Best Practices for Connected Product Development in Medical Devices
Discover best practices for effective connected product development in medical devices.

In product design and development, navigating complexities is essential for delivering innovative solutions. This article explores five best practices employed by leading firms that enhance collaboration while ensuring compliance and user satisfaction. Teams often face difficulties in aligning diverse perspectives, which can hinder effective communication.
How can organizations optimize processes to ensure successful product delivery?
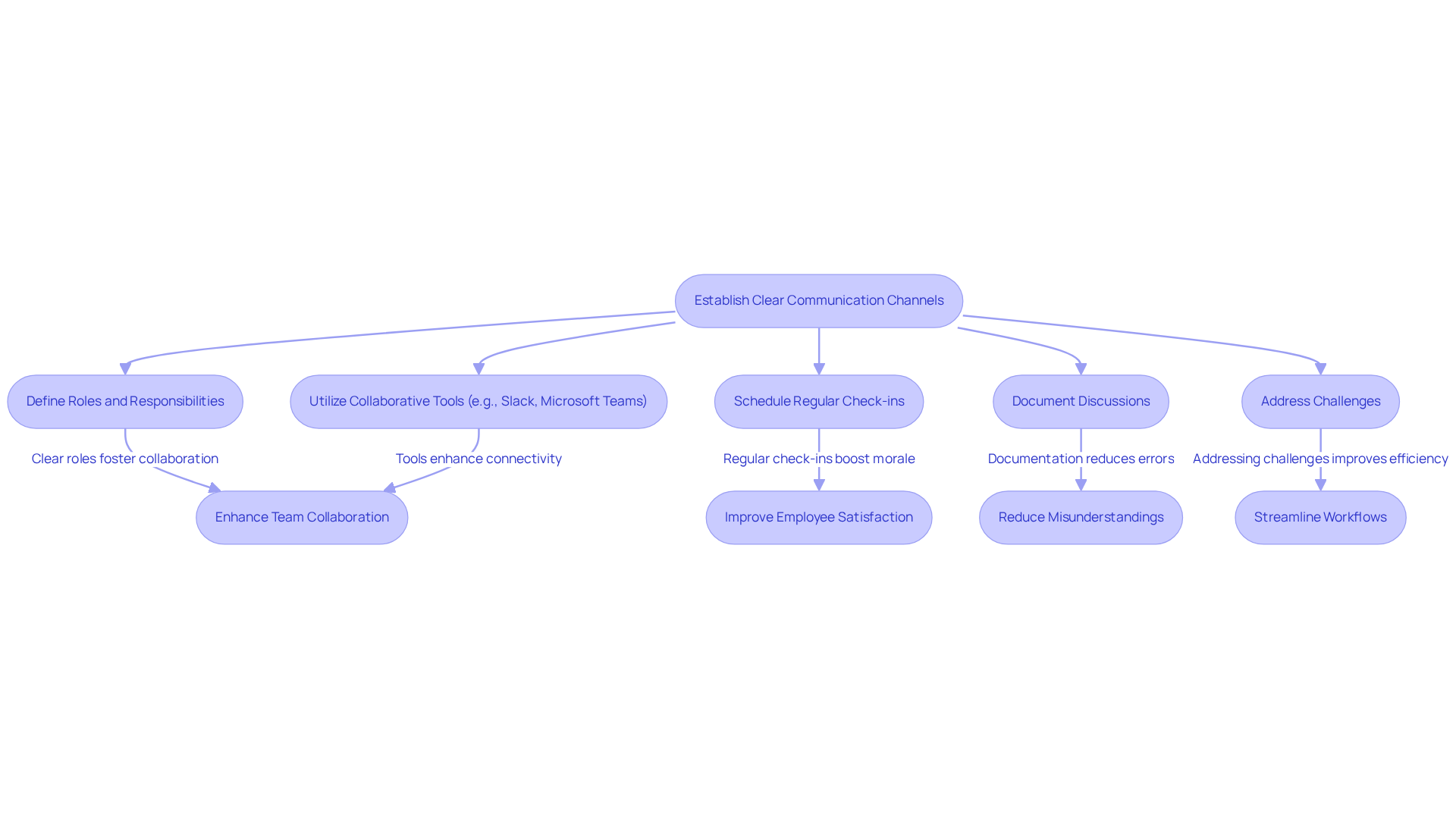
Defining clear roles and responsibilities is fundamental to establishing effective communication channels within teams. Collaborative tools such as Slack and Microsoft Teams facilitate real-time communication, enabling teams to remain connected and responsive. Seventy-five percent of employees use enterprise chat tools for communication, underscoring their critical role in healthcare technology development.
Regular check-ins and updates should be scheduled to ensure alignment and promptly address any emerging issues. Documenting discussions and decisions in a shared space fosters transparency and accountability among team members. For instance, a medical device development team might utilize a management tool to track progress and share updates, ensuring that all stakeholders are informed of any changes or challenges.
This structured approach enhances collaboration and significantly boosts success rates; studies show that effective communication can increase employee productivity by up to 25%. It is crucial to recognize potential pitfalls, as constant connectivity to digital communication tools can lead to heightened stress levels among employees. By addressing these challenges, teams can reduce misunderstandings and streamline workflows, leading to improved outcomes.
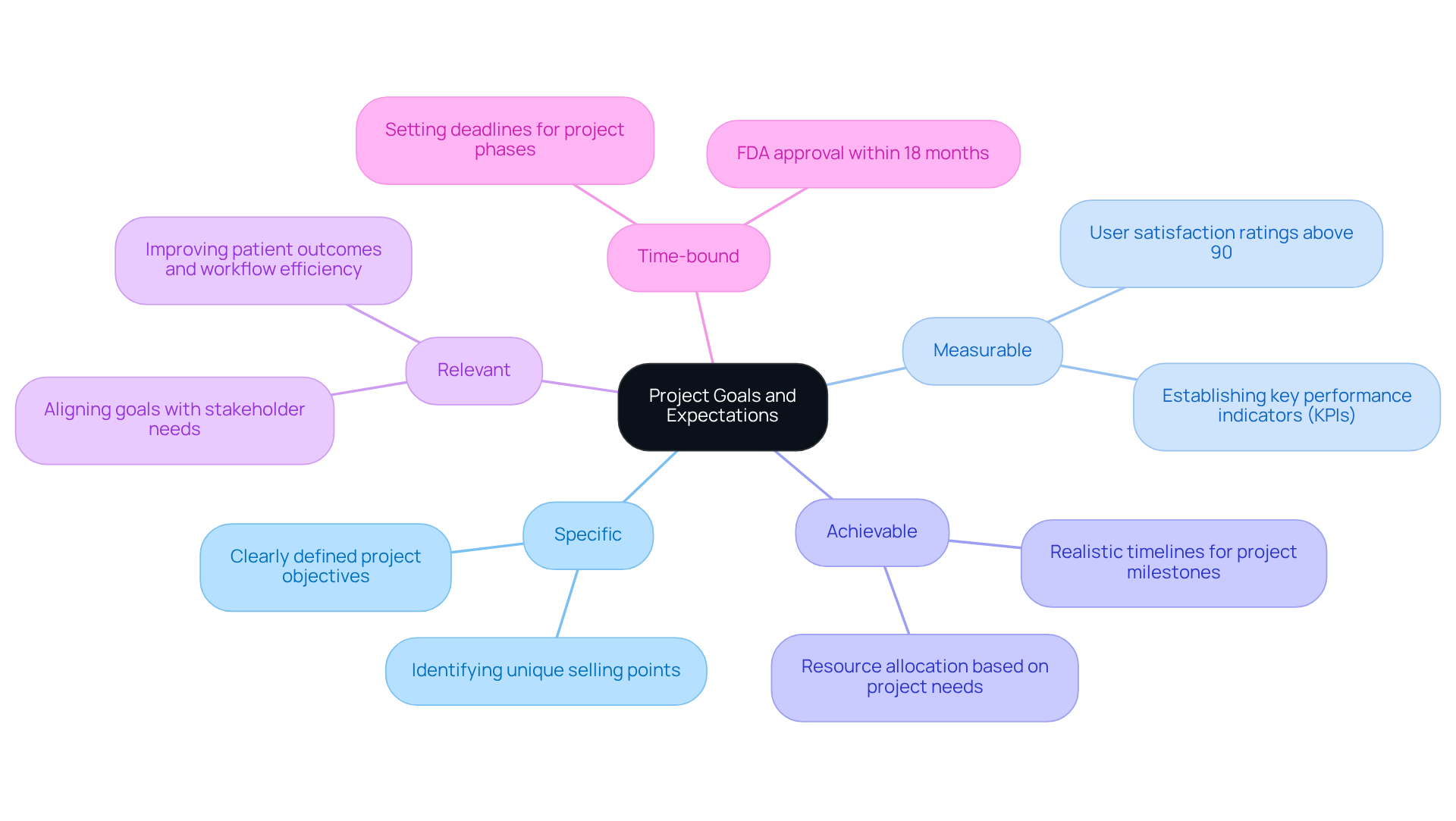
Engaging stakeholders from the outset is crucial for gathering valuable feedback on project goals. Utilizing the SMART criteria - Specific, Measurable, Achievable, Relevant, and Time-bound - ensures that goals are clearly defined. For instance, an initiative focused on developing a wearable health monitor could aim for FDA approval within 18 months while targeting user satisfaction ratings above 90%. Consistently revisiting these objectives during meetings allows teams to evaluate progress and make essential adjustments, fostering accountability and continuous improvement. This approach not only aligns project expectations with stakeholder needs but also significantly influences the success of healthcare technology initiatives.
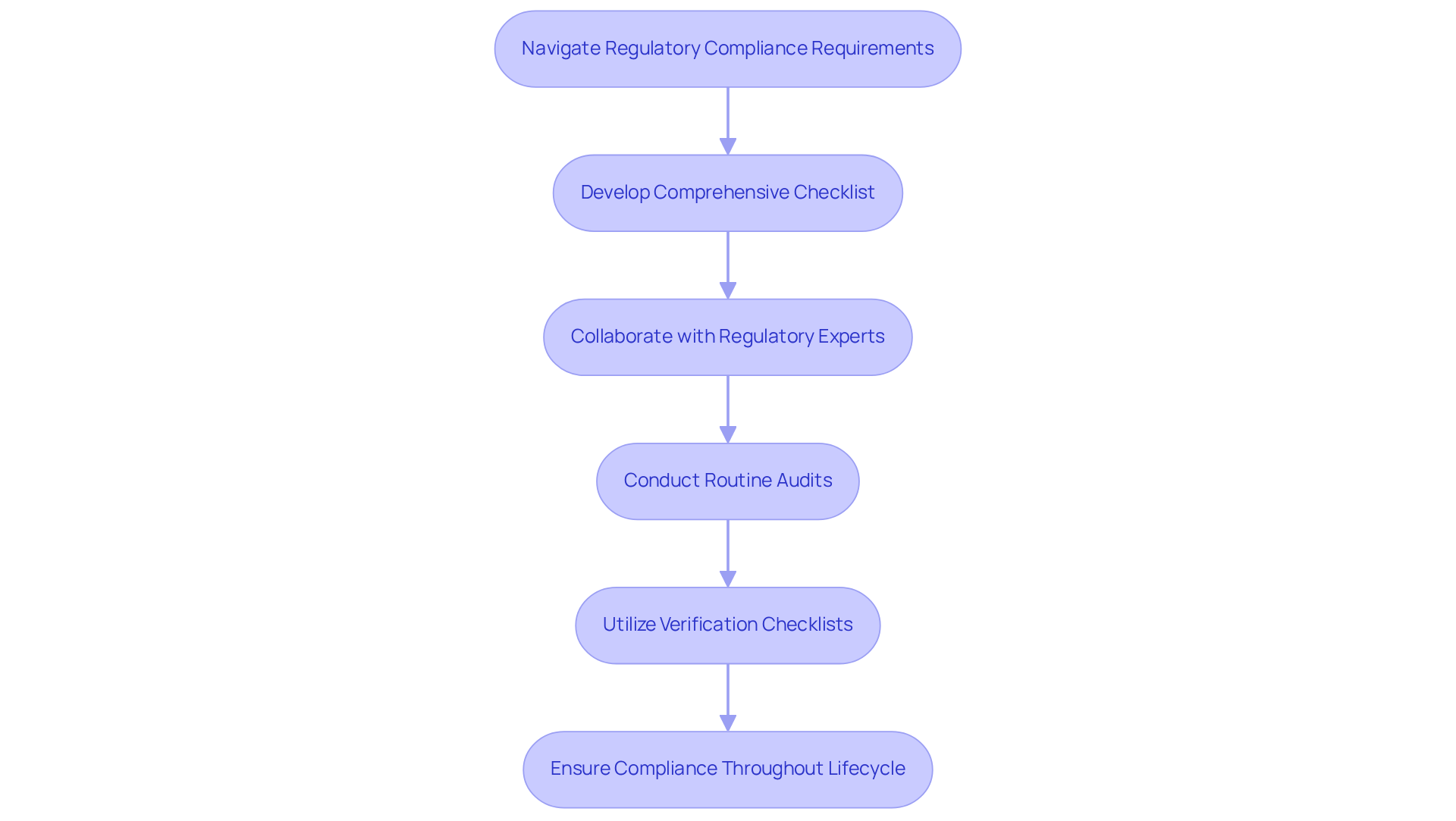
Navigating the regulatory landscape for healthcare instruments requires a thorough understanding of applicable authorities and standards. Start by developing a comprehensive checklist that specifies the necessary documentation, testing protocols, and approvals required for FDA submission. For instance, ensure that all design specifications adhere to FDA guidelines, which have been updated to clarify the distinction between general wellness products and regulated medical devices as of January 2026.
Collaboration with regulatory experts from the outset of the design process is crucial. Involving these experts can assist in recognizing possible regulatory issues early, enabling proactive solutions. Routine audits and assessments of adherence status are also essential; they guarantee that the project stays aligned with regulatory expectations and can adjust to any changes in guidelines.
Manufacturers using organized verification checklists reduce regulatory scrutiny risks. For instance, companies that follow a thorough regulatory framework report a 95% success rate in meeting FDA requirements on their first submission. This highlights the importance of thorough preparation and ongoing adherence management.
Efficient adherence checklists utilized in medical equipment projects frequently encompass sections for risk evaluation, design validation, and post-market monitoring plans. These components are essential for guaranteeing that the apparatus not only satisfies initial regulatory standards but also upholds adherence throughout its lifecycle.
Engaging regulatory experts can significantly strengthen compliance strategies. As noted by industry leaders, maintaining transparency in documentation and aligning product claims with regulatory definitions are best practices that can mitigate risks associated with misclassification and liability claims. By adhering to these guidelines, manufacturers can simplify their route to FDA approval and guarantee the safety and efficacy of their healthcare products. By prioritizing compliance, manufacturers not only enhance their chances of approval but also safeguard their reputation and market position.
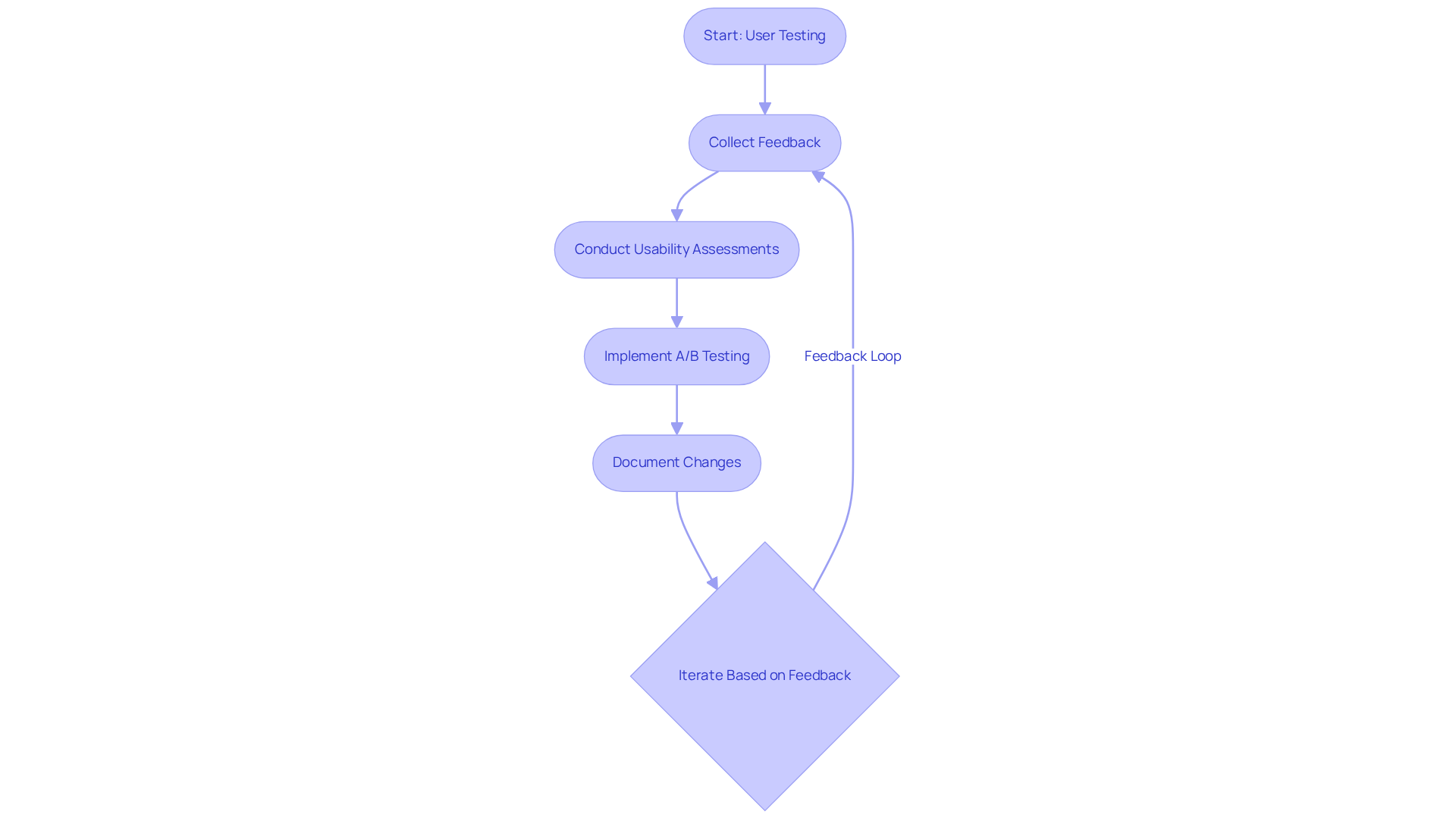
Regular user testing is crucial for a product design and development firm to optimize healthcare products and ensure user satisfaction. Integrating regular user testing sessions throughout the design process allows for the collection of valuable feedback on prototypes.
For example, during the development of a new insulin pump, usability assessments with diabetic individuals can reveal significant pain points, enabling designers to enhance the interface effectively.
Using A/B testing allows teams to evaluate design options effectively, leading to informed decisions that improve usability.
It is essential to document all feedback and subsequent changes meticulously, as this practice not only tracks progress but also fosters a culture of continuous improvement.
Involving users from the beginning ensures the final product meets their needs and expectations, resulting in safer and more intuitive healthcare tools.
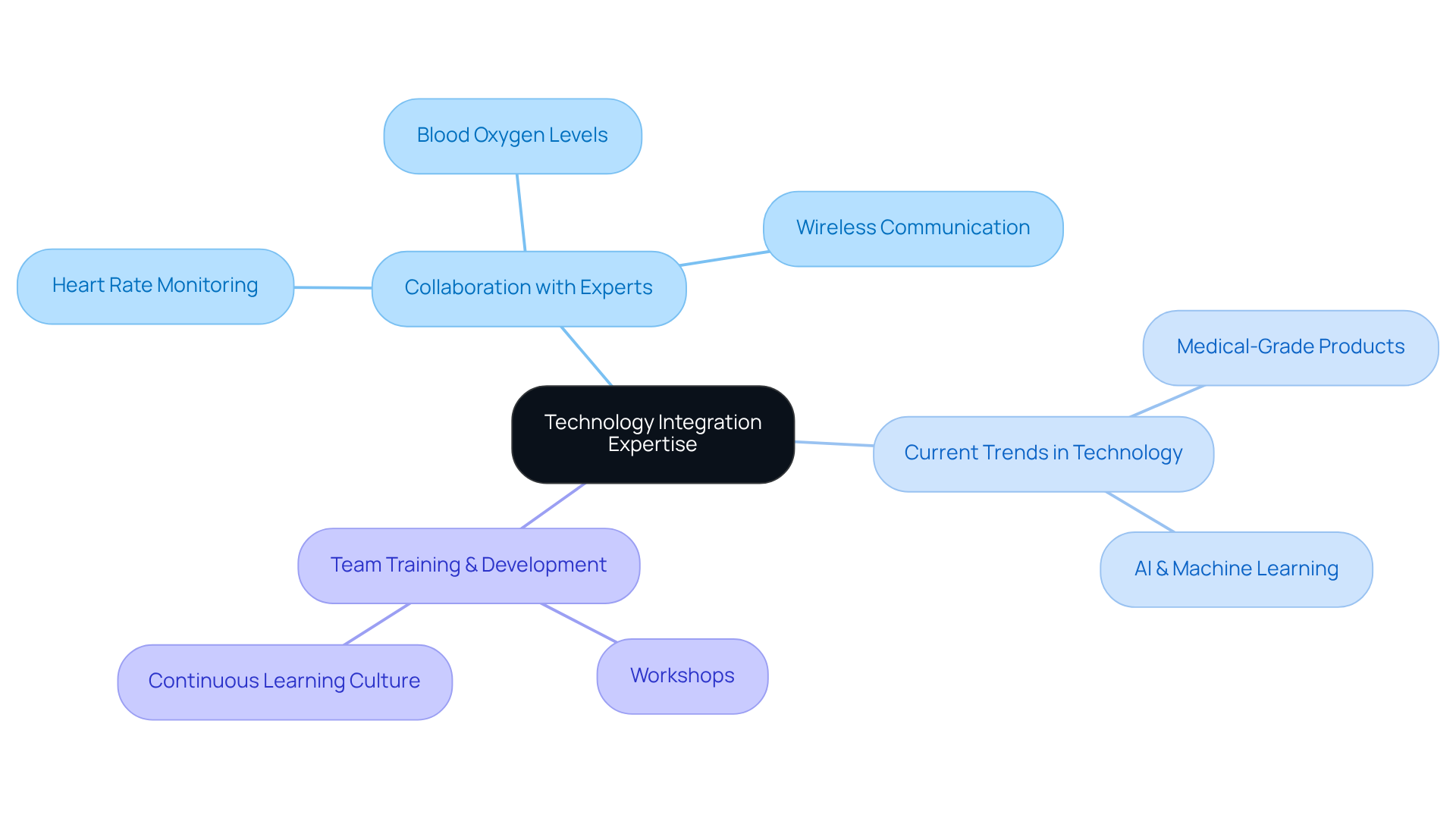
Collaborating with technical experts is essential for integrating advanced technologies into your product design, ensuring optimal functionality and user experience. For instance, when developing a wearable fitness tracker, incorporating advanced sensors such as those for heart rate monitoring, blood oxygen levels, and continuous glucose tracking can significantly improve health monitoring accuracy. Furthermore, utilizing wireless communication technologies guarantees efficient data transmission to users and healthcare providers.
Staying abreast of current trends in sensor technology is essential for maintaining a competitive edge. The market is experiencing a transition towards medical-grade consumer products that provide clinical accuracy, blurring the lines between consumer wellness and medical technology. This evolution is driven by advancements in AI and machine learning, which enable predictive health insights and personalized recommendations.
Regular training and workshops for your team can enhance their understanding of these emerging technologies and their applications in a product design and development firm. Fostering a culture of continuous learning enables your organization to adapt effectively to rapid advancements in health monitoring devices, ultimately leading to innovative solutions that meet the evolving needs of users and healthcare professionals.
Without best practices in product design and development, healthcare technologies risk falling short of user needs and regulatory standards. Establishing these practices is crucial for creating successful solutions. Clear communication, defined project goals, regulatory compliance, iterative feedback, and technology integration are essential for teams to enhance their efficiency and effectiveness in delivering innovative solutions.
Key insights from the article highlight the importance of:
Leveraging technical expertise ensures products are functional and incorporate the latest technological advancements, leading to improved user experiences.
In a rapidly evolving industry, it is essential for product design and development teams to adopt these best practices to remain competitive. The failure to embrace these strategies could hinder the development of safer and more effective healthcare solutions. By prioritizing communication, collaboration, and continuous improvement, organizations can enhance their product offerings and contribute to the overall advancement of healthcare technology.
What is the importance of establishing clear communication channels within teams?
Establishing clear communication channels is fundamental for defining roles and responsibilities, enhancing collaboration, and ensuring teams remain connected and responsive.
What tools are recommended for effective communication in teams?
Collaborative tools such as Slack and Microsoft Teams are recommended as they facilitate real-time communication among team members.
How prevalent is the use of enterprise chat tools among employees?
Seventy-five percent of employees use enterprise chat tools for communication, highlighting their critical role in healthcare technology development.
What practices can enhance communication and collaboration within teams?
Scheduling regular check-ins and updates, documenting discussions in a shared space, and utilizing management tools to track progress can enhance communication and collaboration.
How does effective communication impact employee productivity?
Effective communication can increase employee productivity by up to 25%.
What potential challenges should teams be aware of regarding digital communication tools?
Constant connectivity to digital communication tools can lead to heightened stress levels among employees, which teams should address to reduce misunderstandings and streamline workflows.
Why is it important to define project goals and expectations?
Defining project goals and expectations is crucial for engaging stakeholders, gathering feedback, and aligning project objectives with stakeholder needs, which significantly influences the success of healthcare technology initiatives.
What criteria should be used to ensure project goals are clearly defined?
The SMART criteria - Specific, Measurable, Achievable, Relevant, and Time-bound - should be used to clearly define project goals.
How can teams ensure they are making progress towards their project goals?
Consistently revisiting project objectives during meetings allows teams to evaluate progress and make necessary adjustments, fostering accountability and continuous improvement.
