4 Best Practices for Effective PCB Layout Design in Medical Devices
Discover best practices for effective PCB layout design in medical devices to ensure...

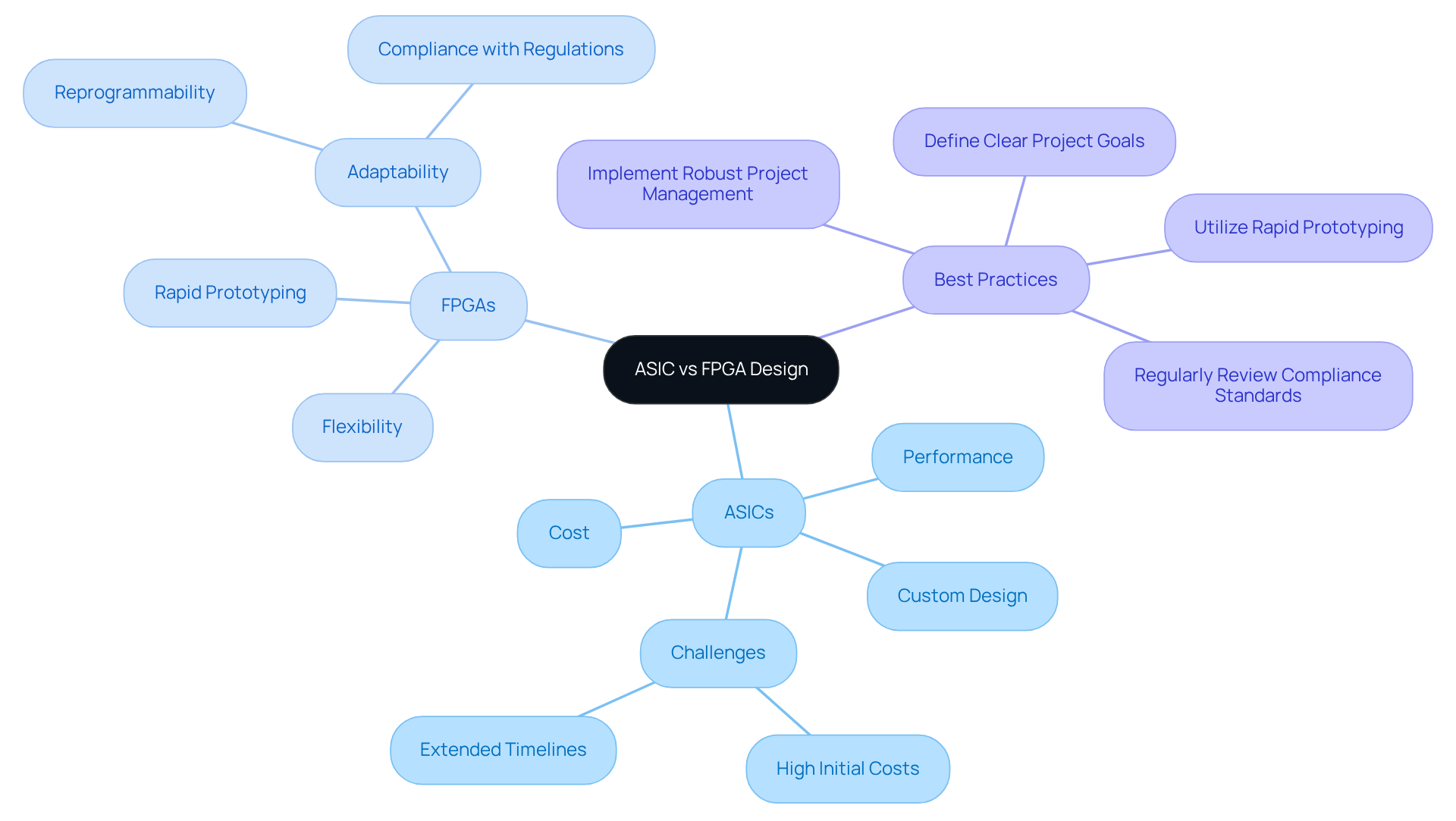
In the medical device industry, the decision between Application-Specific Integrated Circuits (ASICs) and Field-Programmable Gate Arrays (FPGAs) presents a critical challenge that can impact both innovation and compliance. Each technology offers distinct advantages and challenges that significantly influence design efficiency, regulatory compliance, and ultimately, patient safety.
Manufacturers must carefully navigate the complexities of ASIC FPGA design, weighing best practices that optimize performance against the need for regulatory compliance. Engineers must find ways to balance innovation with compliance, employing strategies that lead to successful outcomes in this critical field.
In medical device development, understanding the distinctions between Application-Specific Integrated Circuits (ASICs) and Field-Programmable Gate Arrays (FPGAs) is essential for optimizing ASIC FPGA design and production processes. ASICs are custom-designed for specific applications, delivering exceptional performance and efficiency, especially in high-volume production settings. However, the high initial costs and extended timelines can pose challenges for manufacturers. Conversely, FPGAs offer remarkable flexibility and rapid prototyping capabilities, enabling design modifications even after deployment. This adaptability is crucial in healthcare, where regulatory standards and technology evolve quickly. For example, FPGAs can be reprogrammed to meet new regulations, making them vital for medical equipment engineers.
To ensure timely and budget-friendly delivery, manufacturers should follow these best practices:
By carefully evaluating the strengths of ASICs and FPGAs in ASIC FPGA design, manufacturers can strategically position themselves to meet both market demands and regulatory requirements.
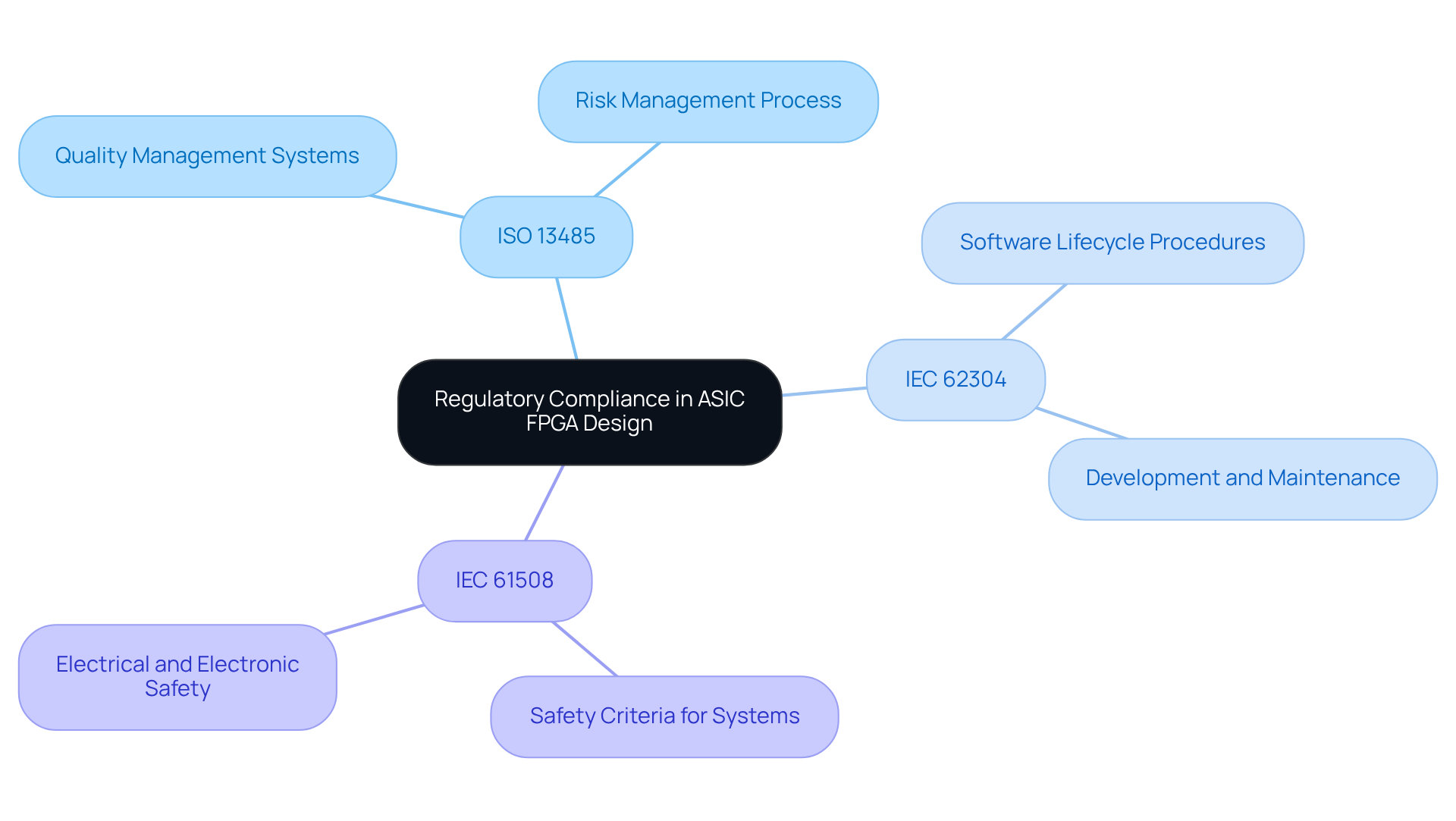
In the medical device sector, regulatory compliance is not just a requirement; it is a critical factor that influences product safety and market success. It is imperative for engineers to align their designs in ASIC FPGA design with the standards set forth by the FDA and ISO. Key regulations include:
Compliance streamlines approval processes and significantly mitigates risks associated with product safety. For instance, a healthcare apparatus utilizing an FPGA for real-time data processing must adhere to IEC 61508, which specifies safety criteria for electrical, electronic, and programmable electronic safety-related systems.
By integrating compliance factors from the outset of the development process, organizations can avoid costly revisions and operational delays. This proactive approach not only reduces risks but also expedites their product's time-to-market and enhances product reliability. Furthermore, adherence to ISO 13485 has been shown to improve overall quality management, with organizations reporting significant enhancements in operational efficiency and customer satisfaction. Ultimately, organizations that prioritize compliance are better positioned to achieve operational excellence and meet customer expectations.
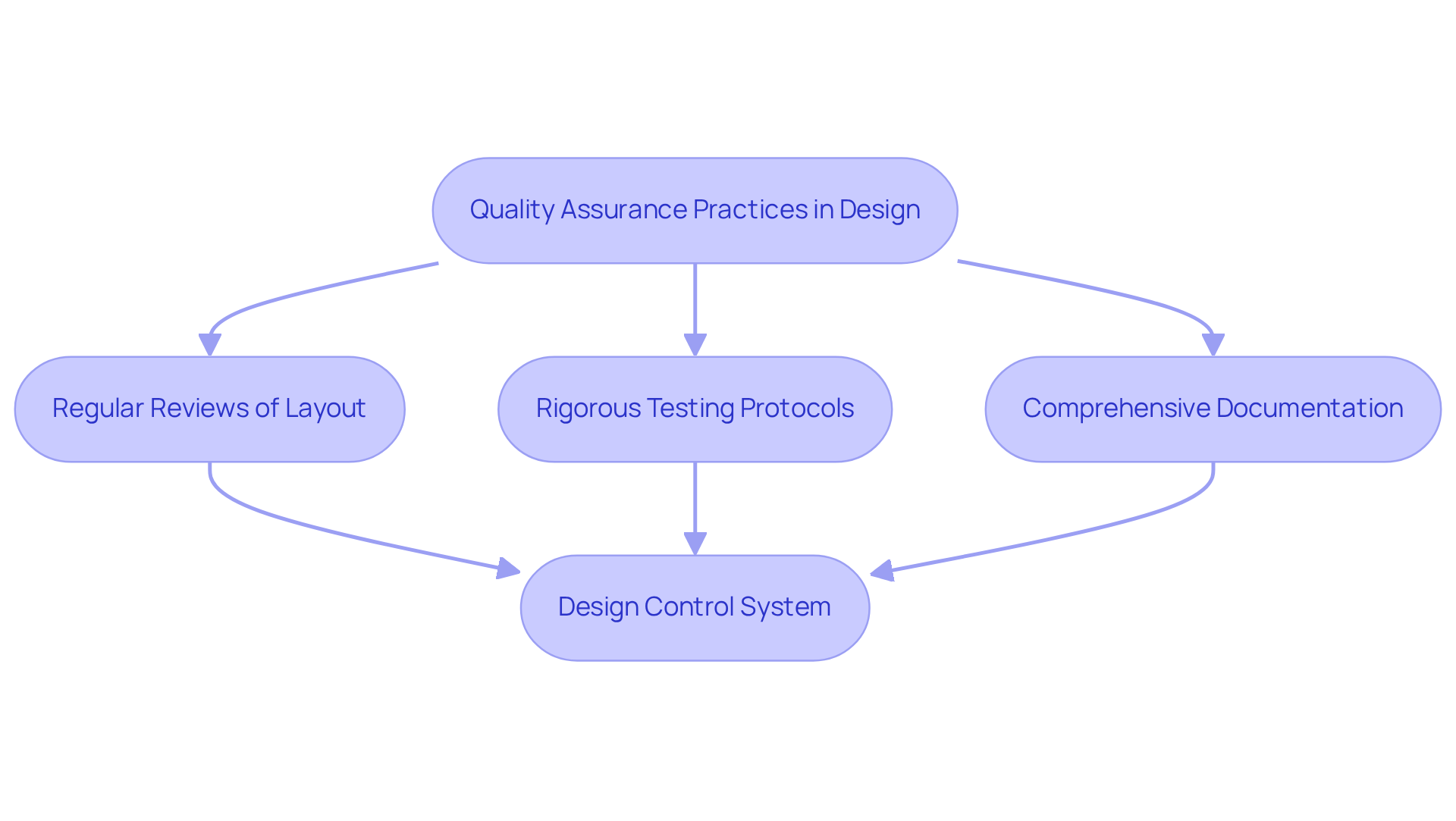
Quality assurance (QA) is a critical component in the development of medical devices, directly impacting safety and regulatory compliance. A structured approach to QA includes:
A Design Control system is crucial for tracking requirements throughout the development process, ensuring compliance with all specifications. For instance, a case study on a wearable health apparatus created by Voler Systems showed that early incorporation of QA practices, such as risk management and design verification, decreased design iterations by 30%, significantly accelerating development timelines.
Furthermore, the use of automated testing tools can simplify verification methods, enabling quicker defect identification. By fostering a proactive QA culture, teams can enhance product reliability and improve patient safety, ultimately resulting in more successful outcomes in the competitive healthcare equipment market.
The FDA's upcoming Quality Management System Regulation (QMSR), effective from February 2, 2026, underscores the imperative for rigorous QA practices, reinforcing the industry's commitment to excellence.
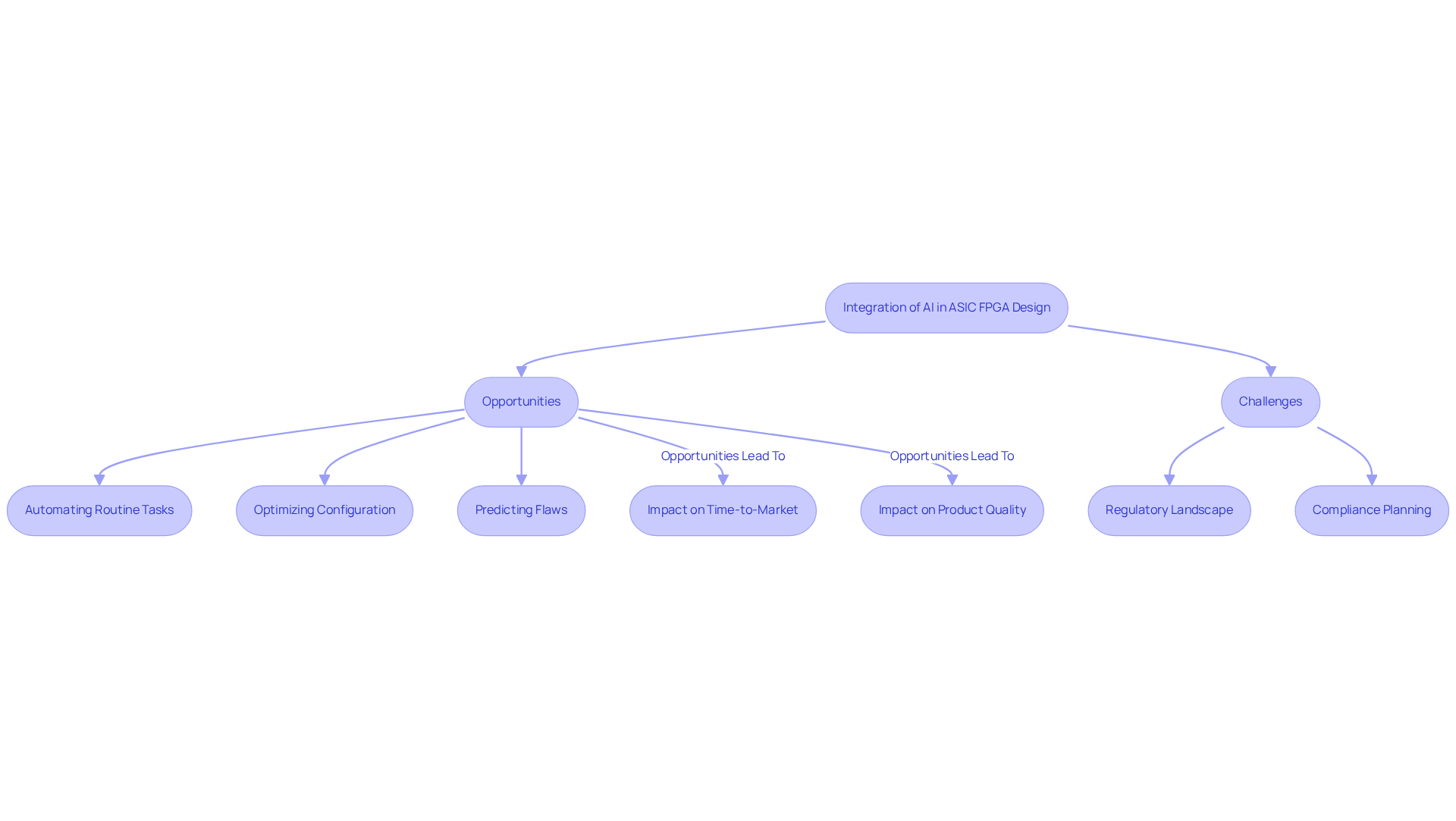
The integration of artificial intelligence (AI) into ASIC FPGA design processes presents both opportunities and challenges for innovation in medical devices. AI plays a crucial role by automating routine tasks, optimizing configuration parameters, and predicting potential flaws prior to their manifestation. For instance, machine learning algorithms can analyze historical development data to uncover patterns that correlate with successful outcomes, empowering engineers to make informed, data-driven decisions. This capability can reduce time-to-market by 30-50% for startups.
A compelling case is the application of AI in a medical imaging device, where predictive analytics shortened the development cycle by 25%, facilitating quicker iterations and elevating product quality. By embracing these advanced technologies, engineers not only improve their creative abilities but also ensure their products stay competitive in a constantly changing market.
However, navigating the regulatory landscape remains a significant challenge for many developers. Moreover, integrating regulatory-ready planning principles from the outset is vital, as it assists in tackling compliance challenges and guarantees that products fulfill essential safety and effectiveness criteria.
Voler Systems' expertise in documentation compliance support can guide medical device manufacturers through these regulatory complexities. As Naveen Shukla noted, aligning design decisions with compliance standards early can reduce redesign risks and accelerate approvals, ultimately benefiting the entire development process.
Ultimately, the strategic incorporation of AI and regulatory planning can redefine success in the medical device industry.
In the competitive landscape of medical device development, integrating ASIC and FPGA design principles effectively is essential for innovation and compliance. Understanding the unique characteristics of ASIC and FPGA technologies allows engineers to leverage their strengths, enhancing performance and regulatory compliance, which ultimately leads to safer medical devices.
This article presents several best practices critical for optimizing ASIC and FPGA design. To optimize ASIC and FPGA design, key strategies include:
These practices streamline development and mitigate risks associated with product safety and market readiness. Incorporating AI enhances design efficiency, allowing teams to make informed decisions that speed up time-to-market while ensuring compliance with changing regulations.
In an evolving medical device industry, embracing these best practices is vital for manufacturers to maintain a competitive edge. The integration of advanced technologies and a proactive approach to regulatory compliance not only enhances product reliability but also drives innovation in healthcare solutions. By prioritizing these strategies, organizations can not only enhance their market position but also significantly improve patient outcomes in a dynamic healthcare environment.
What are ASICs and FPGAs in the context of medical device development?
ASICs (Application-Specific Integrated Circuits) are custom-designed chips optimized for specific applications, offering high performance and efficiency, particularly in high-volume production. FPGAs (Field-Programmable Gate Arrays) are flexible chips that can be programmed and reprogrammed, allowing for rapid prototyping and design modifications even after deployment.
What are the advantages of using ASICs?
ASICs provide exceptional performance and efficiency, especially in high-volume production settings, making them ideal for applications that require consistent, high-quality output.
What challenges are associated with ASICs?
The main challenges of ASICs include high initial costs and extended timelines for design and production, which can be a barrier for manufacturers.
What are the benefits of using FPGAs in medical device development?
FPGAs offer remarkable flexibility, rapid prototyping capabilities, and the ability to make design modifications after deployment. This adaptability is particularly important in healthcare, where regulatory standards and technologies evolve quickly.
How can FPGAs be beneficial in meeting regulatory requirements?
FPGAs can be reprogrammed to comply with new regulations, making them essential for medical equipment engineers who need to adapt to changing standards.
What best practices should manufacturers follow for ASIC and FPGA design?
Manufacturers should define clear project goals and requirements, utilize rapid prototyping with FPGAs, implement a robust project management framework to track progress and budget, and regularly review compliance standards and technology trends to adapt designs accordingly.
How can understanding the differences between ASICs and FPGAs help manufacturers?
By evaluating the strengths of ASICs and FPGAs, manufacturers can strategically position themselves to meet market demands and regulatory requirements effectively.
