Introduction
Navigating the complex landscape of electronic manufacturing services (EMS) in France is crucial for companies focused on producing medical devices. With significant market growth projected, understanding the key players and regulatory requirements is essential for fostering opportunities in innovation and collaboration. As the demand for advanced medical equipment continues to rise, manufacturers must carefully select the right EMS partner to meet their specific needs while ensuring compliance with stringent standards.
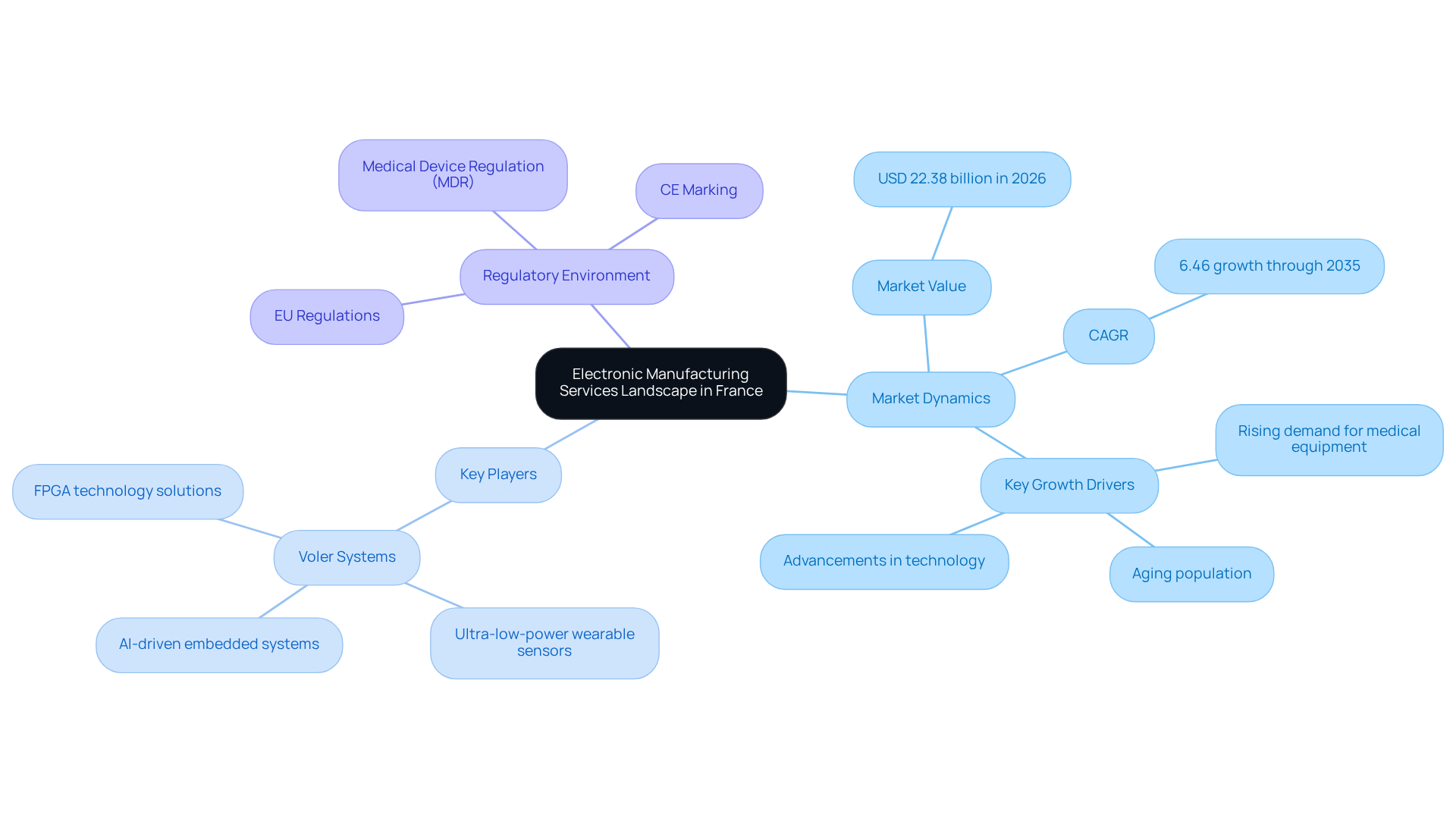
Understand the Electronic Manufacturing Services Landscape in France
To effectively navigate the landscape, understanding the current is essential. The market for was valued at approximately USD 22.38 billion in 2026 and is projected to grow at a compound annual growth rate (CAGR) of 6.46% through 2035. This growth is primarily driven by the rising demand for , particularly in relation to an aging population and advancements in technology.
Key players in the market include established companies that offer a range of services, from PCB assembly to system integration.
- distinguishes itself with its expertise in creating and AI-driven embedded systems, which are crucial for addressing environmental challenges in design.
- Additionally, specializes in , providing flexible and efficient solutions tailored to specific application needs.
Understanding these players and their capabilities can assist in identifying requirements.
Furthermore, it is important to familiarize oneself with the , as compliance with EU regulations is critical for market entry. This includes obtaining CE marking and adhering to the Medical Device Regulation (MDR).
Identify Your Medical Device Manufacturing Requirements
Before engaging with an (EMS) provider, it is essential to clearly define your specific manufacturing requirements. Begin by determining the type of medical apparatus you intend to create, considering its intended use, target market, and any distinctive features. Follow these key steps:
- : Understand the regulatory landscape for your product, including CE marking and adherence to the . The recent introduction of the in 2026 emphasizes the need for compliance with , which aligns U.S. regulations with international standards. This compliance will significantly influence your choice of provider.
- : Clearly outline the of your apparatus, including materials, dimensions, and performance criteria. This detailed information is crucial for of potential EMS partners and ensuring they can meet your specific needs.
- : Estimate the needed for your equipment. This estimation will influence the scalability and flexibility that your EMS supplier must offer, ensuring they can accommodate your production needs as they evolve.
- : Identify the your equipment must adhere to, such as ISO 13485. Ensuring that your EMS partner is capable of meeting these standards is vital for maintaining compliance and delivering a high-quality product.
By following these steps, you can effectively navigate the complexities of choosing an France provider that aligns with your production needs and regulatory obligations.

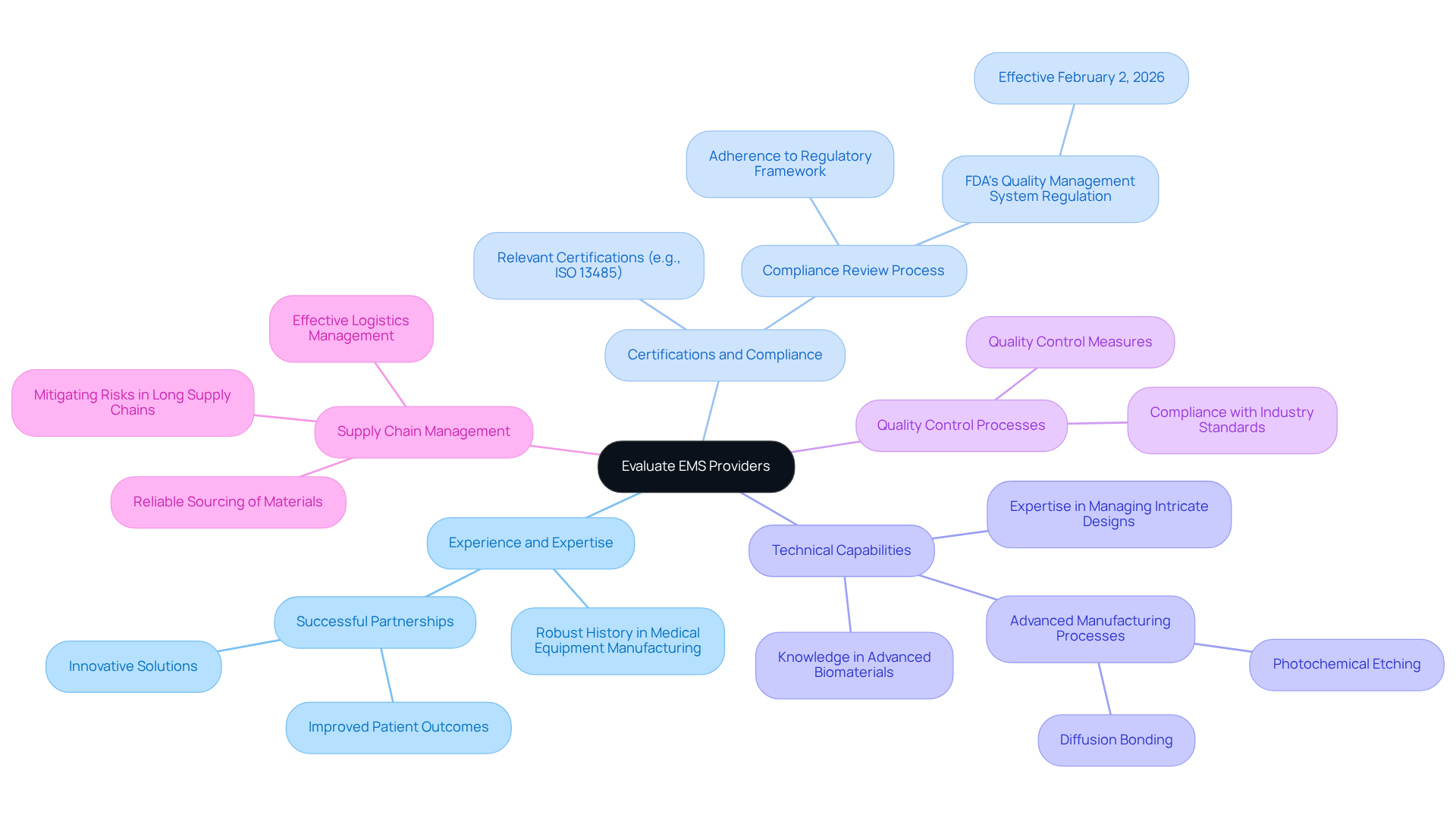
Evaluate Potential EMS Providers Based on Key Criteria
When evaluating potential EMS providers for medical device manufacturing, it is essential to consider several key criteria to ensure alignment with your specific needs:
- Experience and Expertise: Seek suppliers with a robust history in medical equipment manufacturing. in delivering quality products on time and within budget, as demonstrated by successful partnerships that have led to innovative solutions and improved patient outcomes.
- Certifications and Compliance: Verify that the supplier of possesses relevant certifications, such as , which is crucial for ensuring adherence to the regulatory framework governing medical equipment in France. Voler Systems has a that guarantees devices meet emissions and ESD standards, vital for maintaining , particularly with the FDA's Quality Management System Regulation (QMSR) set to take effect on February 2, 2026.
- Technical Capabilities: Assess the supplier's technical abilities, including their expertise in managing intricate designs, prototyping, and comprehensive testing. Advanced manufacturing processes, such as photochemical etching and diffusion bonding, are increasingly important for producing complex components with tight tolerances. Additionally, consider the specialist's knowledge in , which are central to enhancing device-tissue interactions and long-term performance.
- : Inquire about the quality control measures implemented by the supplier. Understanding how they maintain compliance with industry standards throughout the manufacturing process is critical for ensuring the reliability and safety of your devices. , ensuring that every project meets stringent industry standards.
- Supply Chain Management: Evaluate the EMS supplier's . Their ability to source materials reliably and manage logistics effectively is essential, especially in a landscape where reshoring and regionalization are becoming more prevalent to mitigate risks associated with long supply chains. As manufacturers face increasing complexity due to shifting patient expectations and heightened regulatory scrutiny, effective supply chain management becomes even more crucial.
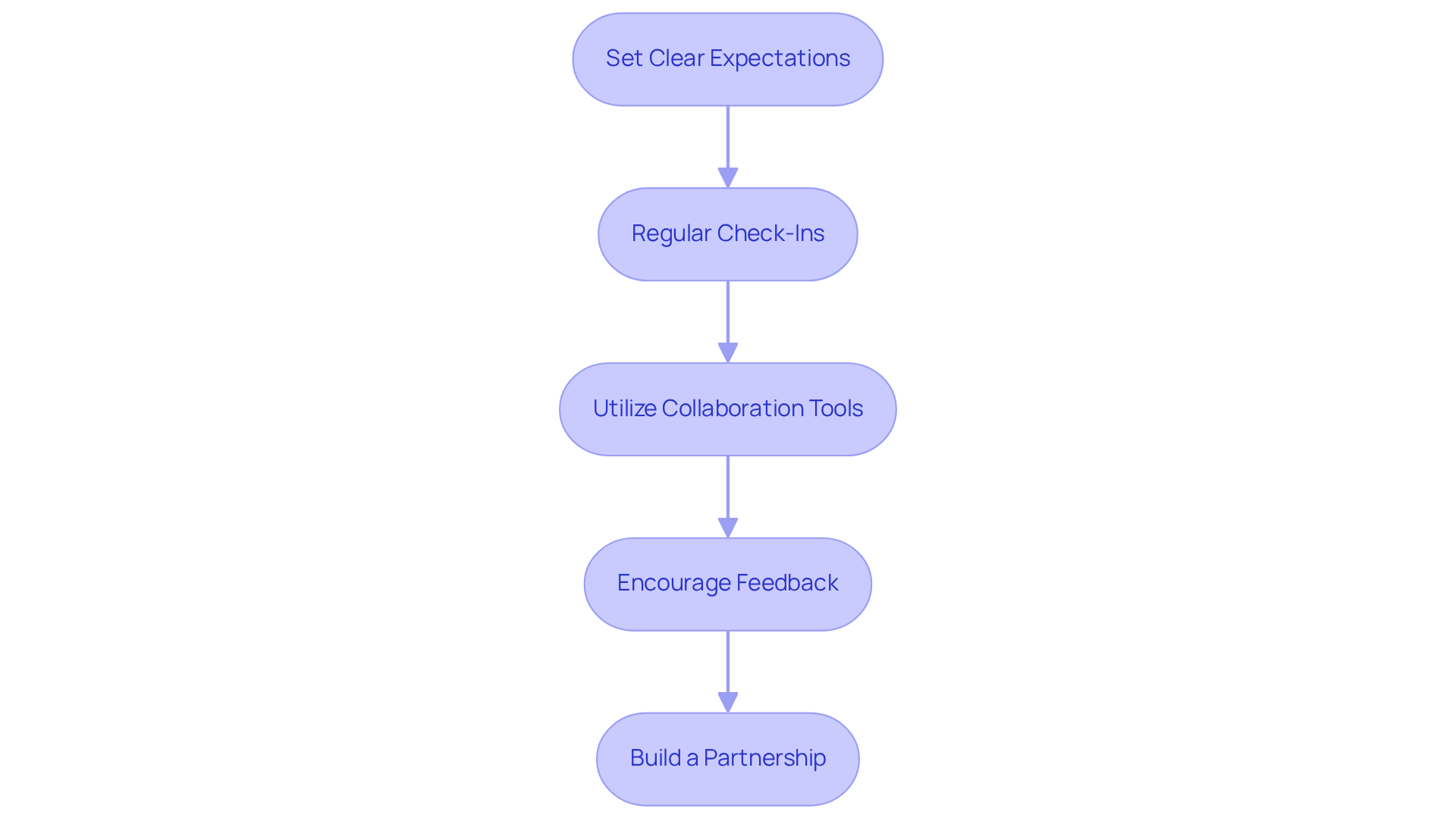
Establish Communication and Collaboration with Your EMS Provider
Choosing is just the beginning; are crucial for project success. Here are key steps to ensure a productive partnership:
- : At the project's outset, it is essential to clearly define goals, timelines, and deliverables. This alignment enables both parties to work towards a common objective.
- : Schedule consistent meetings to review project progress, address challenges, and make necessary adjustments. This practice fosters transparency and accountability, ensuring that all stakeholders remain informed.
- : Implement project management and to streamline communication and document sharing. These tools enhance efficiency and keep all stakeholders aligned.
- : Cultivate an environment where feedback is actively sought and welcomed. This approach promotes continuous improvement and allows for the timely resolution of concerns.
- : View your EMS provider as a partner rather than merely a vendor. This perspective encourages deeper collaboration and can lead to more innovative solutions, ultimately benefiting project outcomes.
Conclusion
Navigating the electronic manufacturing services landscape in France for medical devices necessitates a comprehensive understanding of market dynamics, regulatory compliance, and effective collaboration. With the increasing demand for medical equipment driven by technological advancements and an aging population, identifying the right EMS partner is crucial for success in this competitive environment.
This article outlines essential steps for engaging with EMS providers. It begins with:
- Defining specific manufacturing requirements.
- Evaluating potential partners based on their experience and compliance.
- Establishing effective communication strategies.
Key considerations include:
- Understanding regulatory frameworks such as CE marking and the Medical Device Regulation (MDR).
- Ensuring that technical capabilities align with project needs.
- Fostering a collaborative partnership that encourages innovation and transparency.
In conclusion, as the electronic manufacturing services sector in France evolves, it is imperative for stakeholders to remain informed about trends and best practices. By prioritizing thorough evaluations of EMS providers and maintaining open lines of communication, organizations can enhance their chances of successfully bringing high-quality medical devices to market. Embracing these strategies not only facilitates compliance with regulatory standards but also positions companies to thrive in a rapidly changing industry.
Frequently Asked Questions
What is the current market value of electronic manufacturing services in France?
The market for electronic manufacturing services in France was valued at approximately USD 22.38 billion in 2026.
What is the projected growth rate for the electronic manufacturing services market in France?
The market is projected to grow at a compound annual growth rate (CAGR) of 6.46% through 2035.
What factors are driving the growth of the electronic manufacturing services market in France?
The growth is primarily driven by the rising demand for medical equipment, particularly due to an aging population and advancements in technology.
Who are the key players in the electronic manufacturing services market in France?
Key players include established companies that offer a range of services, such as PCB assembly and system integration.
What specific expertise does Voler Systems offer in the electronic manufacturing services landscape?
Voler Systems specializes in creating ultra-low-power wearable sensors and AI-driven embedded systems, as well as embedded systems utilizing FPGA technology.
Why is it important to understand the regulatory environment in the electronic manufacturing services market?
Familiarizing oneself with the regulatory environment is critical for market entry, as compliance with EU regulations, including obtaining CE marking and adhering to the Medical Device Regulation (MDR), is necessary.
How can understanding key players and their capabilities benefit businesses in the electronic manufacturing services market?
Understanding these players and their capabilities can assist in identifying potential partners for medical equipment production requirements.
List of Sources
- Understand the Electronic Manufacturing Services Landscape in France
- France Electronics Manufacturing Services Market Size, Share & 2031 Growth Trends Report (https://mordorintelligence.com/industry-reports/france-electronics-manufacturing-services-market)
- France Electronic Manufacturing Services Market Size, Share | Growth Report 2035 (https://marketresearchfuture.com/reports/france-electronic-manufacturing-services-market-57562)
- France Medical Devices Market Size, Share | Growth [2032] (https://fortunebusinessinsights.com/france-medical-devices-market-114020)
- MedTech Market in France Hits €37B: Innovation and Global Ties Grow (https://med-tech.world/news/france-medtech-market-growth-2025)
- French MedTech Market: Key Challenges & Solutions (2026 Guide) | GrowthMedics (https://growthmedics.com/blogs-news/challenges-in-entering-french-medtech-market)
- Identify Your Medical Device Manufacturing Requirements
- What the FDA’s New Quality Requirements Mean for U.S. Medical Device Companies (https://amtivo.com/us/resources/insights/fdas-new-quality-requirements-mean-for-us-medical-device-companies)
- qualitymag.com (https://qualitymag.com/articles/99348-medical-device-regulation-in-the-us)
- New Harmonized Standards Added Under EU MDR (https://lne-gmed.com/news/new-harmonized-standards-added-under-eu-mdr)
- EU revised device rules rebalance compliance towards simplification and proportionality (https://osborneclarke.com/insights/eu-revised-device-rules-rebalance-compliance-towards-simplification-and-proportionality)
- Evaluate Potential EMS Providers Based on Key Criteria
- The medical device industry outlook for 2026 | Medical Economics (https://medicaleconomics.com/view/the-medical-device-industry-outlook-for-2026)
- Charting the Future: What lies ahead for medical device manufacturers in 2026 (https://dsm-firmenich.com/en/businesses/health-nutrition-care/segments/biomedical/about-biomedical/who-we-are/global-voices-local-impact/articles/2026-jan-paul-spencer-charting-the-future.html)
- 2026 Forecast: Medical manufacturing industry outlook (https://todaysmedicaldevelopments.com/article/2026-forecast-todays-medical-developments-industry-outlook)
- 10 Key MedTech Themes for 2026 - MedCity News (https://medcitynews.com/2026/01/10-key-medtech-themes-for-2026)
- 4 Medical Device Manufacturing Trends for 2026 (https://mastercontrol.com/gxp-lifeline/2026-medical-device-manufacturing-trends-life-sciences)
- Establish Communication and Collaboration with Your EMS Provider
- Communication Planning and Strategies for Aspiring and New EMS Leaders (https://jems.com/ems-management/leadership-foundations-for-aspiring-and-new-ems-leaders-2)
- Sarasota Herald-Tribune: Local News, Politics & Sports in Sarasota, FL (https://heraldtribune.com/press-release/story/132049/as-burnout-tops-ems-workforce-concerns-leadership-communication-training-gets-a-renewed-focus)
- Leadership Lessons from EMS Frontlines (https://jems.com/ems-management/leadership-lessons-from-ems-frontlines)
- Navigating Conflict Resolution in EMS Teams (https://jems.com/ems-management/navigating-conflict-resolution-in-ems-teams)
- As Burnout Tops EMS Workforce Concerns, Leadership Communication Training Gets a Renewed Focus (https://thenewsstar.com/press-release/story/21020/as-burnout-tops-ems-workforce-concerns-leadership-communication-training-gets-a-renewed-focus)





