Why Medical Device Manufacturers Need an Embedded Design Company
Introduction The world of medical device manufacturing is characterized by a relentless...
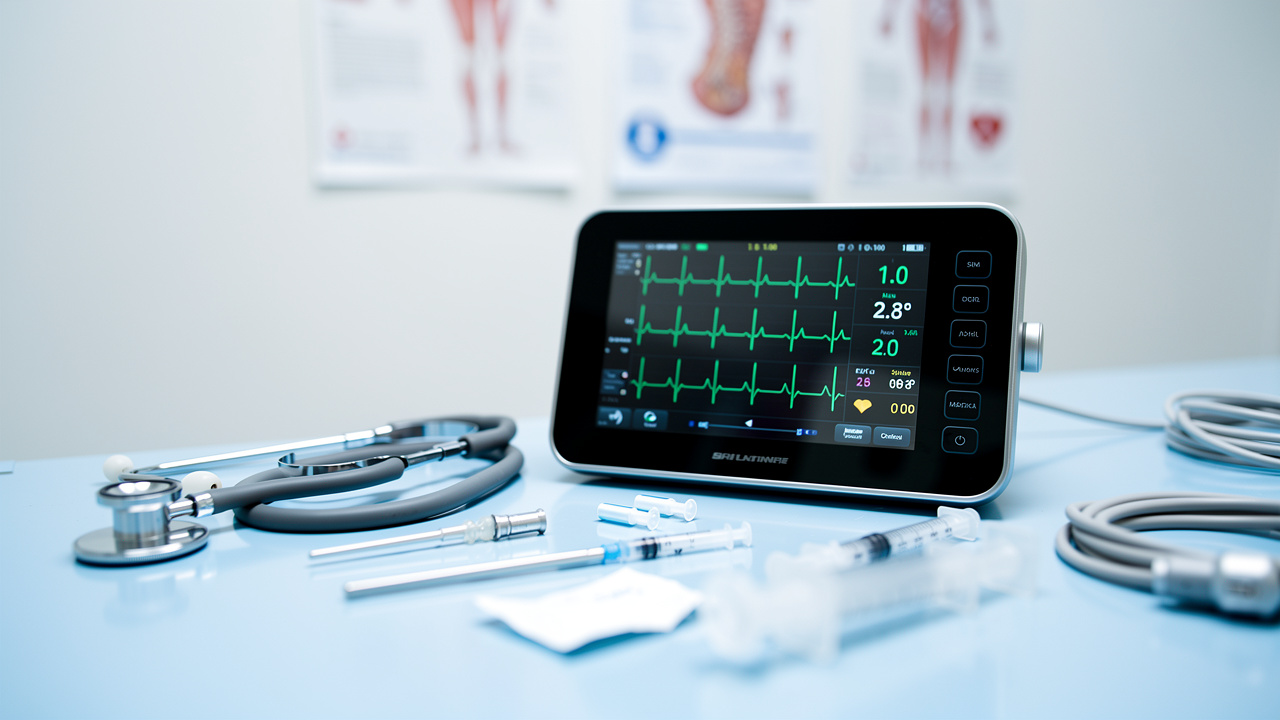
Manufacturers of medical devices face significant challenges in balancing regulatory compliance with the need for innovation in patient care. The integration of embedded systems represents a significant advancement, offering capabilities that enhance reliability and efficiency while navigating the complexities of compliance. However, as the landscape of medical technology evolves, how can manufacturers effectively utilize these systems to foster innovation and maintain a competitive edge? This article explores the essential role of embedded development companies in overcoming these challenges and highlights the advantages of strategic partnerships in developing superior medical devices.
Manufacturers of medical equipment face increasing demands for products that ensure both reliability and efficiency. Embedded systems developed by an embedded development company play a vital role in meeting these demands by integrating advanced sensors and real-time processing capabilities that enable equipment to monitor and respond to patient needs effectively. For instance, a cardiac monitor designed by an embedded development company can continuously track heart rhythms and alert healthcare providers to any anomalies, significantly enhancing patient safety.
Optimized power management and streamlined data processing further enhance the efficiency of these tools. This is particularly crucial in wearable technology, such as those developed by Voler Systems, including heart pumps and liquid biopsy platforms, where battery life is a key consideration. Utilizing ultra-low power technology allows producers to create devices with extended battery life, improving user experience and compliance. The synergy of reliability and efficiency not only satisfies regulatory standards but also builds trust among users and healthcare professionals. Ultimately, the integration of advanced technology in medical devices is essential for improving patient care and outcomes.
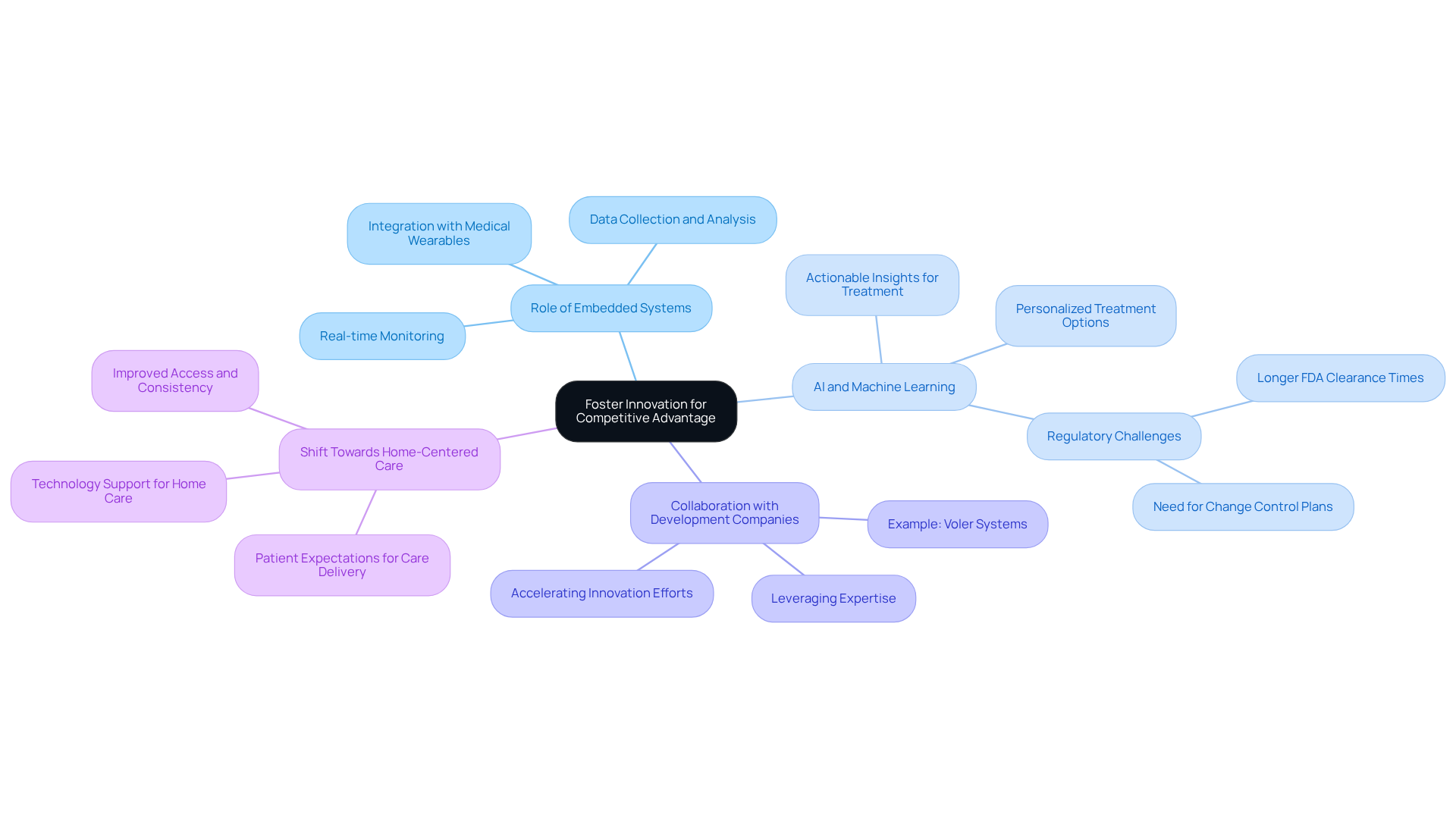
Producers of medical devices face daunting challenges in navigating the complex regulatory landscape, particularly with the stringent requirements set forth by the FDA. The 2026 guidance emphasizes the need for rigorous compliance, necessitating extensive documentation and testing to ensure both safety and efficacy. An embedded development company plays a crucial role by providing expert guidance that helps manufacturers navigate and comply with complex regulations.
For instance, this firm specializes in designing embedded systems that prioritize compliance from the outset, integrating features such as sensor and actuator compatibility that facilitate testing and validation. Innovative technologies like FPGA and AI help optimize battery life, a critical factor for wireless medical devices. This proactive strategy not only streamlines the approval process but also mitigates the risk of costly delays associated with non-compliance.
Furthermore, the knowledge of Voler Systems guarantees that producers stay updated on changing regulatory standards, including the New Inspection CP, which highlights risk management throughout the product lifecycle. This enables manufacturers to focus on product development while effectively managing regulatory risks. By prioritizing compliance, manufacturers not only safeguard their products but also enhance their competitive edge in the market.
In the medical equipment sector, the challenge of balancing cost efficiency with quality assurance is increasingly pressing. Streamlining design processes and utilizing advanced manufacturing techniques allows companies to minimize waste. This approach also reduces time-to-market.
An example of this approach is seen in their innovative electronic product design projects, particularly in the medical, wearable, and IoT sectors. Their expertise ensures that devices not only meet functional requirements but also comply with industry standards, including emissions and ESD requirements.
Additionally, the use of simulation tools during the design phase can identify potential issues early, reducing the need for costly revisions later in the development process. Aditi Basu, Marketing Head at Future Market Insights, emphasizes that "Automation provides many benefits, including cleanliness, speed, and control over production processes," which underscores the importance of integrating automation into manufacturing practices.
Moreover, implementing Just-in-Time (JIT) inventory systems can further enhance efficiency by reducing storage costs and minimizing waste. Collaborating with the company enables producers to leverage their commitment to quality and timely delivery, enhancing profitability. Given the critical shortages in automation and digital manufacturing expertise, partnering with specialized firms like Voler Systems becomes increasingly vital for navigating these challenges and achieving operational excellence. As the landscape evolves, those who adapt through strategic partnerships will likely emerge as leaders in the industry.
In a rapidly evolving technological landscape, medical equipment manufacturers must innovate to maintain their market position. Embedded systems play a crucial role in this innovation, facilitating the creation of advanced, interconnected tools that improve patient care.
The integration of AI and machine learning within embedded systems allows for real-time data analysis and decision-making, resulting in personalized treatment options tailored to individual patient needs. This capability enhances functionality and improves user experience through continuous data collection and analysis. For instance, AI-driven medical devices can provide actionable insights that support earlier and more precise treatment, leading to improved patient outcomes and satisfaction.
Collaborating with an embedded development company like Voler Systems allows manufacturers to leverage expertise and resources, accelerating innovation efforts. This collaboration not only enhances product offerings but also positions manufacturers as leaders in the evolving healthcare landscape, meeting the growing demand for connected health solutions and value-based care models. Furthermore, the shift towards home-centered care by 2026 underscores the necessity for innovative solutions that address patients' changing needs.
The collaboration between medical device manufacturers and embedded development companies is essential for achieving reliability and compliance in a competitive market. By leveraging the expertise of specialized firms, manufacturers can meet regulatory standards while ensuring superior performance and patient safety.
Key insights from the discussion highlight the role of advanced embedded systems in optimizing power management, streamlining production processes, and fostering innovation. These elements are crucial in creating medical devices that are effective, cost-efficient, and compliant with evolving regulations. Collaborating with embedded development experts facilitates a proactive approach to navigating the complex landscape of medical device manufacturing, ultimately leading to improved patient outcomes.
As the medical device industry evolves, partnerships with embedded development companies are essential. This collaboration enhances product offerings and positions manufacturers to adapt to changing healthcare needs and technological advancements. By prioritizing innovation and compliance, manufacturers will not only enhance their product offerings but also ensure they meet the evolving demands of the healthcare sector.
What are the main demands faced by manufacturers of medical equipment?
Manufacturers of medical equipment face increasing demands for products that ensure both reliability and efficiency.
How do embedded systems contribute to medical device design?
Embedded systems integrate advanced sensors and real-time processing capabilities, enabling medical equipment to monitor and respond to patient needs effectively.
Can you provide an example of how embedded systems enhance patient safety?
A cardiac monitor designed by an embedded development company can continuously track heart rhythms and alert healthcare providers to any anomalies, significantly enhancing patient safety.
Why is power management important in medical devices?
Optimized power management is crucial for enhancing the efficiency of medical devices, particularly in wearable technology where battery life is a key consideration.
How does Voler Systems contribute to wearable technology?
Voler Systems develops wearable technology, including heart pumps and liquid biopsy platforms, utilizing ultra-low power technology to create devices with extended battery life.
What is the impact of integrating advanced technology in medical devices?
The integration of advanced technology in medical devices is essential for improving patient care and outcomes, satisfying regulatory standards, and building trust among users and healthcare professionals.
