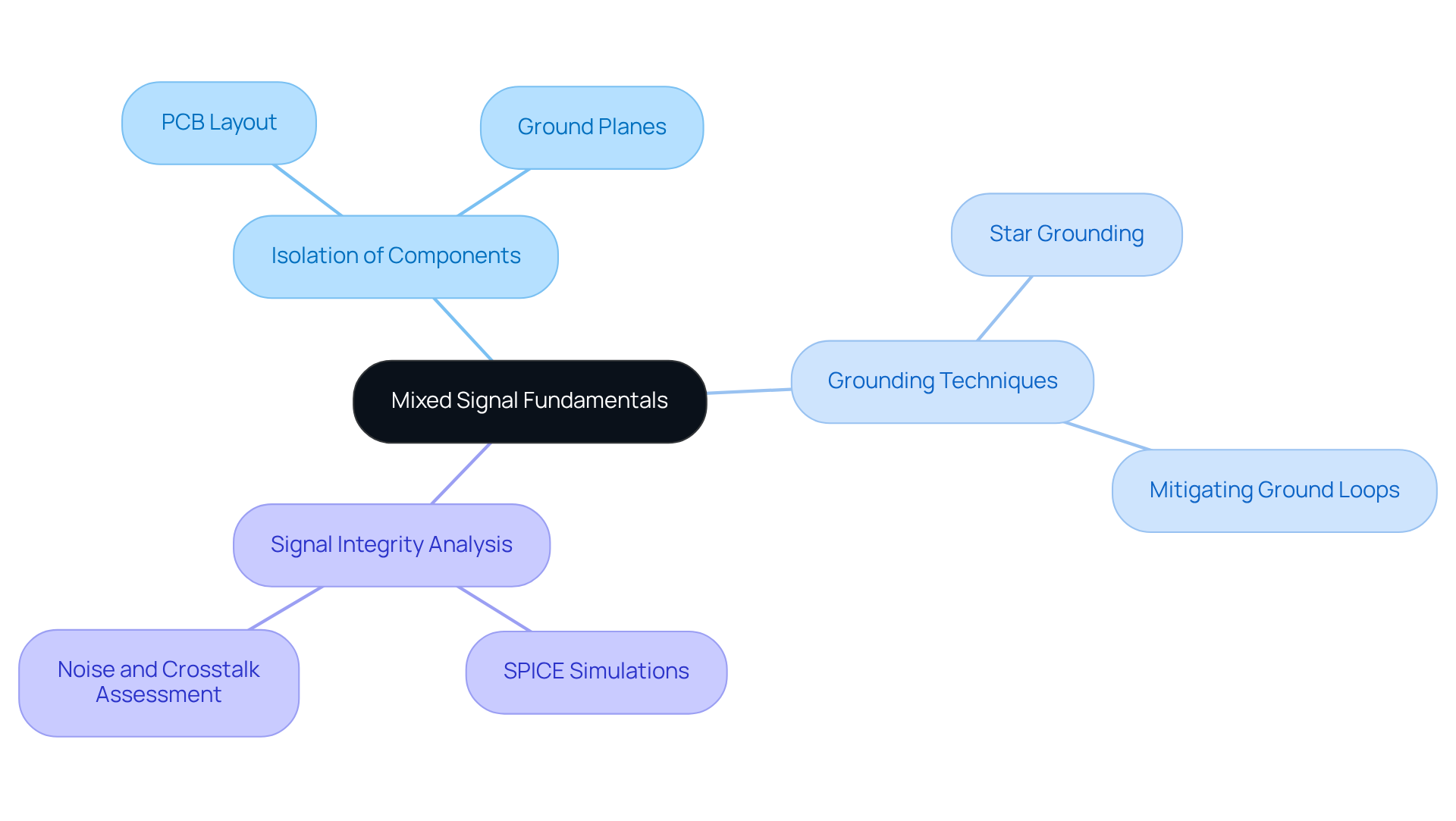
To ensure that s in comply with , engineers should adopt the following practices:
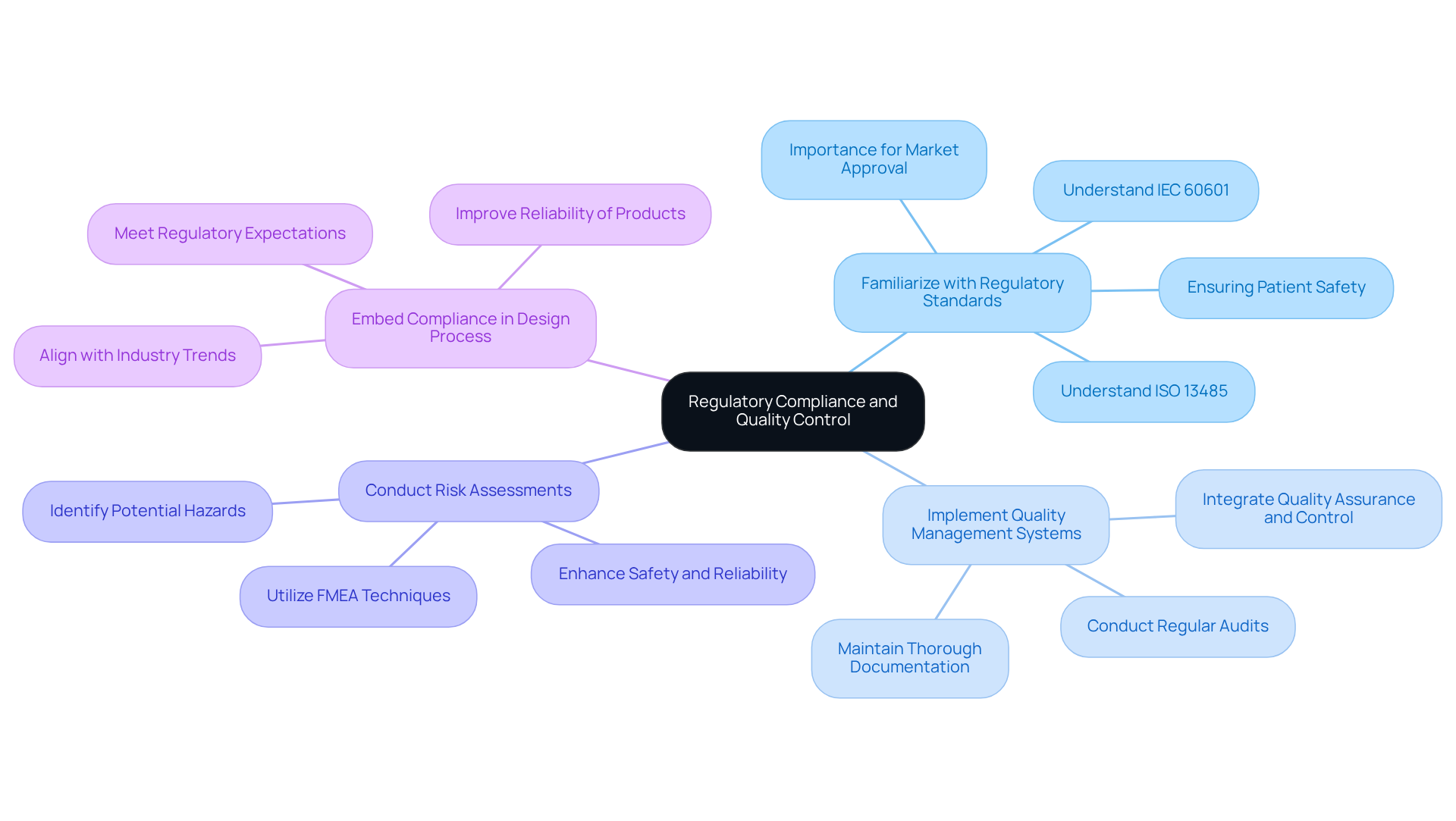
has the potential to significantly enhance in medical devices through several key approaches:
By integrating AI technologies, engineers can elevate their , leading to innovative solutions that address the evolving demands of the .
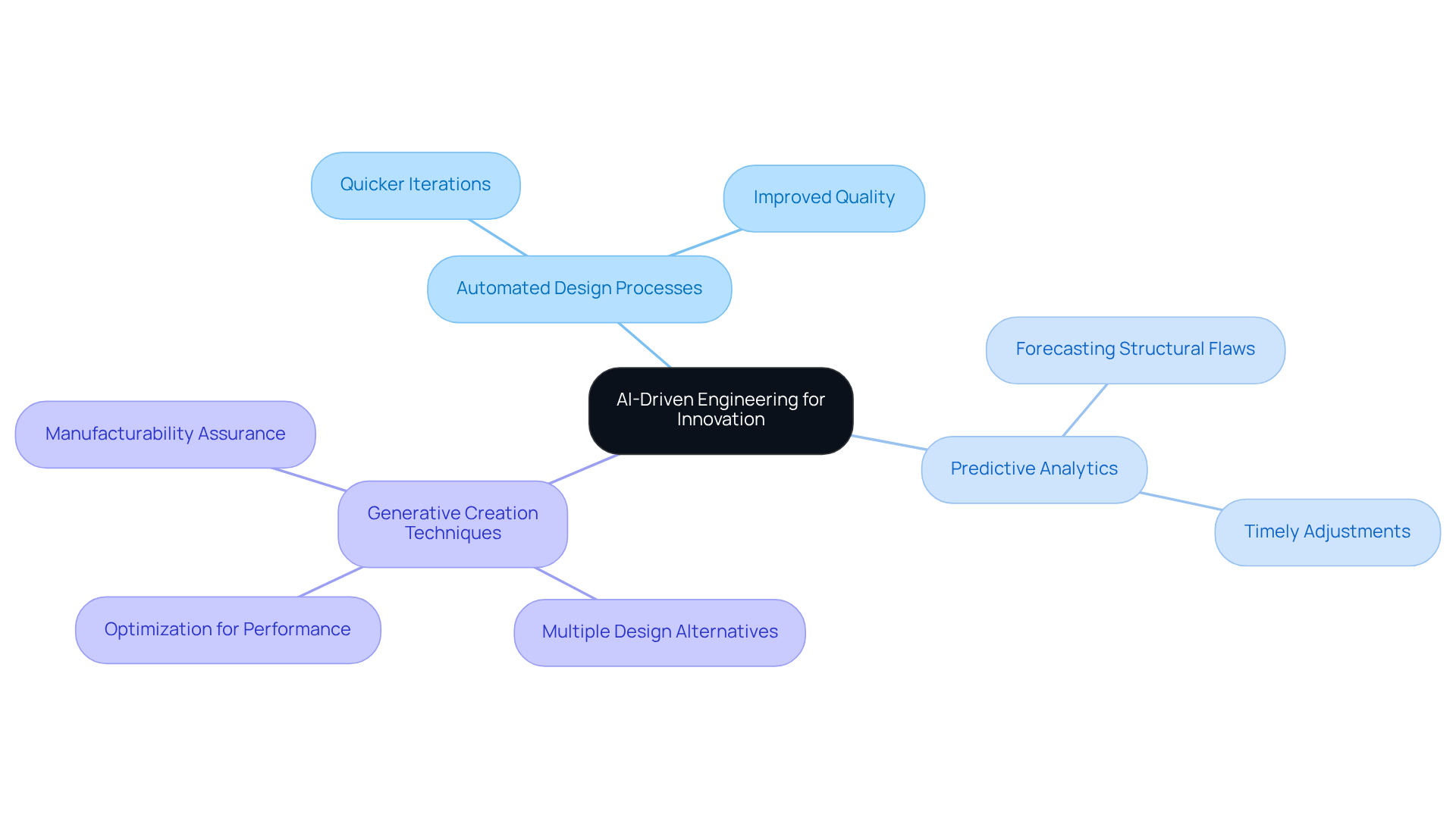
Successful cooperation among diverse teams is essential for enhancing the mixed communication creation process in . Implementing effective practices can significantly improve communication and :
By cultivating a , teams can harness their collective expertise to navigate challenges effectively and drive innovation in , ultimately leading to successful .
The integration of mixed signal design in medical devices marks a significant advancement in healthcare technology, effectively merging analog and digital systems to enhance both functionality and reliability. By mastering the fundamental principles of mixed signal design - such as effective noise management, grounding techniques, and the importance of regulatory compliance - engineers can develop devices that not only meet stringent industry standards but also improve patient outcomes.
Key practices include:
These strategies collectively contribute to the development of innovative healthcare solutions that address the complexities of modern medical requirements. Furthermore, fostering collaboration among cross-functional teams ensures a diverse range of insights and expertise, ultimately driving successful project outcomes.
As the healthcare landscape continues to evolve, embracing these best practices in mixed signal design will be crucial for engineers striving to advance medical device innovation. By prioritizing effective design techniques, regulatory adherence, and collaborative efforts, the industry can enhance the safety, reliability, and efficacy of medical devices, ultimately benefiting both healthcare providers and patients.
What is mixed signal design and why is it important in healthcare equipment?
Mixed signal design integrates both analog and digital inputs within a single system, which is crucial for developing healthcare equipment that requires both types of signals to function effectively.
What are some essential principles engineers must understand for mixed signal design?
Engineers must grasp principles such as noise management, signal integrity, and grounding techniques to create effective mixed analog and digital circuits.
How can engineers minimize interference between analog and digital components?
Engineers can minimize interference by physically separating analog and digital circuits, which can be achieved through careful PCB layout and the use of specific ground planes.
What grounding technique is recommended for mixed signal design in healthcare applications?
A star grounding scheme is recommended as it helps mitigate ground loops, ensuring stable operation, which is critical for maintaining communication quality in healthcare equipment.
Why is signal integrity analysis important in mixed signal design?
Signal integrity analysis is important because it allows engineers to perform simulations that assess the impact of noise and crosstalk on circuit performance, helping to predict and address potential issues.
What tools can engineers use for signal integrity analysis?
Tools such as SPICE are invaluable for performing simulations and predicting circuit behavior under various conditions.
How can effective mixed signal design enhance healthcare equipment?
By mastering mixed signal fundamentals, engineers can develop robust systems that meet the stringent requirements of the healthcare sector, ultimately improving the reliability and effectiveness of healthcare equipment.
What examples illustrate the application of mixed signal design in healthcare?
Voler Systems has pioneered innovative health products, such as wearable sensors and heart pumps, showcasing how effective noise management and grounding techniques can lead to improved performance.
What is the significance of test systems in electronic equipment development for healthcare?
Test systems are crucial for ensuring quality and reliability in electronic equipment development, as highlighted by case studies like 'Resolving EMI and PCB Routing Issues in a Medical Optical Scanner.
