Best Practices in Embedded System Software Development for Medical Devices
Discover essential best practices for embedded system software development in medical...

Navigating the complexities of the medical device industry requires a delicate balance between innovation and regulatory compliance. Manufacturers face significant challenges in balancing innovation with stringent regulatory compliance. Understanding best practices in embedded product engineering is essential for success. Companies must meet regulatory standards while leveraging cutting-edge technologies and collaborative strategies to enhance their product development processes. This article examines four pivotal practices that can enhance the engineering of medical devices, ensuring quality, safety, and market readiness.
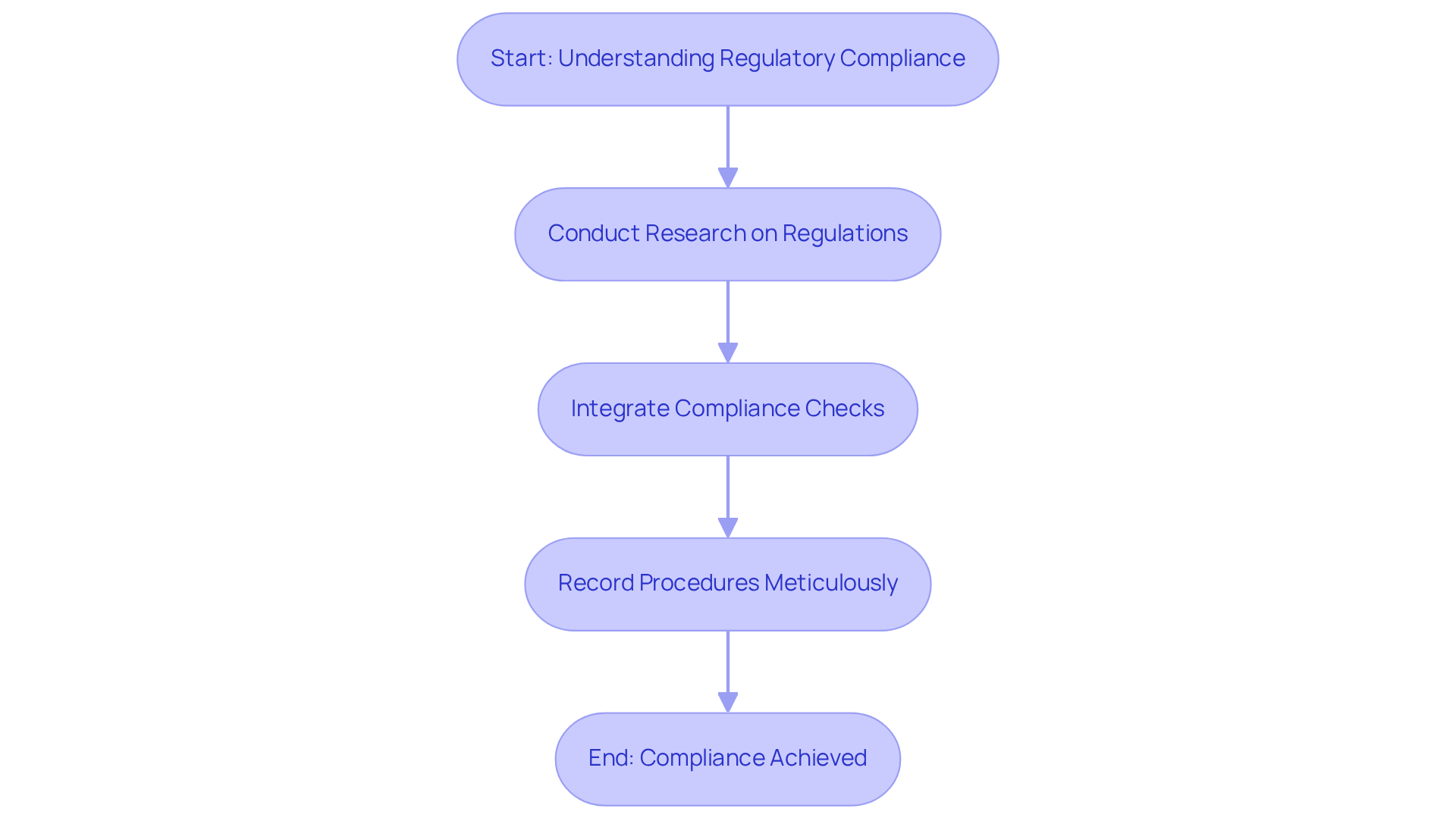
In the healthcare equipment sector, the stakes of regulatory compliance and quality standards are exceptionally high, directly impacting patient safety and product effectiveness. Essential regulations like ISO 13485, which oversees quality management systems, and IEC 62304, which details software lifecycle guidelines, are vital in directing the advancement of medical equipment. These standards not only help in maintaining safety but also streamline the path to market approval.
To effectively implement these standards, manufacturers should:
Recent data indicates that approximately 70% of medical device firms achieve compliance with IEC 62304 standards, demonstrating the critical role of structured compliance strategies. For instance, organizations that align their development methods with these standards often experience quicker time-to-market and a reduced risk of non-compliance penalties. Regulatory expert Julian Russell emphasizes that "maintaining compliance is not merely a checkbox exercise but a vital component of product development that enhances overall quality and safety." Furthermore, companies that have successfully navigated FDA approvals by aligning their development processes with these standards have reported significant improvements in their compliance outcomes. Neglecting these standards can lead to severe repercussions, jeopardizing market access and undermining patient trust and safety.
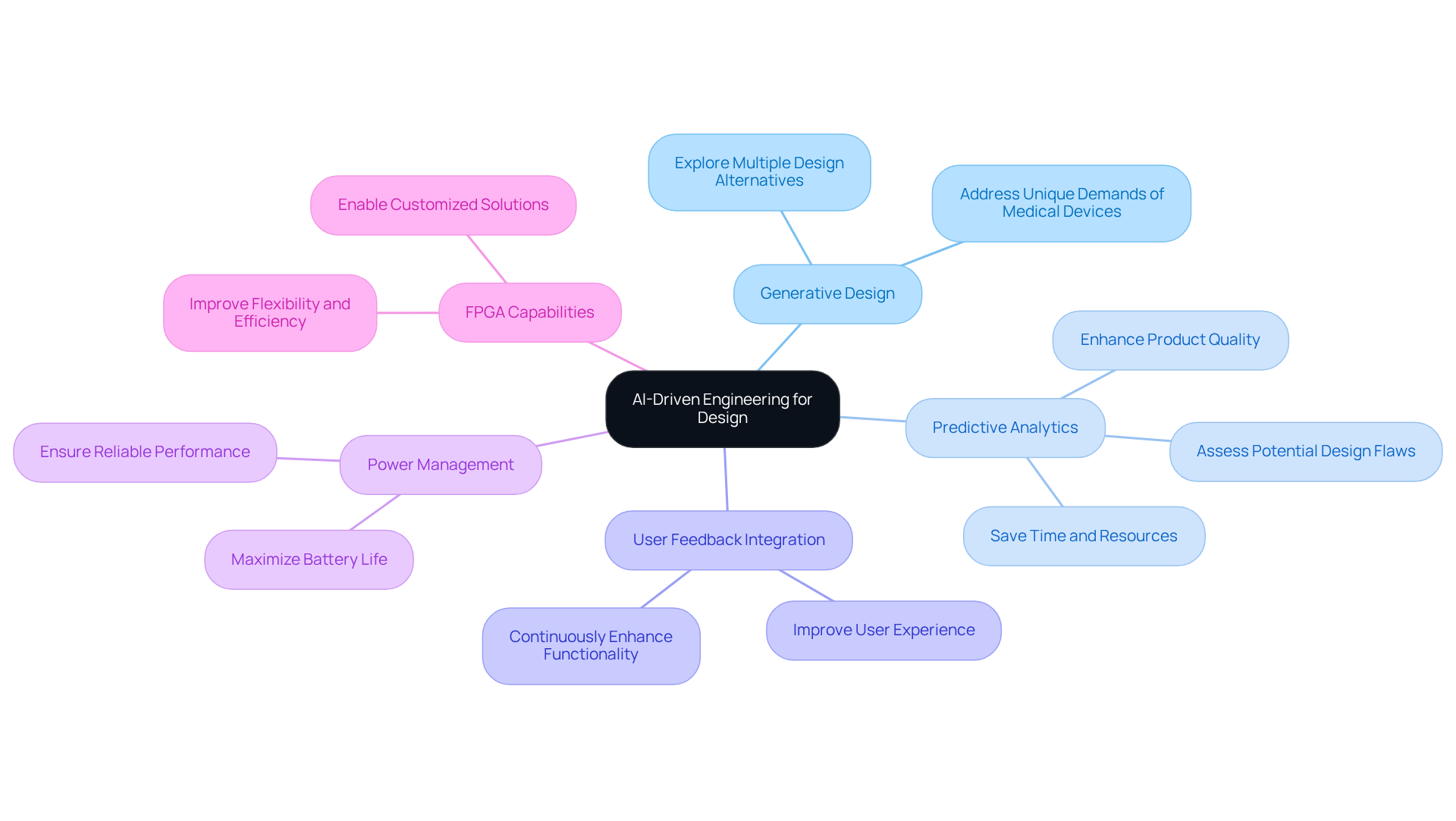
The integration of AI-driven engineering is revolutionizing the development of healthcare instruments, enabling faster iterations and informed decision-making. Engineers at Voler Systems enhance their designs through machine learning algorithms and data analytics, leveraging real-world data and predictive modeling to ensure that products meet the highest standards of performance and reliability.
To effectively leverage AI in your design process:
For example, companies that have incorporated AI into their creation processes, such as Voler Systems, report substantial decreases in development time and expenses, along with improved product performance and user satisfaction. This strategic integration not only enhances efficiency but also positions companies like Voler Systems at the forefront of innovation in healthcare technology.
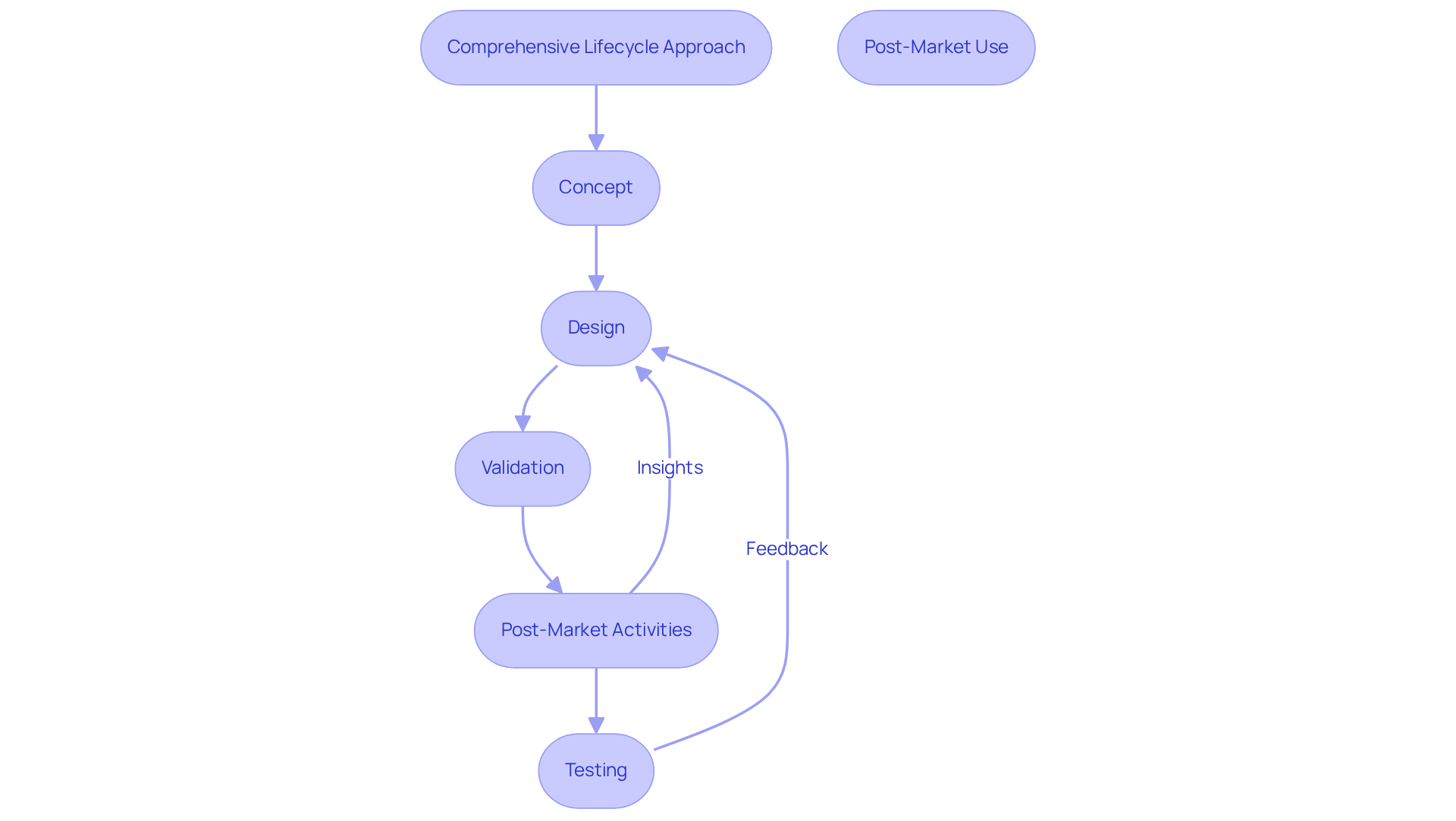
A comprehensive lifecycle method for medical equipment development is crucial for maintaining compliance and effectiveness throughout the product's lifespan. This approach encompasses all phases, including creation, manufacturing, testing, and post-market monitoring.
To implement this approach:
Companies that adopt a lifecycle approach often see improved product quality and reduced time to market, as they can quickly adapt to regulatory changes and user feedback. For instance, organizations that integrate post-market surveillance into their lifecycle management have reported significant enhancements in product performance and safety, demonstrating the value of a proactive compliance strategy. The FDA's authorization of 3,238 510(k)s in 2025 underscores the increasing complexity and regulatory scrutiny in the industry. As René Zölfl, a Global Industry Advisor for MedTech at PTC, observes, 'Integrating quality and regulatory procedures into digital workflows can change compliance from a burden into a strategic advantage.' Furthermore, companies that have successfully implemented lifecycle management practices, such as those highlighted in case studies, have shown marked improvements in their product quality metrics and overall market responsiveness. However, it is crucial to recognize and address potential pitfalls, such as inadequate documentation or failure to adapt to feedback, which can undermine the effectiveness of a lifecycle approach.
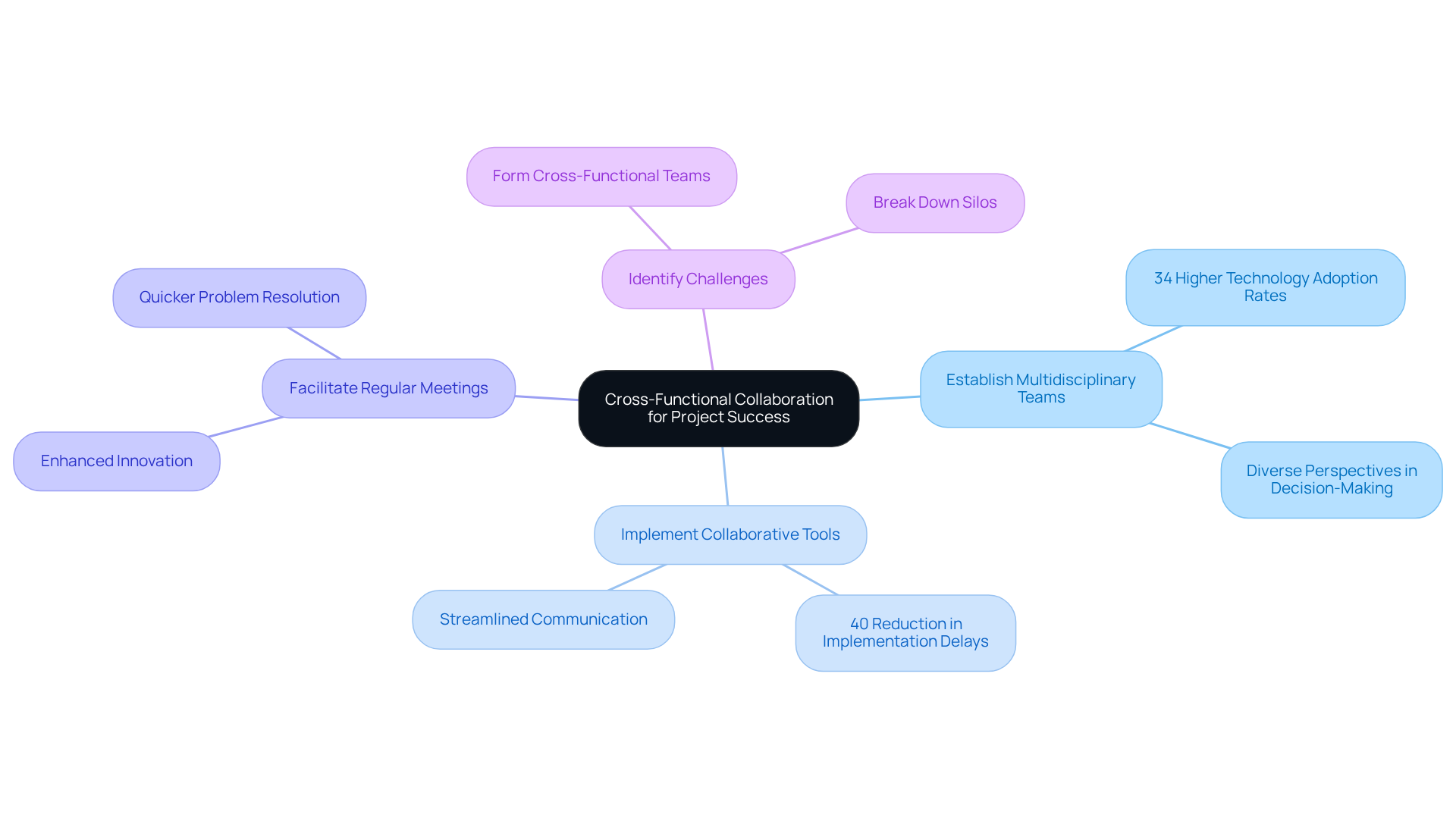
Cross-functional collaboration is essential in the healthcare equipment sector, where successful projects depend on the integration of insights from engineering, regulatory, quality assurance, and marketing teams. Effective collaboration is a catalyst for innovative solutions and significantly improves project outcomes.
To cultivate a collaborative environment:
Industry leaders emphasize that the journey toward effective collaboration begins with identifying pressing challenges and forming cross-functional teams to tackle them. By breaking down silos and fostering a culture of curiosity and cooperation, medical device manufacturers can navigate the complexities of product development more effectively.
Neglecting best practices in embedded product engineering for medical devices can jeopardize safety and compliance. The core message highlights four critical elements:
By prioritizing these elements, manufacturers can enhance product quality and accelerate the development process, ultimately benefiting patient care.
Understanding and implementing regulatory compliance and quality standards is foundational, as it safeguards patient safety and expedites market approval. Leveraging AI-driven engineering allows for more efficient design processes and improved product performance. Additionally, adopting a lifecycle management approach ensures that products remain effective throughout their lifespan, while cross-functional collaboration fosters innovation and enhances project outcomes.
The evolving medical device landscape necessitates a commitment to these best practices. By committing to regulatory compliance, utilizing advanced technologies, and fostering teamwork, organizations can navigate the complexities of product development more effectively. Ultimately, organizations that prioritize these practices will not only excel in product development but also significantly impact patient health outcomes.
Why is regulatory compliance important in the healthcare equipment sector?
Regulatory compliance is crucial in the healthcare equipment sector as it directly impacts patient safety and product effectiveness. Adhering to regulations ensures that medical devices are safe and effective for use.
What are some essential regulations in the healthcare equipment sector?
Key regulations include ISO 13485, which oversees quality management systems, and IEC 62304, which provides guidelines for software lifecycle management. These standards help maintain safety and facilitate market approval.
How can manufacturers effectively implement regulatory standards?
Manufacturers can implement regulatory standards by conducting thorough research on applicable regulations early in the design phase, integrating compliance checks throughout the development lifecycle, and meticulously recording all procedures to aid in compliance and transparency.
What percentage of medical device firms achieve compliance with IEC 62304 standards?
Approximately 70% of medical device firms achieve compliance with IEC 62304 standards, highlighting the importance of structured compliance strategies.
What benefits do organizations experience by aligning their development methods with regulatory standards?
Organizations that align their development methods with regulatory standards often experience quicker time-to-market and a reduced risk of non-compliance penalties.
What are the consequences of neglecting regulatory standards in medical device development?
Neglecting regulatory standards can lead to severe repercussions, including jeopardized market access and undermined patient trust and safety.
