Why Medical Device Manufacturers Need a Product Development Studio
Introduction The medical device industry is at a pivotal juncture, balancing innovation...

The rapid evolution of healthcare technology necessitates a new approach to medical device development, where connectivity and user-centric design are essential. By integrating structured processes and cross-functional teams, organizations can harness diverse expertise to improve product functionality and compliance.
However, with the increasing complexities of regulations and the imperative for continuous innovation, how can developers ensure that their connected medical devices not only meet stringent standards but also resonate with end-users?
This article explores best practices that empower teams to navigate these challenges and create impactful healthcare solutions.
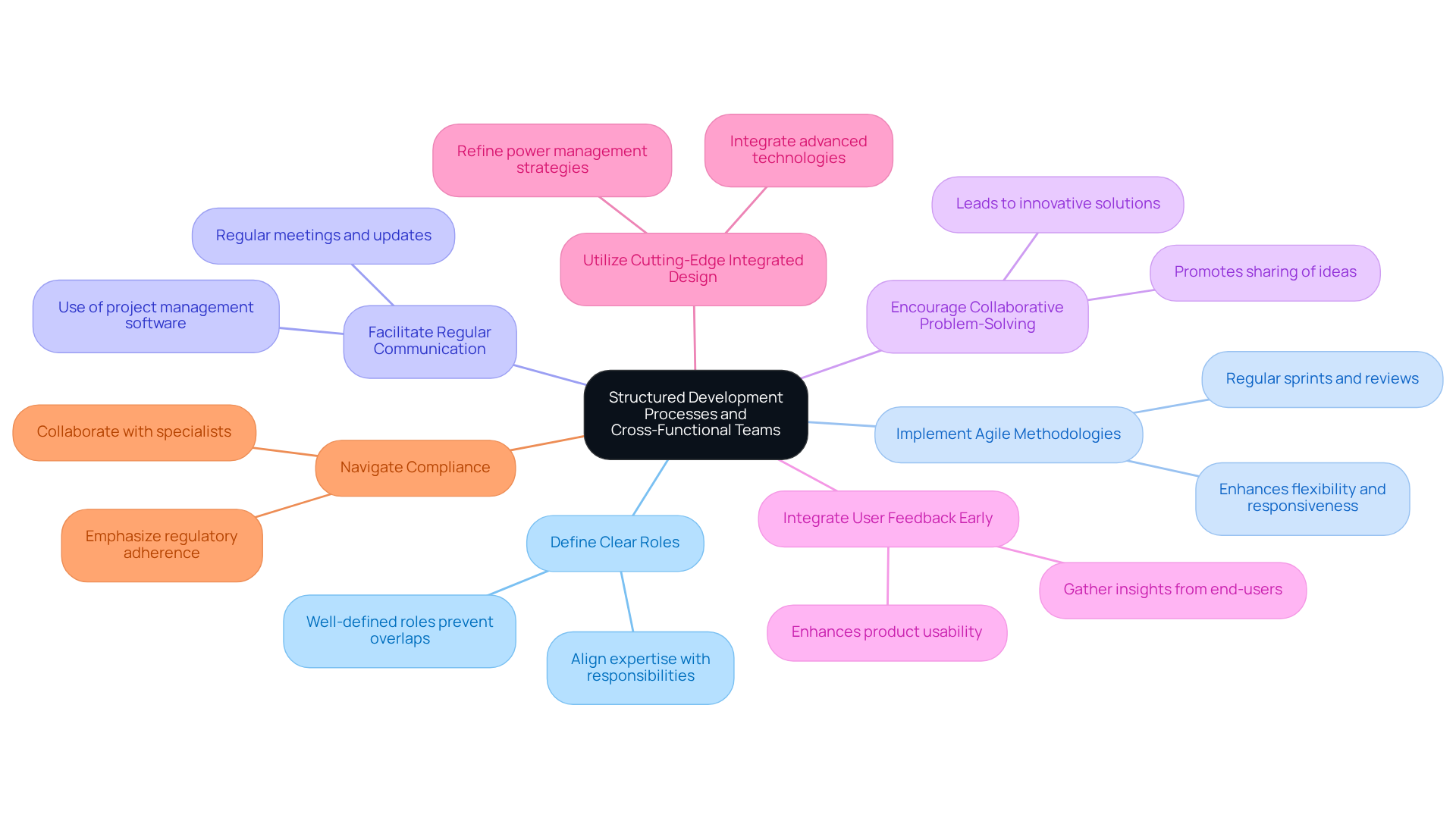
To effectively create interconnected health instruments, establishing organized development procedures that involve cross-functional teams is crucial. This approach ensures that all relevant expertise is integrated from the outset, including engineering, regulatory affairs, quality assurance, and clinical insights. Below are key practices to consider:
By adopting these practices, organizations can establish a robust framework for connected product development of interconnected health instruments that meet both user requirements and regulatory standards.
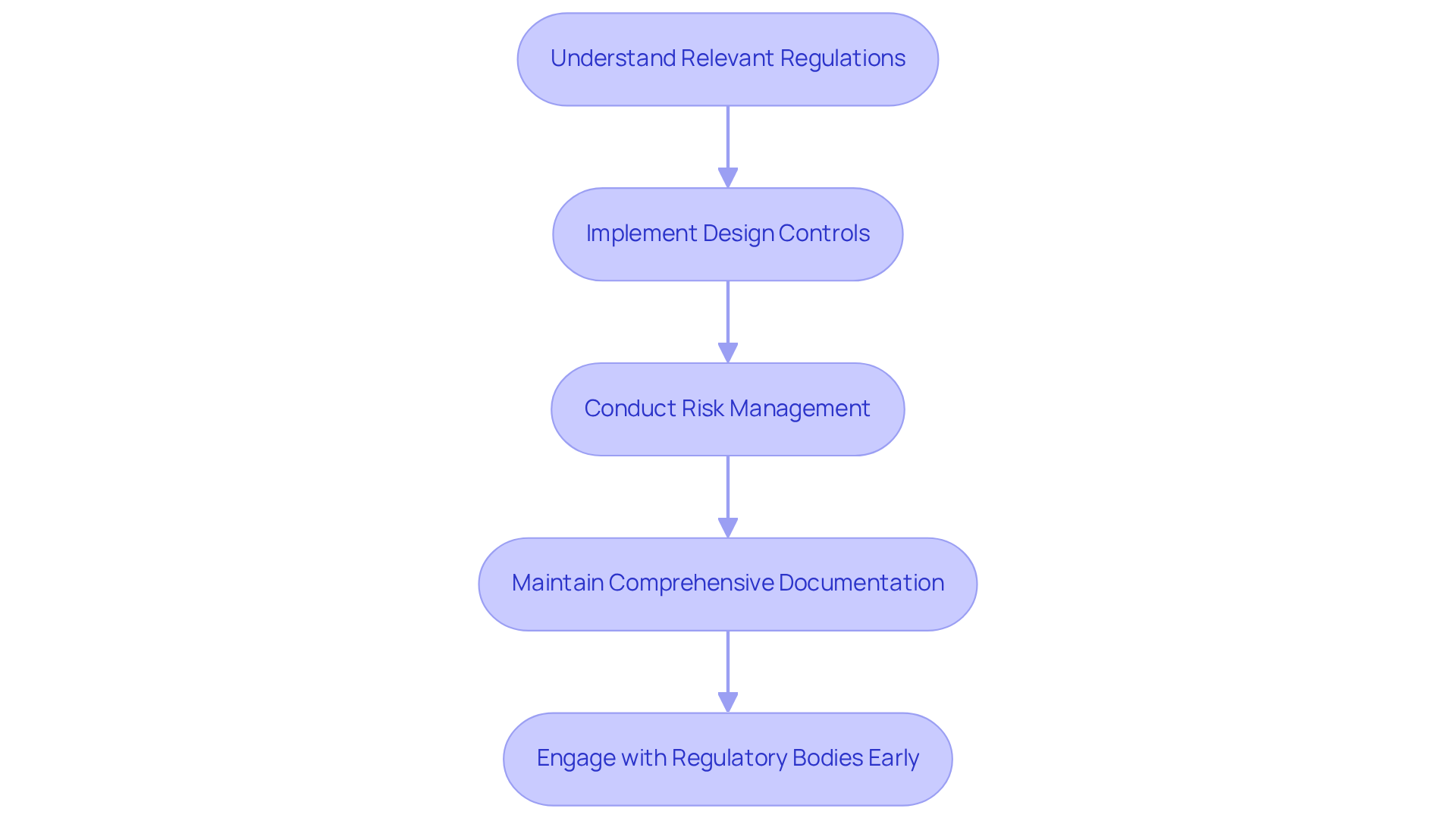
Regulatory adherence and quality assurance are essential components of medical product development. The company is dedicated to ensuring that its offerings meet the necessary safety and effectiveness standards. To enhance compliance and product quality, consider the following best practices:
By integrating these practices into the design process, organizations can improve product quality and ensure compliance, ultimately leading to successful market entry.
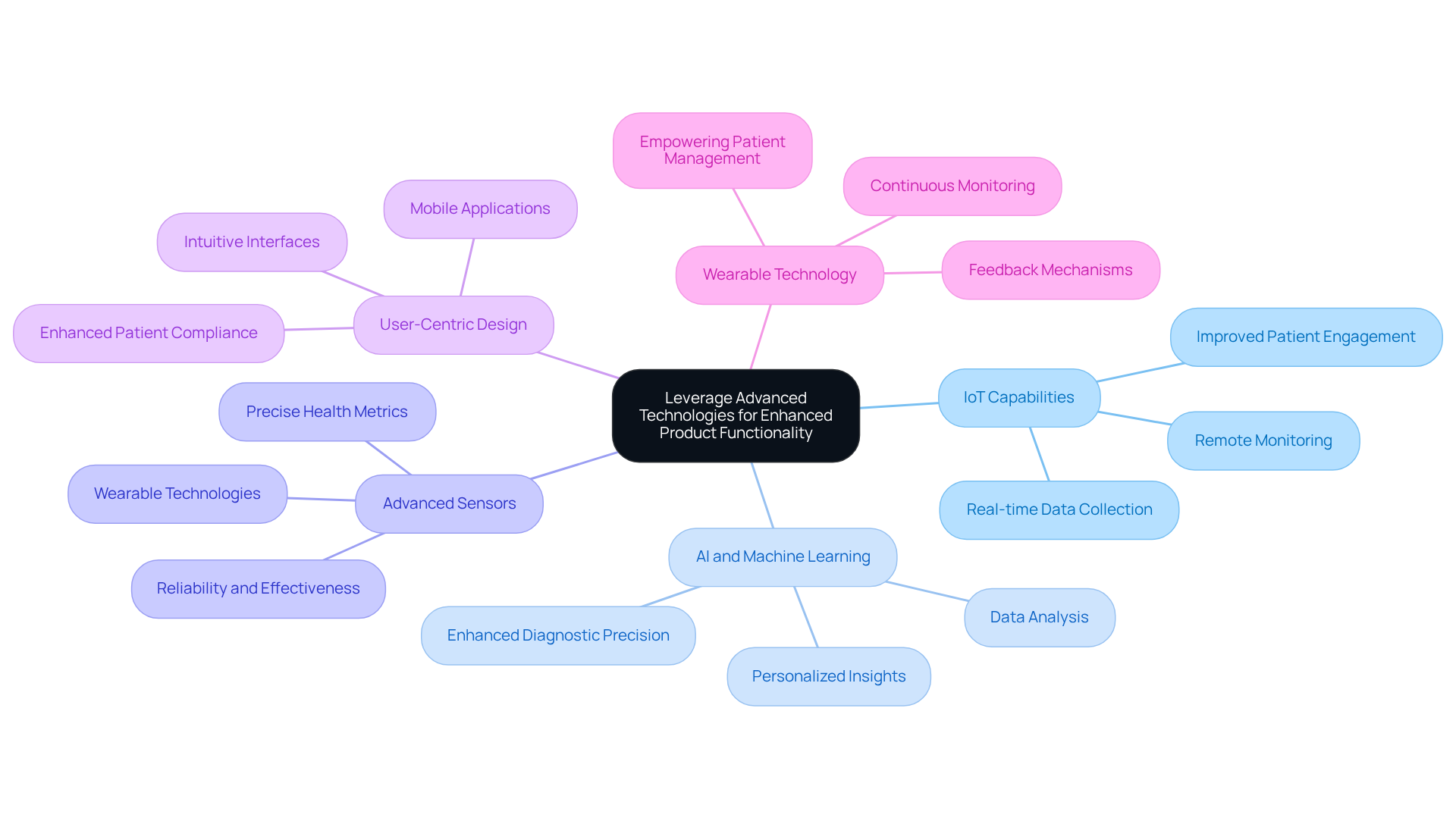
Integrating advanced technologies into healthcare equipment development can significantly enhance product functionality and user experience. The following best practices outline how to effectively leverage these technologies:
By adopting these advanced technologies, organizations can create innovative healthcare instruments that meet the evolving needs of patients and providers.
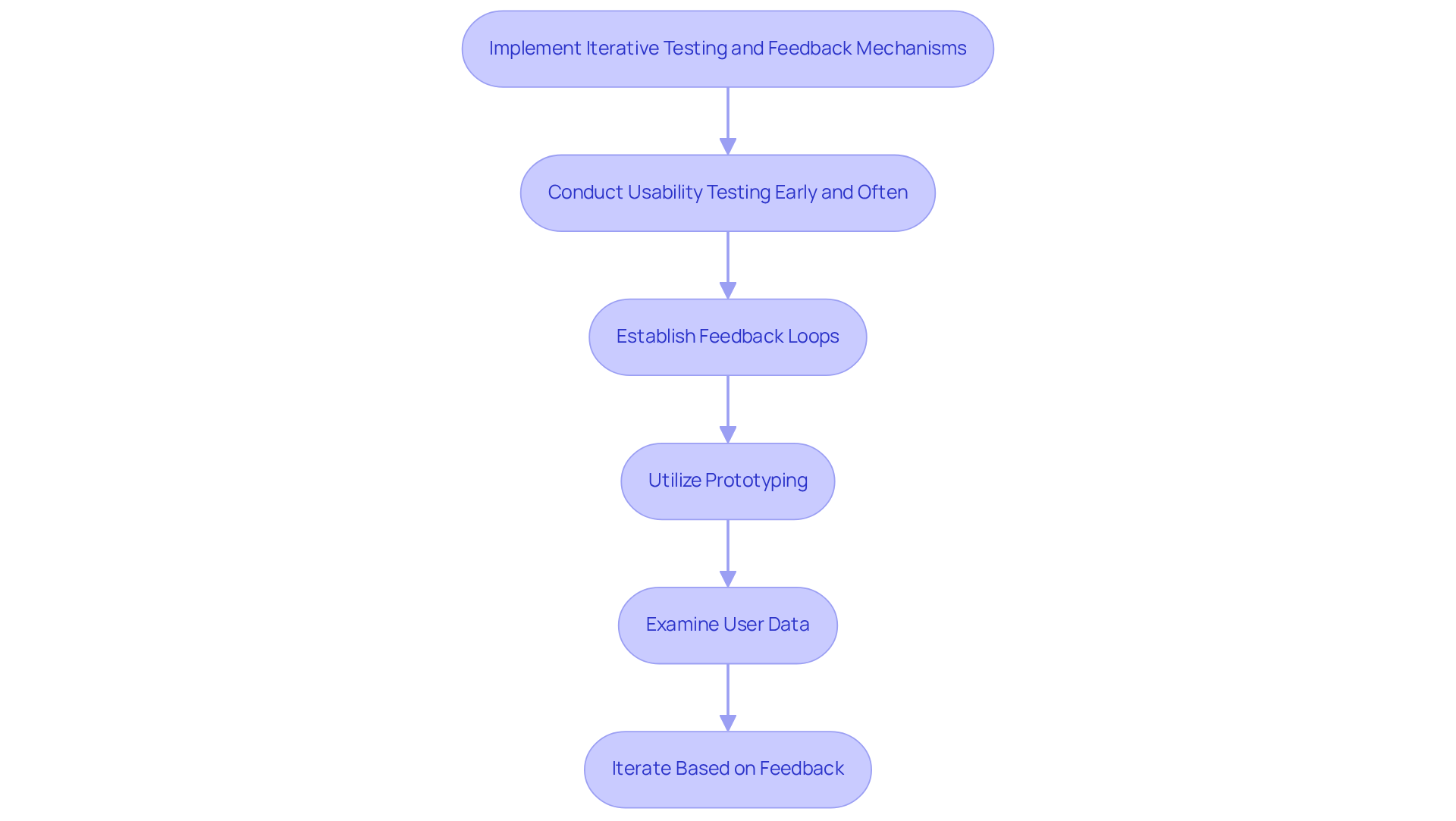
Iterative testing and feedback systems are crucial for refining healthcare tools and ensuring alignment with user expectations. Here are some best practices to implement:
By implementing these iterative testing and feedback mechanisms, organizations can enhance the effectiveness and user satisfaction of their medical devices.
Establishing effective practices for connected product development in medical devices is crucial for creating innovative and compliant healthcare solutions. By prioritizing structured development processes and leveraging cross-functional teams, organizations can ensure that every aspect of the product lifecycle is addressed comprehensively. This approach not only enhances collaboration but also cultivates an environment where innovative ideas can thrive, resulting in devices that genuinely meet user needs.
Key insights presented in this article underscore the importance of:
Additionally, navigating regulatory compliance and utilizing advanced technologies such as IoT and AI can significantly enhance product functionality and ensure quality control. Each of these components contributes to a robust framework that supports successful market entry and user satisfaction.
The significance of these practices is paramount. As the medical device industry continues to evolve, adopting these strategies will empower organizations to develop connected products that not only adhere to stringent regulations but also enhance patient outcomes. It is imperative for stakeholders to act on these insights, fostering a culture of innovation and collaboration that will ultimately propel the future of healthcare technology.
Why is it important to establish structured development processes for health instruments?
Establishing structured development processes ensures that all relevant expertise, including engineering, regulatory affairs, quality assurance, and clinical insights, is integrated from the outset, leading to effective interconnected health instruments.
What are the key practices for creating effective cross-functional teams?
Key practices include defining clear roles and responsibilities, implementing agile methodologies, facilitating regular communication, encouraging collaborative problem-solving, integrating user feedback early, utilizing cutting-edge integrated design, and navigating compliance from prototype to production.
How can defining clear roles and responsibilities benefit a project team?
Clear roles and responsibilities prevent overlaps and gaps in responsibilities, ensuring comprehensive coverage of all project aspects and enhancing team efficiency.
What is the role of agile methodologies in development processes?
Agile methodologies enhance flexibility and responsiveness to changes, allowing teams to adapt quickly to new information or regulatory updates through regular sprints and reviews.
Why is regular communication important in project development?
Regular communication through meetings and updates ensures that all team members are aligned on project goals and timelines, facilitating effective collaboration and progress tracking.
How does encouraging collaborative problem-solving contribute to project success?
Fostering a collaborative environment allows team members to share ideas and challenges, leading to innovative solutions and quicker problem resolution.
What is the significance of integrating user feedback early in the development process?
Involving end-users early allows for gathering insights that inform design decisions, enhancing product usability and aligning the product more closely with market needs.
How does utilizing cutting-edge integrated design improve healthcare equipment?
Integrating advanced technologies like FPGA and AI improves functionality and efficiency, enhancing battery life and performance, which are essential for wireless healthcare tools.
What should be emphasized in the development process for wearable health products?
The development process should emphasize regulatory adherence, and collaborating with specialists can help manage documentation and regulatory challenges effectively.
What is the overall benefit of adopting these structured practices in health instrument development?
Adopting these practices establishes a robust framework for connected product development that meets both user requirements and regulatory standards.
